Abundancia y diversidad de semillas dispersadas por el conejo castellano (Sylvilagus floridanus) en una reserva urbana de la Ciudad de México
Yury Glebskiy a, b, *, Zenón Cano-Santana a
a Universidad Nacional Autónoma de México, Facultad de Ciencias, Departamento de Ecología y Recursos Naturales, Laboratorio de Interacciones y Procesos Ecológicos, Circuito Exterior s/n, Ciudad Universitaria, 04510 Ciudad de México, Mexico
b Universidad Nacional Autónoma de México, Posgrado en Ciencias Biológicas, Facultad de Ciencias, Circuito Exterior s/n, Ciudad Universitaria, 04510 Ciudad de México, Mexico
*Corresponding author: agloti@ciencias.unam.mx (Y. Glebskiy)
Received: 05 February 2025; accepted: 07 October 2025
Abstract
Lagomorphs are potentially important but understudied seed dispersers. In particular, there are no studies about the role of the Eastern Cottontail as a seed disperser. Therefore, the aim of this article is to evaluate the abundance and diversity of seeds dispersed by Sylvilagus floridanus in a conservation reserve within Mexico City. We collected fecal pellets during 2 periods, the highest seed production and the start of seed germination in the Reserva Ecológica del Pedregal de San Ángel. Pellets were subjected to different treatments to end seed dormancy (left in the field, stored in laboratory, and no treatment) and put to germinate in germination chambers and the field. We found that pellets left in the field had the most seeds germinating. Cottontails dispersed up to 0.77 seeds/g of excrete. A total of 15 species were observed, but the Chao estimator suggested that there were 29.8 species. Many of the dispersed seeds belong to rare species, most noticeable Jaegeria hirta. We conclude that cottontails are important dispersers due to the amount of excretes produced and the identity of the plants they disperse.
Keywords: Dormancy; Germination; Jaegeria hirta; Opuntia tomentosa;REPSA
Resumen
Los lagomorfos son dispersores de semillas potencialmente importantes, pero subestudiados. En particular, no existen estudios sobre el papel del conejo castellano como dispersor de semillas; por tanto, el objetivo de este artículo fue evaluar la abundancia y diversidad de semillas dispersadas por Sylvilagus floridanus en una reserva dentro de la Ciudad de México. Colectamos pastillas fecales durante 2 periodos, la temporada de mayor producción de semillas y el inicio de la germinación en la Reserva Ecológica del Pedregal de San Ángel. Las pastillas se sometieron a distintos tratamientos para terminar la dormancia (almacenar en el campo, en laboratorio y sin tratamiento) y fueron puestas a germinar en cámaras y en el campo. Encontramos que las excretas almacenadas en el campo tuvieron más semillas germinadas. Los conejos dispersaron hasta 0.77 semillas/g de excreta. Observamos 15 especies, pero el estimador de Chao sugirió que hubo 29.8 especies dispersadas. Muchas especies dispersadas son raras, en particular Jaegeria hirta. Concluimos que los conejos son importantes dispersores por la cantidad de excretas que producen y la identidad de las plantas diseminadas.
Palabras clave: Dormancia; Germinación; Jaegeria hirta; Opuntia tomentosa; REPSA
Introduction
There is a general consensus among ecologists that seed dispersal by animals is a very important service and that many plant species and even ecosystems could be dependent on this process (Gelmi-Candusso & Hämäläinen, 2019; Godó et al., 2022; Iluz, 2011). And there is an important amount of information on seed dispersal by birds, primates, and ungulates, among other groups (Albert et al., 2015; Beaune et al., 2013; Iluz, 2011); however, information on some other potentially important dispersers, particularly the lagomorphs, is scarce (Godó et al., 2022).
Lagomorphs are a widely distributed and abundant group of mammals that can be found across many ecosystems in all continents, except Antarctica (Chapman & Flux, 2008). Previous studies have shown that lagomorphs are not particularly important seed dispersers when compared to other animals by the number of seeds per gram of excrete (Borchert & Tyler, 2023; Malo & Suárez, 1995). However, they do disperse seeds, and given their abundance and the high amount of fecal matter they produce, they still have the potential to be important seed dispersers. Despite that, there are few studies that evaluate the role of lagomorphs as seed dispersers, and most of them used Oryctolagus cuniculus as the study model (Godó et al., 2022). Therefore, the aim of this study is to describe the diversity and abundance of seeds dispersed by endozoochory by the Eastern cottontail (Sylvilagus floridanus) in an urban reserve in Mexico City.
There are several studies about seed dispersal by the genus Sylvilagus, and they have found that species of this genus can be important seed dispersers of Opuntia (Borchert & Tyler, 2023) and Juniperus (Lezama-Delgado et al., 2016). At the same time, S. floridanus has a diverse diet (Chapman et al., 1980; Hudson et al., 2005). Therefore, our prediction was that Opuntia seeds would be common in excretes, and that the Eastern cottontail will disperse more species than previously reported for the genus. However, to our knowledge, this is the first publication that studies endozoochory by S. floridanus, which is quite surprising given that this is one of the most studied rabbits in North America.
Materials and methods
The Eastern cottontail inhabits a wide variety of environments ranging from deserts to rain forests from southern Canada to Venezuela (Chapman et al., 1980) and has been introduced to other territories like Italy (Rosin et al., 2008). This rabbit produces around 350 fecal pellets/day (Cochran & Stains, 1961), making it a potentially important seed disperser. Its diet consists mainly of grasses and herbaceous plants (Hudson et al., 2005), but it can switch to trees if its preferred food is scarce, for example, during the winter (Chapman et al., 1980). There are also reports of Eastern cottontails consuming fruits and seeds (Lorenzo & Cervantes, 2005); thus, it is a generalist herbivore.
We performed this study in the Reserva Ecológica del Pedregal de San Ángel located in Mexico City (19°18’55” N, 99°11’32” W). The reserve is dominated by a xerophytic scrub with a mean precipitation of 752 mm and an average annual temperature of 18.2 °C (Rzedowski, 1954; SMN, 2025). The reserve has 2 seasons, the wet (from May to October) and the dry (from November to April), and most seeds are only able to germinate during the wet period (Rzedowski, 1954). There is only 1 species of rabbit, S. floridanus, found in this reserve (Hortelano-Moncada et al., 2009).
We collected excretes of S. floridanus twice, the first collection between October 28 and 30, 2016 in 4 locations separated by at least 100 m (we used different locations to capture the variability of the reserve, and it is not the purpose of this study to compare between locations, and after collection, pellets from different locations were mixed together). The second collection was made between May 17 and 22, 2017. We chose those dates because the October collection represents the point of highest seed production in the ecosystem (Cesar-García, 2002) and therefore probably the greatest amount of seeds per excrete, and the second because it’s the beginning of the rainy season and thus germination. After collection, we cleaned the pellets from possible additions of seeds by sieving. The method resulted effective since we observed seeds of Muhlenbergia robusta (Poaceae) fall on the collected pellets, but that plant did not appear in our experiment suggesting, that we were able to get rid of seeds added after defecation.
We performed 2 germination experiments, and the purpose of the first was to determine how many seeds can germinate immediately after the October collection and to compare methods of germination. In the field we observed that fecal pellets could be found entire or disintegrated and could be laying on soil or on rock; therefore, we used 4 treatments: 1) entire pellets in sterilized soil (we sterilized the soil to kill all possible seeds by microwaving before adding the pellets); 2) disintegrated pellets in sterilized soil; 3) entire pellets with no soil, and 4) disintegrated pellets with no soil. This also allowed us to determine the best germination method for the second phase of the experiment. For each treatment we used 1,000 fecal pellets divided among 10 trays. All treatments were put in germination chambers with 12/12 light/dark periods at 25 °C during 3 months. We used the Kruskal-Wallis and post hoc Dunn tests to compare the number of germinated seeds between treatments (using trays as replicas) and calculated the number of seeds per gram of excrete to facilitate comparison with other species.
The purpose of the second germination period was to determine how many seeds can germinate from the October pellets after being stored in different conditions and the pellets collected in May with no storage, in germination chambers, and in the field. In our study location, seeds that are dropped in October cannot germinate in normal field conditions because there is almost no rain, and most seeds in xerophytic shrubs have some type of dormancy to avoid germinating before the rainy season (85% of the seeds have dormancy, according to Baskin and Baskin [2014]). Therefore, we tested 2 methods of pellet storage: laboratory conditions, pellets were dried and stored at room temperature with no exposure to sun. And in the field, pellets were dropped in the field in a location with no rabbits and recollected in May. At the same time, we wanted to test if seeds germinate the same in laboratory and in field conditions; therefore, we put disintegrated pellets in sterilized soil (based on the result of the first experiment) using pellets collected in October and kept in the laboratory, pellets collected in October and kept in the field, and pellets collected in May with no storage in germination chambers (with the same conditions as the previous experiment, we inspected and watered those treatments 3 times a week during 3 months) and in the field (Table 1). The field experiment used the same sterilized soil, but the trays were higher to allow plants to grow and were covered by fabric with 0.04 mm openings to prevent seed addition from the exterior. In this case, the trays were left in the field, and no watering or other treatment was performed. For these experiments we only considered vascular plants. We inspected the field treatments once every week for 3 months. We used Kruskal-Wallis and Dunn tests to compare between treatments (including the disintegrated pellets in the soil treatment from the first experiment) and the Chao estimator for the species richness to estimate the number of species that are dispersed but did not appear in our study.
Results
In the first experiment, we observed the germination of 1.4 ± s.d. 1.3 seeds/100 pellets in the disintegrated pellets in soil treatment, 1.0 ± 0.8 seeds/100 pellets in the entire pellets in soil treatment, and 0.1 ± 0.3 seeds in both treatments with no soil. The Kruskal-Wallis test showed that there are differences between the treatments (p < 0.05), and according to the Dunn test, there are differences between treatments with and without soil but no differences between disintegrated and entire pellets. During this stage we observed 4 morphologically distinctive plants, but they all died early and could not be identified. The main reason plants could not develop was that all pellets were being actively degraded by fungi, which ultimately killed all emerging plants. We also observed that in the treatments with no soil, it was hard to maintain the adequate humidity of the substrate.
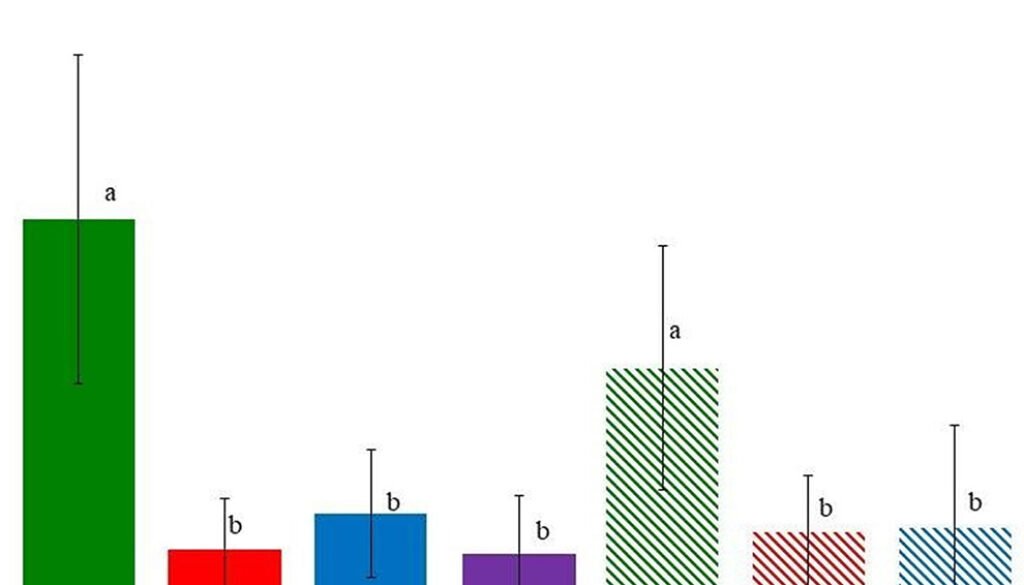
In the second experiment, the Kruskal-Wallis test also showed differences between groups (p < 0.05), and according to the Dunn test, the pellets collected in October and left in the field (both the germination cameras and the field) had significantly more seeds germinating from them (Fig. 1, Table 1). Cottontail excretes contained between 1.4 and 8.8 seeds/100 pellets, which equals to 0.12 and 0.77 seeds/g of excrete (considering that the weight of an excrete is 0.115, according to Glebskiy [2016]). We observed 15 species of dispersed seeds, and according to the Chao estimator, there are 29.8 species dispersed (with a 95% confidence interval of ± 13.4). The most common group of dispersed seeds was the Poaceae family (34.5% of all seeds), followed by Opuntia tomentosa (25.9%), Physalis glutinosa (16.4%), and Jaegeria hirta (12.7%). The Poaceae family was composed of Aegopogon tenellus, Eragrostis Mexicana, and Chloris gayana; however, due to their similarities in the initial stages and that some of the plants died, we were unable to determine the exact number of plants for each species (Table 1).
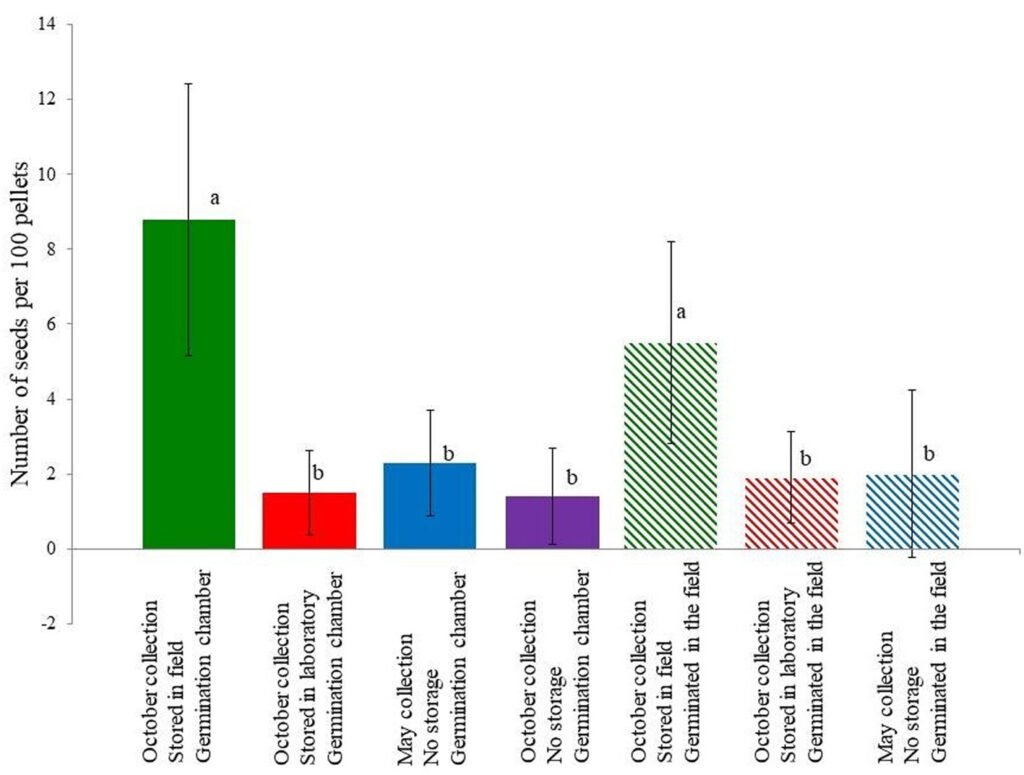
Figure 1. Number of seeds germinated per 100 cottontail fecal pellets ± standard deviation. Letters represent significant differences between treatments according to the Dunn test. Full bars represent treatments in germination chambers.
Discussion
The first experiment showed that seeds from both entire and disintegrated pellets can germinate equally well. We used disintegrated pellets in the following experiments because they are easier to homogeneously disperse in the experiment tray. However, germination was very low in the experiments with no soil. This suggests that if the pellets are placed on soil, the seeds inside are able to germinate, but if the cottontail forms a latrine on a rock or some other hard object (as it is commonly observed in our study location; pers. obs.), the pellets are not a good substrate for the plants to develop (at least in the fresh state). An interesting observation that we made was that in the October germination experiment, fungi were an important problem for plant development, but the same did not happen in the following experiment. This could be due to the fact that July-October are the months when most fungi expel their spores and infect the pellets (Valenzuela et al., 2009), but they apparently (at least partially) die before the germination period and by May do not represent an important threat for seeds or seedlings. Therefore, fungi are unlikely to strongly affect the process of seed dispersion by cottontails, since our October germination experiment would never happen in field conditions because of the dry season.
Baskin and Baskin (2014) mention that in xerophitic scrubs, 85% of seeds have some sort of dormancy; this is close to the results of our experiment. If we compare the number of seeds germinated from pellets stored in laboratory and field conditions, we can see that pellets exposed to the field yielded 5.9 (in germination chambers) and 2.9 (in the field) times more plants (Fig. 1). This suggests that most seeds have some sort of dormancy that is broken by the conditions in the field, most likely by heat, as it was shown for Opuntia tomentosa (Olvera-Carrillo et al., 2003). Notice that the Opuntia seeds almost never germinate after being stored in laboratory conditions (Table 1).
Table 1
Number and identity of the seeds germinated per 1,000 fecal pellets in each treatment. Chao estimator ± 95% confidence interval.
| Germinated in chambers | Germinated in the field | Total | |||||
| Collection | October collection | May collection | October collection | May collection | |||
| Storage | Field | Lab. | None | Field | Lab. | None | |
| Poaceae1 | 29 | 9 | 5 | 25 | 7 | 1 | 76 |
| Opuntia tomentosa | 13 | 1 | 14 | 12 | 0 | 17 | 57 |
| Physalis glutinosa | 15 | 2 | 2 | 8 | 7 | 2 | 36 |
| Jaegeria hirta | 17 | 1 | 0 | 8 | 2 | 0 | 28 |
| Drymaria laxiflora | 6 | 0 | 1 | 1 | 0 | 0 | 8 |
| Crusea longiflora | 5 | 0 | 0 | 0 | 0 | 0 | 5 |
| Jaltomata procumbens | 1 | 1 | 0 | 1 | 1 | 0 | 4 |
| Galinsoga parviflora | 1 | 0 | 0 | 0 | 0 | 0 | 1 |
| Evolvulus alsinoides | 1 | 0 | 0 | 0 | 0 | 0 | 1 |
| Bidens bigelovii | 0 | 0 | 0 | 0 | 1 | 0 | 1 |
| Solanum bulbocastanum | 0 | 1 | 0 | 0 | 0 | 0 | 1 |
| Buddleja cordata | 0 | 0 | 1 | 0 | 0 | 0 | 1 |
| Unidentified | 0 | 0 | 0 | 0 | 1 | 0 | 1 |
| Total seeds | 88 | 15 | 23 | 55 | 19 | 20 | 220 |
| Pellets used | 1,000 | 1,000 | 1,000 | 1,000 | 1,000 | 1,000 | 6,000 |
| Observed richness | 9 | 6 | 5 | 6 | 6 | 3 | 15 |
| Chao2 richness | 11.7 ± 4.1 | 10.6 ± 4.8 | 10.1 ± 6.5 | 7.9 ± 2.0 | 11.1 ± 6.5 | 3.5 ± 1.2 | 29.8 ± 13.4 |
1Includes: Aegopogon tenellus, Eragrostis Mexicana, and Chloris gayana (the last one is the only exotic species).
The Eastern cottontails showed to be poor seed dispersers if considering the amount of seeds per gram of excretes, 0.77 under the best conditions, compared with the 7.13 seeds/g of excrete dispersed by ringtails in the same location (Peña-Herrera et al., 2024). However, cottontails produce around 350 pellets/day (Cochran & Stains, 1961), which at a weight of 0.115 g/pellet (Glebskiy, 2016) which equals 40 g of excretes and thus up to 31 seeds dispersed per day per cottontail (and their population can rise up to 14 individuals/ha (Glebskiy, 2016). Another important consideration is the identity of the seeds, from the 14 species that we were able to identify; only 2 (Opuntia tomentosa and Buddleja cordata) could be considered dominant in the region (according to the list provided by Cano-Santana [1994]), and the other species are less common or even rare. Most noticeable Jaegeria hirta, which composes 12.7% of all seeds found and is a native but rare species for the location. It has to be noted that of the dispersed species (Table 1), only Chloris gayana is exotic to the location. When comparing to the seeds dispersed by ringtails in the same location (Peña-Herrera et al., 2024), we can see that only 3 species are shared (Opuntia tomentosa, Drymaria laxiflora, and Bidens sp.) and cottontails tend to disperse more Poaceae plants. This suggests that ringtails and rabbits have a complementary role as seed dispersers.
Therefore, we conclude that while being poor dispersers of seeds/g of excrete (the most commonly used estimator to measure dispersion efficiency), cottontails compensate for this by the sheer amount of excretes produced, their high population density, and the fact that they disperse mostly uncommon or even rare plants, and most are native to the region. Thus, cottontails are important seed dispersers and can have a great effect on some rare plants, especially Jaegeria hirta.
Acknowledgements
We are thankful to Díaz Rico A. and Zúñiga Ruíz B. for allowing us to use their germination chambers, Martínez Orea Y. for the plant identification, SEREPSA working team for the permits to collect pellets, List Sánches R. and Godínez Álvarez H. for their useful comments on this project, and Castellanos Vargas I. for technical support. This project was financially supported by PAPIIT grant IV200117 “Análisis ecosocial de una reserva urbana para la sustentabilidad en el campus de Ciudad Universitaria” to ZCS and a scholarship by Conacyt 817316 to YG.
References
Albert, A., Auffret, A. G., Cosyns, E., Cousins, S. A., D’hondt, B., Eichberg, C. et al. (2015). Seed dispersal by ungulates as an ecological filter: A trait-based meta-analysis. Oikos, 124, 1109–1120. https://doi.org/10.1111/oik.02512
Baskin, C. C., & Baskin, J. M. (2014). Seeds ecology, biogeography, and evolution of dormancy and germination. 2nd. Edition. San Diego: Elsevier.
Beaune, D., Bretagnolle, F., Bollache, L., Bourson, C., Hohmann, G., & Fruth, B. (2013). Ecological services performed by the bonobo (Pan paniscus): seed dispersal effectiveness in tropical forest. Journal of Tropical Ecology, 29, 367–380. https://doi.org/10.1017/S0266467413000515
Borchert, M., & Tyler, C. (2023). Fruit production and seed dispersal of Opuntia phaeacantha (Cactaceae) in the southwest Mojave Desert. Haseltonia, 29, 49–56. https://doi.org/10.2985/026.029.0108
Cano-Santana, Z. (1994). Flujo de energía a través de Sphenarium purpuracens(Orthoptera: Acrididae) y productividad primaria neta aérea en una comunidad xerófila. (Ph.D. Thesis). Mexico City: Universidad Nacional Autónoma de México.
César-García, S. F. (2002). Análisis de algunos factores que afectan la fenología reproductiva de la comunidad vegetal de la Reserva del Pedregal de San Ángel, D.F. (México) (Bachelor´s Thesis). Mexico City: Universidad Nacional Autónoma de México.
Chapman, J. A., & Flux, J. E. (2008). Introduction to the Lagomorpha. In P. C. Alves, N. Ferrand, & K. Hackländer (Eds.), Lagomorph Biology: evolution, ecology, and conservation (pp. 1–9). Berlin: Springer. https://doi.org/10.1007/978-3-540-72446-9
Chapman, J. A., Hockman, J. G., & Ojeda, C. M. M. (1980). Sylvilagus floridanus. Mammalian Species, 136, 1–8.
Cochran, G. A., & Stains, H. J. (1961). Deposition and decomposition of fecal pellets by cottontails. The Journal of Wildlife Management, 25, 432–435. https://doi.org/10.2307/3798835
Gelmi-Candusso, T. A., & Hämäläinen, A. M. (2019). Seeds and the city: the interdependence of zoochory and ecosystem dynamics in urban environments. Frontiers in Ecology and Evolution, 7, 41. https://doi.org/10.3389/fevo.2019.00041
Glebskiy, Y. (2016). Factores que afectan la distribución y abundancia del conejo castellano (Sylvilagus floridanus) en la Reserva del Pedregal de San Ángel, D.F. (México) (Bachekor’s Thesis). Mexico City: Universidad Nacional Autónoma de México.
Godó, L., Valkó, O., Borza, S., & Deák, B. (2022). A global review on the role of small rodents and lagomorphs (clade Glires) in seed dispersal and plant establishment. Global Ecology and Conservation, 33, e01982. https://doi.org/10.1016/j.gecco.2021.e01982
Hortelano-Moncada, Y., Cervantes, F. A., & Trejo-Ortiz, A. (2009). Mamíferos silvestres de la Reserva Ecológica del Pedregal de San Ángel en Ciudad Universitaria, Universidad Nacional Autónoma de México, México, D. F. Revista Mexicana de Biodiversidad, 80, 507–520. https://doi.org/10.22201/ib.20078706e.2009.002.614
Hudson, R., Rodríguez-Martínez, L., Distel, H., Cordero, C., Altbäcker, V., & Martínez-Gómez, M. (2005). A comparison between vegetation and diet records from the wet and dry season in the cottontail rabbit Sylvilagus floridanus at Ixtacuixtla, central Mexico. Acta Theriologica, 50, 377–389. https://doi.org/10.1007/BF03192633
Iluz, D. (2011). Zoochory: the dispersal of plants by animals. In Z. Dubinsky, & J. Seckbach (Eds.), All flesh is grass: plant-animal interrelationships (pp. 199–214). Dordrecht: Springer. https://doi.org/10.1007/978-90-481-9316-5_9
Lezama-Delgado, E., Sainos-Paredes, P., López-Portillo, J., Ángeles, G., Golubov, J., & Martínez, A. J. (2016). Association of Juniperus deppeana (Cupressaceae: Pinales) seeds with Mexican cottontail rabbit (Sylvilagus cunicularius; Leporidae: Lagomorpha) latrines. Journal of Natural History, 50, 2547–2555. https://doi.org/10.1080/
00222933.2016.1200685
Lorenzo, C., & Cervantes, F. (2005). Silvialgus floridanus. In G. C. G. Oliva, & G. Ceballos (Eds.), Los mamíferos silvestres de México. Mexico City: Comisión Nacional para el Conocimiento y Uso de la Biodiversidad/ Fondo de Cultura Económica.
Malo, J. E., & Suárez, F. (1995). Herbivorous mammals as seed dispersers in a Mediterranean dehesa. Oecologia, 104, 246–255. https://doi.org/10.1007/bf00328589
Olvera-Carrillo, Y., Márquez-Guzmán, J., Barradas, V. L., Sánchez-Coronado, M. E., & Orozco-Segovia, A. (2003). Germination of the hard seed coated Opuntia tomentosa
SD, cacti from the México valley. Journal of Arid Environments, 55, 29–42. https://doi.org/10.1016/s0140-1963(02)00268-9
Peña-Herrera, J. J., Glebskiy, Y., Hernández-Trejo, T., & Cano-Santana, Z. (2024). Ringtails (Bassariscus astutus) as seed dispersers in an urban gradient under conditions of low human activity due to COVID-19. Revista Mexicana de Biodiversidad, 95, e955351. https://doi.org/10.22201/ib.20078706e.2024.95.5351
Rosin, A. V., Gilio, N., & Meriggi, A. (2008). Introduced lagomorphs as a threat to “native” lagomorphs: the case of the Eastern cottontail (Sylvilagus floridanus) in northern Italy. In P. C. Alves, N. Ferrand, & K. Hackländer (Eds.), Lagomorph Biology: evolution, ecology, and conservation (pp. 153–164). Berlin: Springer. https://doi.org/10.1007/978-3-540-72446-9_11
Rzedowski, J. (1954). Vegetación del Pedregal de San Ángel. Anales de la Escuela Nacional de Ciencias Biológicas, Instituto Politécnico Nacional, 8, 59–129.
SMN (Servicio Meteorológico Nacional). (2025). Normales climatológicas 1991-2020, estación 9071 Colonia Educación. https://smn.conagua.gob.mx/tools/RESOURCES/Nor
males_Climatologicas/Normales9120/df/nor9120_09071.txt
Valenzuela, V. H., Herrera, T., & Pérez-Silva, E. (2009). Macromicetos. In A. Lot, & Z. Cano-Santana (Eds.), Biodiversidad del ecosistema del Pedregal de San Ángel (pp. 95–100). Mexico City: Universidad Nacional Autónoma de México.