Complejo Prorocentrum lima asociado a macrofitas en dos sitios de afloramiento de borde oriental: laguna Estero de Urías (México) y bahía de Paracas (Perú)
Tomasa Cuellar-Martinez a, *, Ana Carolina Ruiz-Fernández a, Joan Albert Sanchez-Cabeza a, Arturo Aguirre-Velarde b, Haydeé Felícita López-Cabanillas b, Jorge Tam c,
Sonia Sánchez c, François Colas d
a Universidad Nacional Autónoma de México, Instituto de Ciencias del Mar y Limnología, Unidad Académica Mazatlán, Capitán Joel Montes Camarena s/n, Cerro del Vigía, 82040 Mazatlán, Sinaloa, Mexico
b Universidad Nacional Agraria La Molina, Facultad de Pesquería, Av. La Molina s/n, La Molina, Lima, Peru
c Instituto del Mar del Perú, Esquina Gamarra y General Valle s/n, Chucuito Callao, Lima, Peru
d Institut de Recherche pour le Développement (IRD), Laboratoire LOPS, Institut Universitaire Européen de la Mer (IUEM), 29280 Plouzané, France
*Corresponding author: tcuellar@ola.icmyl.unam.mx (T. Cuellar-Martinez)
Received: 20 November 2024; accepted: 17 September 2025
Abstract
Benthic or epibenthic dinoflagellates (EDs) are a potential risk to the environment and human health due to the production of toxins by some species. This study explored for the first time the presence of EDs mainly associated with macrophytes (macroalgae and seagrass) at 2 sites influenced by upwellings: Estero de Urías Lagoon (EUL), at the entrance of the Gulf of California, and Paracas Bay (PB), on the southern Peruvian coast. Prorocentrum lima complex was present at low abundances: ≤ 25 cells g-1 wet weight in EUL and ≤ 867 cells g-1 wet weight in PB. It was recorded in a wide range of temperatures from 22.2 to 31.6 °C in EUL and from 18.0 to 22.2 °C in PB. Despite its low abundance, monitoring the EDs community is essential to detect changes in the distribution patterns of harmful species in the context of climate change.
Keywords: Macroalgae; Caulerpa; Artificial substrate; California Current; Humboldt Current
Resumen
Los dinoflagelados epibentónicos (DE) representan un riesgo potencial al ambiente y a la salud del ser humano debido a la producción de toxinas por parte de algunas especies. En este estudio, se exploró por primera vez la presencia de DE principalmente asociados a macrofitas (macroalgas y pastos marinos) en 2 sitios con influencia de afloramientos: laguna Estero de Urías (LEU), localizado en la entrada del golfo de California y la bahía de Paracas (BP) en la costa sur de Perú. El complejo Prorocentrum lima estuvo presente con bajas abundancias: ≤ 25 células g-1 peso húmedo en LEU y ≤ 867 células g-1 peso húmedo en BP. La especie se registró en un amplio intervalo de temperatura de 22.2 a 31.6 °C en LEU y de 18.0 a 22.2 °C en BP. A pesar de las bajas abundancias encontradas, el monitoreo de la comunidad de DE es importante para detectar cambios en los patrones de distribución de las especies nocivas en un contexto de cambio climático.
Palabras clave: Macroalgas; Caulerpa; Sustrato artificial; Corriente de California; Corriente de Humboldt
Introduction
Benthic or epibenthic dinoflagellates (EDs) are common in shallow waters. They are frequently attached to many substrate types, such as algal turf, macroalgae, rocks, coral rubble, sand, or seagrasses (Honsell et al., 2013; Yong et al., 2018). Although they prefer substrate attachment, the vegetative cells of free-living species are flagellated and are fully capable of detachment and motility (Durán-Riveroll et al., 2019). The excessive growth of these species, particularly the toxigenic ones, can produce benthic harmful algal blooms, and toxins can be transferred along the food web; in humans, consumption of contaminated seafood with these toxins can cause diarrhetic shellfish poisoning (DSP) or ciguatera fish poisoning (Berdalet et al., 2016).
Although EDs have a high relative cell abundance and diversity in subtropical and tropical waters, they are found globally, including in temperate, sub-arctic, and polar environments (Álvarez et al., 2022; Tester et al., 2010). In recent decades, evidence suggests a shift in biogeographical distribution patterns for some species (Gobler et al., 2017; Tester et al., 2020), such as Gambierdiscus,previously considered tropical or sub-tropical. Recent studies have shown that it is well established in temperate areas such as Korea, Japan, New Zealand, Australia, the northern Gulf of Mexico, and the Mediterranean Sea (Chinain et al., 2021). Furthermore, there is concern that floating plastics in the ocean can act as global vectors for transporting algal cells and transferring toxins along marine food webs (do Prado-Leite et al., 2022).
In Mexico, about 60 species from 18 EDs genera have been identified. The species that form dense benthic blooms with potentially harmful consequences are Prorocentrum rhathymum, Blixaea quinquecornis, and Amphidinium cf. carterae (Okolodkov et al., 2022). In South America, 31 EDs taxa have been reported (Mafra et al., 2023). The most frequent and widespread species was the P. lima complex (Mafra et al., 2023). On the Peruvian coast, the cold coastal waters of the Humboldt Current System could restrict the presence of these species (Durán-Riveroll et al., 2019). The P. lima complex has been frequently observed over the past ~ 50 years, though studies have primarily focused on the planktonic community (Mafra et al., 2023).
Benthic microalgae are expected to benefit from climate change conditions, such as warmer waters and changes in marine current patterns, which could promote their invasive colonization and range of expansion (Tester et al., 2010). Estero de Urías Lagoon (EUL) and Paracas Bay (PB) host harbors with high levels of activity (i.e., the harbors of Mazatlán and San Martín, respectively), which may represent a potential risk since ballast waters remain a vector for the introduction of non-native and potentially harmful aquatic species (Lee et al., 2021). This study aimed to explore the EDs associated with macrophytes in Estero de Urías Lagoon, Mazatlán (Mexico), and Paracas Bay, Pisco (Peru), and the environmental variables associated with the presence of these species.
Materials and methods
Estero de Urías Lagoon (EUL; 23.2° N) is located at the entrance to the Gulf of California (Fig. 1) and covers an area of 18 km2 with water depths of 1-3 m except in the navigation channel where it is up to 13 m (Montaño-Ley et al., 2008). It is surrounded by the industrialized city and harbor of Mazatlán (Raygoza-Viera et al., 2014). It is influenced by seasonal coastal upwelling events (Herrera-Becerril et al., 2022). EUL does not receive continuous freshwater, for which it exhibits an inverse estuarine circulation, and during the warmest months high salinity occurs in the upper lagoon (Cardoso-Mohedano et al., 2018).
The climate in the area is warm and humid, with a mean annual temperature of 21.4-29.6 °C (1951-2017) and a mean annual precipitation of 372 mm (SMN, 2024). EUL is an anthropized coastal lagoon affected by urban development, shrimp farming, and discharges of domestic, industrial, and wastewater (Hernández-Cornejo & Ruiz-Luna, 2000; Méndez, 2002). The Mazatlán harbor is the second largest on the Mexican Pacific coast, and in 2023 it received almost 500,000 cruise ship passengers from the largest cruise ships navigating the globe (API, 2024).
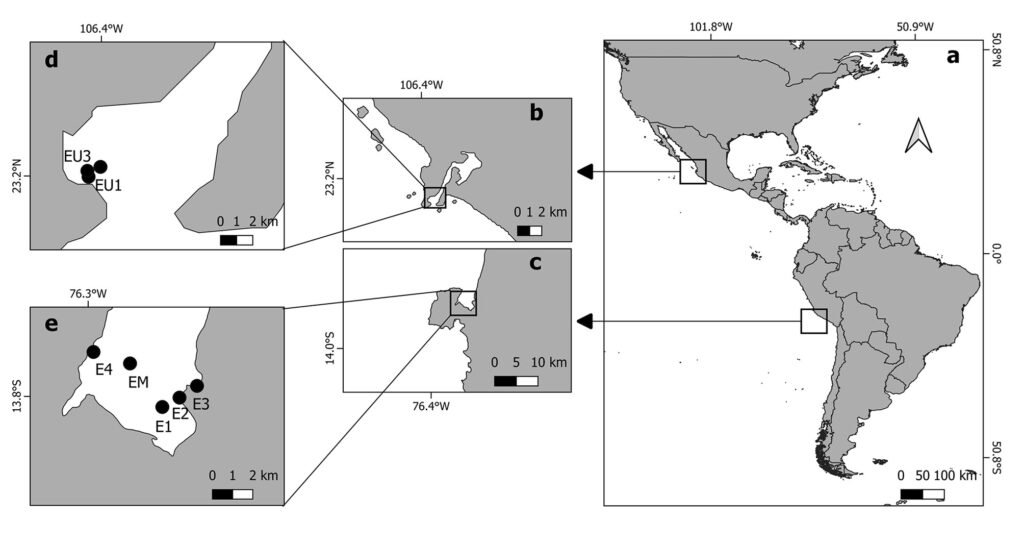
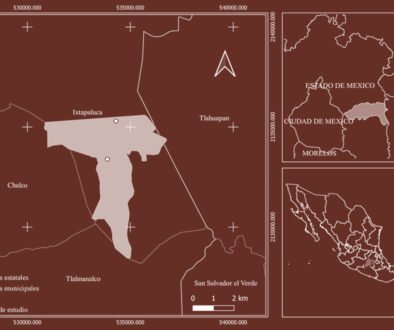
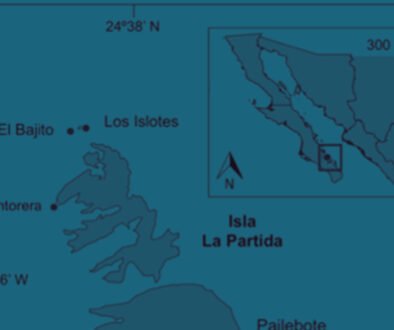
Figure 1. a, Map of the study sites; b, d, sampling sites in Estero de Urías (EU1, EU2, EU3), Mexico; c, e, Paracas Bay (E1, E2, E3, E4, EM), Peru.
Paracas Bay (PB; 13.8° S) is located in Pisco, off South-Central Peru (Fig. 1), with an area of 30 km2 and < 15 m of depth (Merma-Mora et al., 2024). The southern part of the bay is included in the Paracas National Reserve (Sernanp, 2019). It is influenced by one of the main upwelling cells of the highly productive Peruvian Upwelling System (Chávez et al., 2008). PB is seasonally influenced by river discharge, and monthly averages range from 0.7 to 282 m3 s−1, with the higher values in austral summer (Cuellar-Martinez et al., 2023).
The climate in the region is arid, with an average annual rainfall of 1.83 mm and air temperatures ranging from 15 to 22 °C (Reyes, 2009). Anthropogenic activities around the bay include artisanal and industrial fishing, tourism, bay scallop Argopecten purpuratus aquaculture, fish processing industries, non-metallic minerals, and guano extraction (Reyes, 2009). The Terminal Portuario General San Martín, the country’s third most important port for cargo traffic (APN, 2018), is in the surroundings.
The most abundant macrophytes were collected manually or by diving and placed in polypropylene bags; the macrophyte masses ranged from 5 to 342.2 g in wet weight. The 3 sampling sites at EUL were close to the lagoon mouth, where a greater diversity of macroalgae species is found (Ochoa-Izaguirre et al., 2002). The sites were located in a total area of 0.01 km2 with fish cage cultures (~ 8 m depth). The most abundant macroalgae attached to the floating cage frames were collected monthly between August and November 2018 and in January, February, April, and June 2019. A total of 25 samples were collected, including species of Gracilaria sp., Hynea sp., Sargassum sp., Padina sp., Grateloupia sp., and Gelidium sp. In general, 1 to 4 samples were collected per sampling station. In addition, between August and October 2018, artificial substrates consisting of rectangular pieces (3 x 33 cm) of fiberglass screen fixed on a rigid frame were used (Jauzein et al., 2016). The artificial substrates were collected using scissors. For retrieval, 24 hours after installation, the substrate was cut on one side and carefully placed into a plastic bottle filled with 250 ml of ambient seawater. Then, the other side of the substrate was cut and placed inside the bottle, which was capped underwater (Jauzein et al., 2016).
In PB, the most abundant macrophytes, such as Caulerpa filiformis, Chondracanthus chamissoi, and Ruppia maritima were collected at 5 sites during June and November 2021, and April 2022. The sites were shallow (~ 1 m depth), whereas E1 and EM were 5 m and 10 m deep, respectively. A total of 24 samples were obtained. Additionally, surface water was sampled daily in E1 to evaluate the presence of EDs in the water (cells L-1), and HOBO data loggers (Onset UA-001-64) recorded surface and bottom temperatures every 20 minutes.
Environmental parameters (temperature, salinity, dissolved oxygen, and chlorophyll a) were measured in both PB and EUL using EXO (YSI) multiparameter sondes. In EUL, the chlorophyll a sensor was calibrated using rhodamine standards according to the manufacture’s guidelines (YSI Inc., 2014), allowing chlorophyll a concentrations to be expressed in absolute units (μg L⁻¹). In contrast, no calibration was performed in PB; thus, chlorophyll a values are reported in RFU (relative fluorescence units), reflecting the raw sensor response. Although not directly comparable to absolute concentrations, RFU values are widely used to identify temporal patterns in relative chlorophyll a concentrations (Foster et al., 2022). In the EUL, measurements were taken near the sampling sites (1-2 km) at stations 3 and 4 for the coastal observatory of global change in Mazatlán (Fig. 1 in Sanchez-Cabeza et al., 2019) and at station E1 for PB.
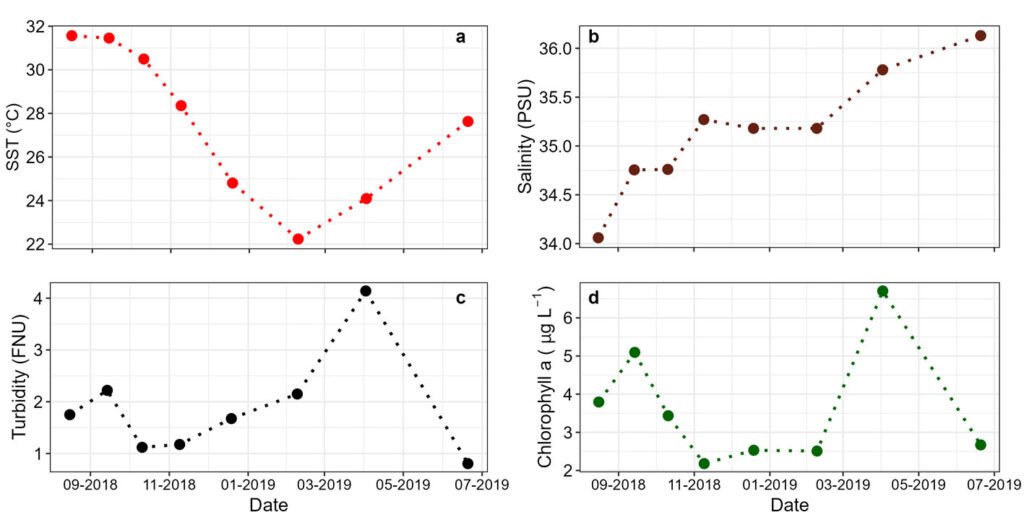
Figure 2. Mean values of the sea surface temperature (SST in °C). a) Salinity (PSU; b), turbidity (in nephelometric turbidity units, NTU); c) chlorophyll a concentration (in µg L-1; d) in the Estero de Urías Lagoon, at the entrance of the Gulf of California.
In the laboratory, the bags with macrophytes and bottles with artificial substrates were vigorously agitated for 2 minutes. The suspension was filtered through sieves with 250 µm, 150 µm, and 20 µm mesh to remove larger particles. The fraction retained in the 20 µm sieve was concentrated and transferred to a vial of 50 ml filled with seawater filtered through Whatman GF/F filters (nominal pore size ~ 0.7 µm), and preserved with 1% acidic Lugol’s iodine fixative. The EDs counting was performed on 3 replicate subsamples from the same macrophyte sample, using a Sedgwick-Rafter chamber in an optical microscope (LM Leica DMR, Wetzlar, Germany) with 200× magnification. Cell abundances were reported as cells per g w.w. of macrophyte (Reguera et al., 2011). The reported uncertainty is the standard deviation of the mean of 3 replicate counts.
The Shapiro test confirmed that species densities were not normally distributed. Therefore, Kruskal-Wallis one-way ANOVA and Dunn’s multiple comparison post hoc tests were used to assess whether abundances and environmental variables exhibited significant variations during the study period. The significance level (α) was set at < 0.05. All analyses and plots were performed with R version 2024.04.2 (R Core Team, 2024).
Results
Estero de Urías Lagoon
During the study period, the sea surface temperature (SST) varied from 22.2 to 31.6 °C, salinity from 34.0 to 36.1, turbidity from 0.8 to 4.1 NTU, and chlorophyll a from 2.2 to 6.7 mg L-1. The highest SST was observed in August-September, 2018 (Fig. 2).
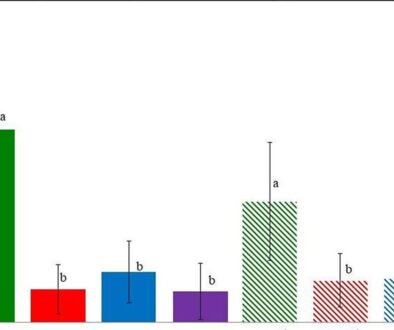
No EDs were observed attached to the artificial substrates in the EUL. Macroalgae were not found in EU2 in November 2018 and June 2019. Dinoflagellates on the EUL macroalgae belonged to the genus Prorocentrum; the highest abundances corresponded to P. micans (Supplementary material; Fig. S1). Prorocentrum lima complex was frequently observed in the samples, with abundances ranging from 0 to 78±45 cells g-1. No significant differences in the abundances were observed between seasons or among stations (Fig. 3a, b). Spearman’s correlation between the abundances of EDs and environmental variables was not significant.
Paracas Bay
During the study period, temperatures were from 14.1 to 22.2 °C. The sea surface and bottom temperatures were significantly higher in autumn (surface temperatures: 18.0-22.2 °C; bottom temperatures: 17.5-21.6 °C) than in summer and spring (surface temperatures: 14.5-22.1 °C; bottom temperatures: 14.1-19.7 °C; Fig. 4a). In the 3 seasons, the surface temperatures were significantly higher than the bottom temperatures. When the P. lima complex was observed in the water column and associated with macrophytes (May 14-Jun 04), SST ranged from 18.0 to 22.2 °C. Chlorophyll a ranged from 0.45 to 7.12 RFU. The highest values were observed in autumn, while spring and summer values were similar. The salinity varied between 34.4 and 35.2, with higher values in autumn than in spring and summer (4b).
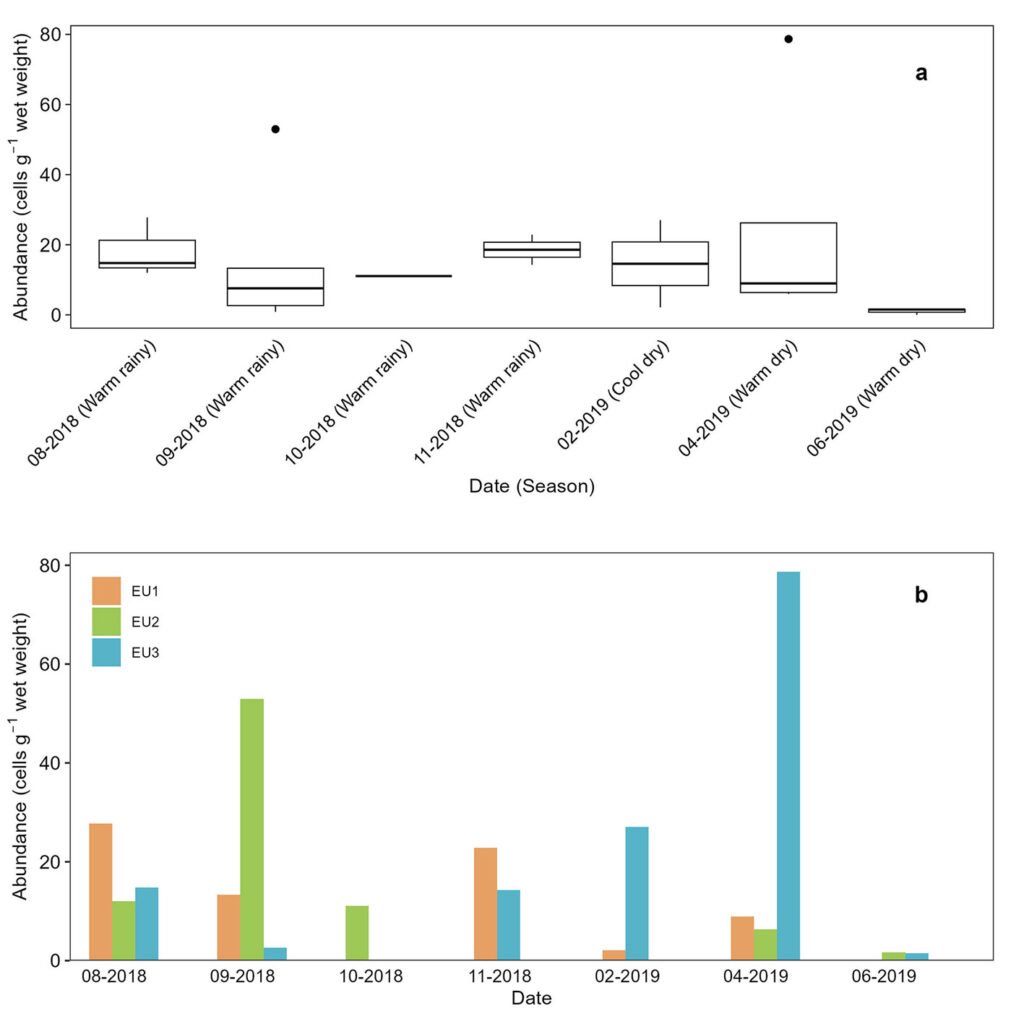
Figure 3. a) Abundance of the Prorocentrum lima complex, with seasons indicated in parentheses. Boxplots show the median (horizontal line), the interquartile range (the difference between the third and first quartile; box). Whiskers extend to values within 1.5 times the interquartile range, and data beyond this range are shown as outliers (dots); b, total abundance by station sites in the Estero de Urías Lagoon, located at the entrance of the Gulf of California.
In Paracas Bay, EDs were absent during most of the study period, and the P. lima complex was detected only in the austral autumn (June 2-4, 2021; Fig. 5, Table 1) with the highest abundances (867±172 cells g-1 in station E3). The P. lima complex was attached mostly to C. filiformis (Table 1).
According to the high-frequency monitoring of Prorocentrum species in surface water, P. micans was a common dinoflagellate in austral autumn and summer, and its abundances were from 40 to 1,840 cells L-1 (Supplementary material; Fig. S2). The P. lima complex was observed only in the austral autumn with a maximum abundance (1,360 cell L-1) on May 14, and between May 17 and June 01, its abundances were ≤ 280 cells L-1 (Fig. 4c).
Discussion
Artificial substrates have been successfully used for quantifying and monitoring EDs, particularly in sheltered, shallow, and subtidal sites (Tester et al., 2022). The lack of EDs attached to the artificial substrates in EUL is likely related to the low abundance of these organisms and the lagoon’s turbulent hydrodynamics. Low abundances of EDs were also observed associated with macroalgae. These results agree with observations from the coast of Tonga in an area exposed to high wave action, where Argyle (2018) detected low abundances of Gambierdiscus cells in association with macrophytes, but the artificial substrate failed to collect them. In contrast, Gambierdiscus concentrations were high on natural and artificial substrates deployed at sheltered, shallow, and subtidal sites. Despite EUL mouth having the highest macroalgae diversity (Ochoa-Izaguirre et al., 2002), it is not a sheltered zone. It is influenced by the tidal regime since the energy available for water circulation is primarily provided by tidal pumping, with tidal velocities of up to 0.60 m s-1 at the main channel (Montaño-Ley et al., 2008). The EDs can be attached by coating mucus or can live freely within the macroalgae interstices, so the turbulence and currents make them vulnerable to cell dispersal. They are mainly found in areas with low to moderately low energy environments (Foden et al., 2005).
Table 1
Abundance of Prorocentrum species in Paracas Bay in the austral autumn.
| Species | Date | Season | Station | Macrophyte species | Abundance (cells g wet weight)* |
| Prorocentrum lima complex | 2021-06-02 | Autumn | E3 | Caulerpa filiformis | 861±172 |
| Prorocentrum lima complex | 2021-06-02 | Autumn | E3 | Caulerpa filiformis | 250±17 |
| Prorocentrum lima complex | 2021-06-02 | Autumn | E3 | Caulerpa filiformis | 13±3 |
| Prorocentrum micans | 2021-06-02 | Autumn | E3 | Caulerpa filiformis | 6±5 |
| Prorocentrum lima complex | 2021-06-04 | Autumn | E3 | Chondracanthus chamissoi +Ulva sp. | 0 |
| Prorocentrum lima complex | 2021-06-04 | Autumn | E3 | Red algae | 0 |
| Prorocentrum lima complex | 2021-06-04 | Autumn | E3 | Red algae | 0 |
| Prorocentrum lima complex | 2021-06-04 | Autumn | E4 | Chondracanthus chamissoi | 1±1 |
| Prorocentrum lima complex | 2021-06-04 | Autumn | E4 | Ruppia marítima | 5±5 |
| Prorocentrum lima complex | 2021-06-04 | Autumn | E4 | Ruppia maritima | 5±2 |
* Abundances represent the average of 3 independent counts performed on the same sample. Values are reported as the mean ± standard deviation.
Among the Prorocentrum species associated with macrophytes in this study, only P. lima complex is recognized as a benthic species (Okolodkov et al., 2022), and it is the first time that P. lima complexis reportedattached to macrophytes in EUL and PB. This cosmopolitan taxon is widely distributed in South America (Mafra et al., 2023). Although low abundances were observed in PB, P. lima has been recorded in the water column with abundances of up to 4.5×104 cells L-1 (Cuellar-Martinez et al., 2023). Caulerpa filiformis is the most representative species in the macroalgae assemblage of PB (Olivas-Valverde, 2013), and the highest abundances of P. lima complex were observed on this species. Although information about the toxicity of P. lima complex is not available in strains from PB or EUL, reports worldwide reveal that all studied P. lima strains produce toxins related to DSP, such as okadaic acid, and several strains produce dinophysistoxins-1 (DTX1; Nishimura et al., 2020). Thus, monitoring this species on different substrates is relevant.
The presence of P. lima complex in both the water column and on macroalgae in Paracas Bay coincided with the highest temperatures recorded during the study period. Aissaoui et al. (2014) mentioned that this species appears to be eurythermal and euryhaline, as it grows over a wide interval of temperatures and salinities. In this work, P. lima complex was observed over a wide range of temperatures (18.0-31.6 °C), suggesting a remarkable ecological plasticity and ability to colonize diverse niches. Although P. lima complex was previously observed in PB at temperatures ranging from 15 to 25 °C (Mafra et al., 2023), in this study it was detected during autumn, when the highest temperatures reached 17.5-22.2 °C. Aquino-Cruz et al. (2018) highlighted the importance of temperature on cell growth rates, toxin levels, and photosynthetic efficiency. They determined that the optimum growth of P. lima strains from the United Kingdom occurred at 15-25 °C, with an increment in the production of okadaic acid at 15 °C.
Temperatures also affect the range of species distribution. It is expected that warmer temperatures associated with climate change could benefit harmful benthic dinoflagellate species, expanding their geographical distribution range (Tester et al., 2020). Another factor associated with distribution expansion is the artificial introduction of discharged ballast ship water (Park et al., 2021). The sampling period of this study limits the potential for making strong extrapolations regarding the temporal dynamics of the DEs and their ecological dynamics. Nevertheless, the detection of DEs with potential toxin production is a relevant finding that highlights the need for longer-term monitoring to better understand their ecological dynamics and potential harmful implications for environmental health and human activities.
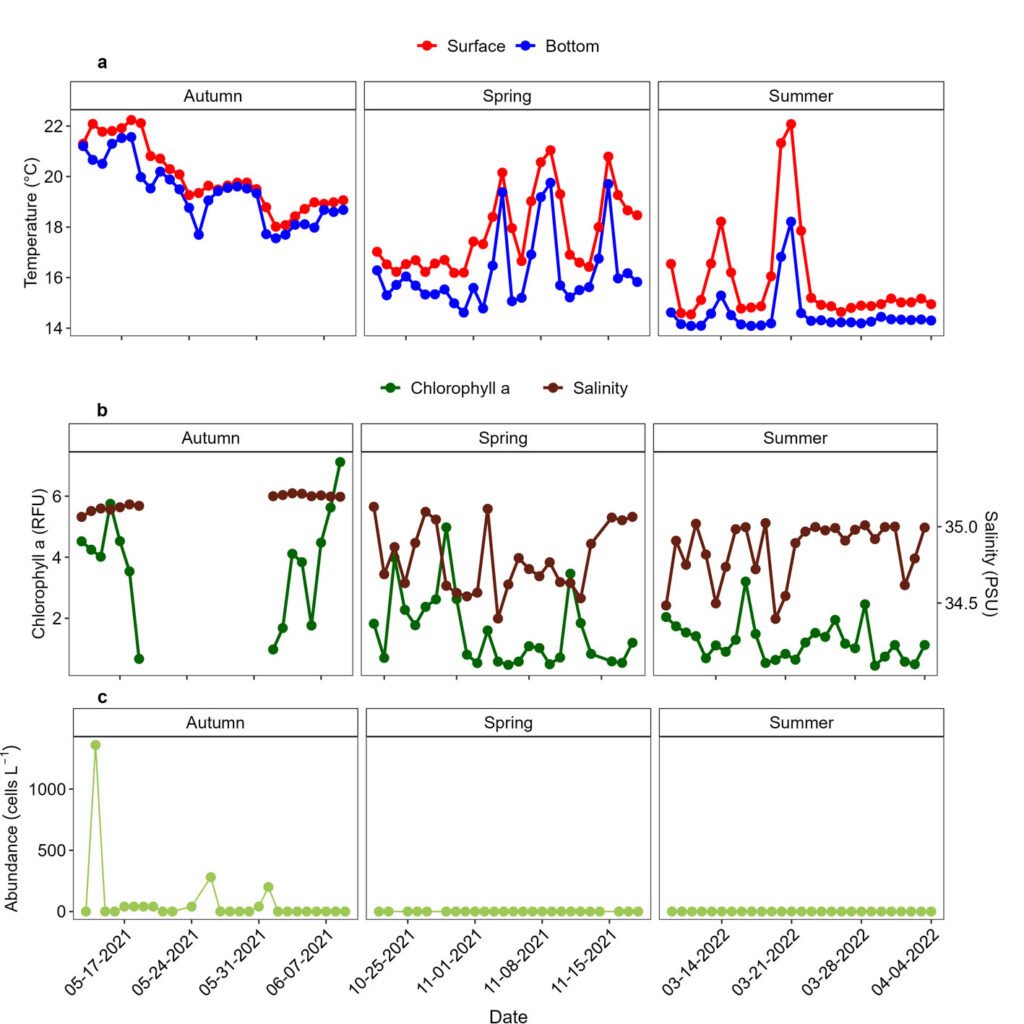
Figure 4. Surface and bottom temperatures (in °C). a) Surface salinity (PSU) and chlorophyll a (in relative fluorescence units, RFU; b), and seasonal abundances of Prorocentrum lima complex in surface waters (c) at the E1 station in Paracas Bay, Peru.
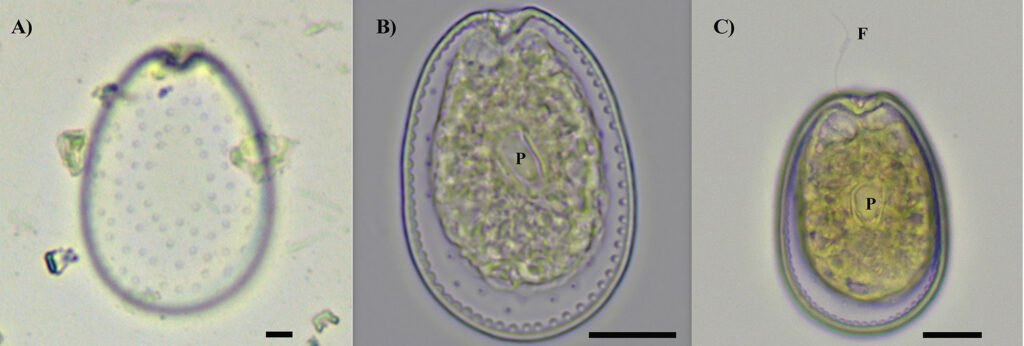
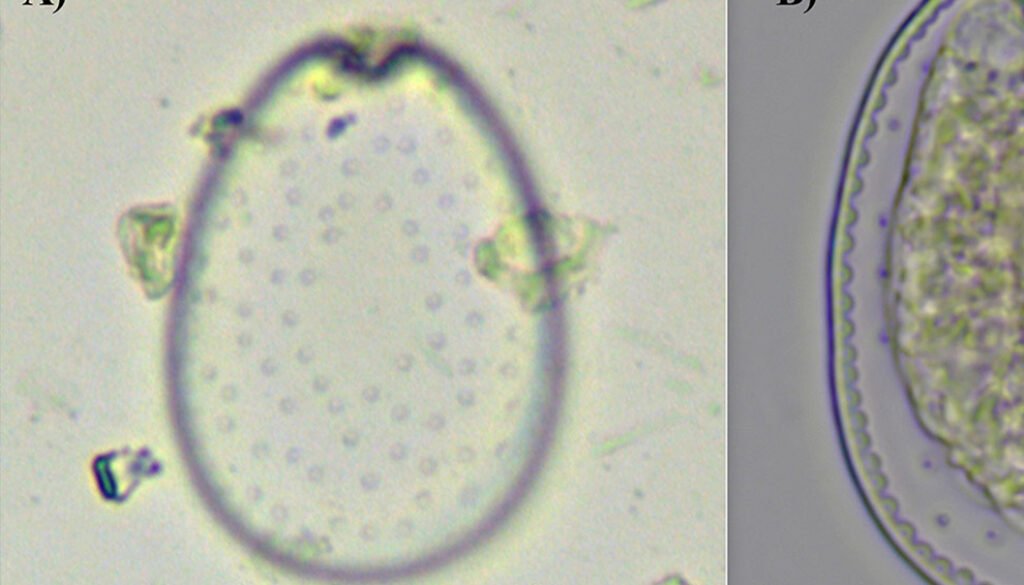
Figure 5. Vegetative cells of Prorocentrum lima complex collected from Paracas Bay, Peru, observed under light microscopy. A) Surface focus on a vegetative cell showing the pore pattern, B) cell showing pyrenoid (P), C) cell showing pyrenoid and flagellu. Scale bar: 10 µm.
The epibenthic dinoflagellate community was explored at 2 sites influenced by upwellings: Estero de Urías Lagoon, at the entrance to the Gulf of California, and Paracas Bay, off south-central Peru. The Prorocentrum lima complex was commonly found with low abundances, mainly associated with macrophytes in Estero de Urías Lagoon and sporadically detected in Paracas Bay (mainly on Caulerpa filiformis). In the water column of Paracas Bay, the P. lima complex was observed during the period with the highest recorded temperature in the study, suggesting potential environmental influences. These initial findings highlight the need for long-term studies with higher temporal resolution and increased detection frequency to better understand the environmental relationships of the benthic dinoflagellate at the studied sites. Given the known production of diarrhetic shellfish toxins by P. lima, its possible sensitivity to climate-related changes, and the anthropogenic pressures present in both study areas —such as intense marine traffic— further monitoring and multidisciplinary research, including toxicological, physiological, and molecular studies, are strongly recommended to assess the potential risks posed by P. lima to marine ecosystems and public health.
Acknowledgments
This work was supported by UNAM ICML (#347), IAEA-RLA/7014 and 7028, Project Concytec – World Bank 05-2019-FONDECYT-BM. This is a contribution to the Marine-Coastal Research Stressors Network for Latin America and the Caribbean (REMARCO) www.remarco.org and to the IRN DEXICOTROP of the Institut de Recherche pour le Développement (IRD). The authors thank M. Rangel, O. López Ramos, M. Fregoso-López, B. Yáñez-Chávez, H. Bojórquez-Leyva, and F. Rioual for the help provided in the fieldwork; R. Alonso-Rodríguez, J. A. Cabello Coral, B. Yáñez-Rivera, for microscopy facilities. F. Ramos-Huamaní and L. H. Pérez-Bernal for the laboratory analyses, and M. Ochoa-Izaguirre for the identification of macroalgae from the Estero de Urías Lagoon.
References
Aissaoui, A., Armi, Z., Akrout, F., & Ben-Hassine, O. K. (2014). Environmental factors and seasonal dynamics of Prorocentrum lima population in coastal waters of the Gulf of Tunis, South Mediterranean. Water Environment Research, 86, 2256–2270. https://doi-org/2443/10.2175/106143014X13975035526266
Álvarez, E. A., Klemm, K., Hoppenrath, M., Cembella, A., John, U., & Karlson, B. (2022). Temporal and spatial distribution of epibenthic dinoflagellates in the Kattegat-Skagerrak, NE Atlantic-Focus on Prorocentrum lima and Coolia monotis. Harmful Algae, 118, 102318. https://doi.org/10.1016/j.hal.2022.102318
API (Administración Portuaria Integral). (2024). Puerto de Mazatlán: plataforma logística integral. Available at https://www.puertomazatlan.com.mx/APIWEB/PRESENTA
TION_APIMAZ.PDF (Accessed May 20 2024).
APN (Autoridad Portuaria Nacional). (2018). Plan Maestro del Terminal Portuario General San Martin. Lima, Peru. Available at: http://webaplicacion.apn.gob.pe/proyecto/wp-
content/uploads/2020/10/Plan-Maestro-Portuario-TP-GENERAL-SAN-MARTIN.pdf (Accessed: March 11, 2024).
Aquino-Cruz, A., Purdie, D. A., & Morris, S. (2018). Effect of increasing sea water temperature on the growth and toxin production of the benthic dinoflagellate Prorocentrum lima. Hydrobiologia, 813, 103–122. https://doi.org/10.1007/s10750-018-3512-4
Argyle, P. A. (2018). The ecology and toxin production of Gambierdiscus and Fukuyoa species from the Pacific (Ph.D. Thesis). University of Canterbury, New Zealand.
Berdalet, E., Fleming, L. E., Gowen, R., Davidson, K., Hess, P., Backer et al. (2016). Marine harmful algal blooms, human health and wellbeing: challenges and opportunities in the 21st century. Journal of the Marine Biological Association of the United Kingdom, 96, 61–91. https://doi.org/10.1017/S0025315415001733
Cardoso-Mohedano, J. G., Lima-Rego, J., Sanchez-Cabeza, J. A., Ruiz-Fernández, A. C., Canales-Delgadillo, J., Sánchez-Flores, E. I. et al. (2018). Sub-tropical coastal lagoon salinization associated to shrimp ponds effluents. Estuarine, Coastal and Shelf Science, 203, 72–79. https://doi.org/10.1016/j.ecss.2018.01.022
Chávez, F. P., Bertrand, A., Guevara-Carrasco, R., Soler, P., & Csirke, J. (2008). The northern Humboldt Current System: Brief history, present status and a view towards
the future. Progress in Oceanography, 79, 95–105. https://doi.org/10.1016/j.pocean.2008.10.012
Chinain, M., Gatti, C. M. I., Darius, H. T., Quod, J. P., & Tester, P. A. (2021). Ciguatera poisonings: A global review of occurrences and trends. Harmful Algae, 102, 101873. https://doi.org/10.1016/j.hal.2020.101873
Cuellar-Martinez, T., Ochoa, A. D. R. H., Sánchez, S., Aguirre-Velarde, A., Ocas, E. O. M., Velasquez, A. M. R. et al. (2023). Abundance and distribution of potentially toxic phytoplankton in aquaculture sites along the Peruvian coast. Journal of Marine Systems, 240, 103865. https://doi.org/10.1016/j.jmarsys.2023.103865
do Prado-Leite, I., Menegotto, A., da Cunha-Lana, P., & Júnior, L. L. M. (2022). A new look at the potential role of marine plastic debris as a global vector of toxic benthic algae. Science of the Total Environment, 838, 156262. https://doi.org/10.1016/j.scitotenv.2022.156262
Durán-Riveroll, L. M., Cembella, A. D., & Okolodkov, Y. B. (2019). A review on the biodiversity and biogeography of toxigenic benthic marine dinoflagellates of the coasts of Latin America. Frontiers in Marine Science, 6, 148. https://doi.org/10.3389/fmars.2019.00148
Foden, J., Purdie, D. A., Morris, S., & Nascimento, S. (2005). Epiphytic abundance and toxicity of Prorocentrum lima populations in the Fleet Lagoon, UK. Harmful Algae, 4, 1063–1074. https://doi.org/10.1016/j.hal.2005.03.004
Foster, G. M., Graham, J. L., Bergamaschi, B. A., Carpenter, K. D., Downing, B. D., Pellerin, B. A. et al. (2022). Field techniques for the determination of algal pigment fluorescence in environmental waters. Principles and guidelines for instrument and sensor selection, operation, quality assurance, and data reporting (No. 1-D10). Virginia: US Geological Survey. https://doi.org/10.3133/tm1D10
Gobler, C. J., Doherty, O. M., Hattenrath-Lehmann, T. K., Griffith, A. W., Kang, Y., & Litaker, R. W. (2017). Ocean warming since 1982 has expanded the niche of toxic algal blooms in the North Atlantic and North Pacific oceans. Proceedings of the National Academy of Sciences, 114, 4975–4980. https://doi.org/10.1073/pnas.1619575114
Hernández-Cornejo, R., & Ruiz-Luna, A. (2000). Development of shrimp farming in the coastal zone of southern Sinaloa (Mexico): operating characteristics, environmental issues, and perspectives. Ocean & Coastal Management, 43, 597–607. https://doi.org/10.1016/S0964-5691(00)00049-1
Herrera-Becerril, C. A., Sanchez-Cabeza, J. A., Sánchez, L. F. Á., Lara-Cera, A. R., Ruiz-Fernández, A. C., Cardoso-Mohedano, J. G. et al. (2022). Statistical identification of coastal hypoxia events controlled by wind-induced upwelling. Continental Shelf Research, 233, 104634. https://doi.org/10.1016/j.csr.2021.104634
Honsell, G., Bonifacio, A., De Bortoli, M., Penna, A., Battocchi, C., Ciminiello, P. et al. (2013). New insights on cytological and metabolic features of Ostreopsis cf. ovata Fukuyo (Dinophyceae): A multidisciplinary approach. Plos One, 8, e57291. https://doi.org/10.1371/journal.pone.0057291
Jauzein, C., Fricke, A., Mangialajo, L., & Lemée, R. (2016). Sampling of Ostreopsis cf. ovata using artificial substrates: optimization of methods for the monitoring of benthic harmful algal blooms. Marine Pollution Bulletin, 107, 300–304. https://doi.org/10.1016/j.marpolbul.2016.03.047
Lee, E. S., Hwang, J., Hyung, J. H., & Park, J. (2021). Detection of the benthic dinoflagellates, Ostreopsis cf. ovata and Amphidinium massartii (Dinophyceae), Using Loop-Mediated Isothermal Amplification. Journal of Marine Science and Engineering, 9, 885. https://doi.org/10.3390/jmse9080885
Mafra, Jr., L. L., Sunesen, I., Pires, E., Nascimento, S. M., Álvarez, G., Mancera-Pineda, J. E. et al. (2023). Benthic harmful microalgae and their impacts in South America. Harmful Algae, 127, 102478. https://doi.org/10.1016/j.hal.2023.102478
Méndez, N. (2002). Annelid assemblages in soft bottoms subjected to human impact in the Urías Estuary (Sinaloa, Mexico). Oceanologica Acta, 25, 139–147. https://doi.org/
10.1016/S0399-1784(02)01193-3
Merma-Mora, L., Colas, F., Cardich, J., Sánchez, S., Flores, E., Lorenzo, A. et al. (2024). Bottom-water hypoxia in the Paracas Bay (Peru, 13.8° S) associated with seasonal and synoptic time scale variability of winds and water stratification. Journal of Marine Systems, 241, 103918. https://doi.org/10.1016/j.jmarsys.2023.103918
Montaño-Ley, Y., Peraza-Vizcarra, R., & Páez-Osuna, F. (2008). Tidal hydrodynamics and their implications for the dispersion of effluents in Mazatlán Harbor: An urbanized shallow coastal lagoon. Water, Air, and Soil Pollution, 194, 343–357. http://dx.doi.org/10.1007/s11270-008-9721-0
Nishimura, T., Uchida, H., Noguchi, R., Oikawa, H., Suzuki, T., Funaki, H. et al. (2020). Abundance of the benthic dinoflagellate Prorocentrum and the diversity, distribution, and diarrhetic shellfish toxin production of Prorocentrum lima complex and P. caipirignum in Japan. Harmful Algae, 96, 101687. https://doi.org/10.1016/j.hal.2019.101687
Ochoa-Izaguirre, M. J., Carballo, J. L., & Páez-Osuna, F. (2002). Qualitative changes in macroalgal assemblages under two contrasting climatic conditions in a subtropical estuary. Botanica Marina, 45, 130–138. https://doi.org/10.1515/BOT.2002.014
Okolodkov, Y. B., Durán-Riveroll, L. M., Band-Schmidt, C. J., Leyva-Valencia, I., Gárate-Lizárraga, I., & Cembella, A. D. (2022). A review on marine benthic dinoflagellates in Mexico. Hidrobiológica, 32, 183–210. https://doi.org/10.24275/uam/izt/dcbs/hidro/2022v32n3/okolodkov
Olivas-Valverde, J. A. (2013). Informe de evaluación e identificación de potenciales riesgos ambientales en flora y fauna en la Bahía de Paracas, provincia de Pisco, departamento de Ica. Organismo de Evaluación y Fiscalización Ambiental. Gobierno de Perú. Available at https://repositorio.oefa.gob.pe/handle/20.500.12788/1214 (Accessed May 28, 2024).
Park, J. S., Li, Z., Kim, H. J., Kim, K. H., Lee, K. W., Youn, J. Y. et al. (2021). First report of the marine benthic dinoflagellate Bysmatrum subsalsum from Korean tidal pools. Journal of Marine Science and Engineering, 9, 649. https://doi.org/10.3390/jmse9060649
Raygoza-Viera, J. R., Ruiz-Fernández, A. C., Ruelas-Inzunza, J., Alonso-Hernández, C., Pérez-Bernal, L. H., & Páez-Osuna, F. (2014). Accumulation and distribution of Hg and 210Pb in superficial sediments from a coastal lagoon in the SE Gulf of California associated with urban-industrial and port activities. Environmental Earth Sciences, 72, 2729–2739. https://doi.org/10.1007/s12665-014-3178-9
R Core Team. (2024). R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/
Reguera, B., Alonso, R., Moreira, A., & Méndez, S. (2011). Guía para el diseño y puesta en marcha de un plan de seguimiento de microalgas productoras de toxinas. Proyecto ARCAL RLA 7/014. París, Viena: UNESCO/ OIEA.
Reyes, J. (2009). Paracas, nuestra Reserva. Información básica sobre la Reserva Nacional de Paracas (2ª ed.). Áreas Costeras y Recursos Marinos, [ACOREMA]. http://humboldt.iwlearn.org/es/sitios-piloto/-1/paracas.pdf
Sanchez-Cabeza, J. A., Álvarez Sánchez, L. F., Cardoso-Mohedano, J. G., Escalante-Mancera, E., Díaz-Asencio, M., López-Rosas, H. et al. (2019). A low-cost long-term model of coastal observatories of global change. Journal of Operational Oceanography, 12, 34–46. https://doi.org/10.1080/1755876X.2018.1533723
Sernanp (Servicio Nacional de Áreas Naturales Protegidas). (2019). Reserva Nacional de Paracas. Servicio Nacional de Áreas Naturales Protegidas por el Estado. Available at: https://www.gob.pe/institucion/sernanp/informes-publicaciones/1718991-reserva-nacional-de-paracas (Accessed October 23, 2024).
SMN (Servicio Meteorológico Nacional). (2024). Normales climatológicas Mazatlán 1951-2017. Available at https://smn.cna.gob.mx/tools/RESOURCES/Diarios/25062.txt
Tester, P. A., Feldman, R. L., Nau, A. W., Kibler, S. R., & Litaker, R. W. (2010). Ciguatera fish poisoning and sea surface temperatures in the Caribbean Sea and the West Indies. Toxicon, 56, 698–710. https://doi.org/10.1016/j.toxicon.2010.02.026
Tester, P. A., Litaker, R. W., & Berdalet, E. (2020). Climate change and harmful benthic microalgae. Harmful Algae, 91, 101655. https://doi.org/10.1016/j.hal.2019.101655
Tester, P. A., Litaker, R. W., Soler-Onís, E., Fernández-Zabala, J., & Berdalet, E. (2022). Using artificial substrates to quantify Gambierdiscus and other toxic benthic dinoflagellates for monitoring purposes. Harmful Algae, 120, 102351. https://doi.org/10.1016/j.hal.2022.102351
Yong, H. L., Mustapa, N. I., Lee, L. K., Lim, Z. F., Tan, T. H., Usup, G. et al. (2018). Habitat complexity affects benthic harmful dinoflagellate assemblages in the fringing reef of Rawa Island, Malaysia. Harmful Algae, 78, 56–68. https://doi.org/10.1016/j.hal.2018.07.009
YSI Inc. (2014). EXO Rhodamine WT Smart Sensor Calibration Guide. Available at: https://www.fondriest.com/pdf/ysi_exo_rhodamine_cal.pdf