Vascular epiphyte community of an inland mangrove of Tabasco: composition and similarity with coastal mangroves and adjacent tropical vegetation
Comunidad epífita vascular de un manglar interior de Tabasco: composición y similitud con manglares costeros y vegetación tropical adyacente
Neil Ebeth Meled Morales-Rodríguez a, *, Carlos Manuel Burelo-Ramos a, José G. García-Franco b, Octavio Aburto-Oropeza c y María Eugenia Molina-Paniagua a
a Universidad Juárez Autónoma de Tabasco, Laboratorio de Manglares Interiores, Carretera Villahermosa-Cárdenas Km. 0.5 s/n, Entronque a Bosques de Saloya, 86150 Villahermosa, Tabasco, Mexico
b Instituto de Ecología, Carretera antigua a Coatepec #351, Col. El Haya, 91073 Xalapa, Veracruz, Mexico
c Scripps Institution of Oceanography, 9500 Gilman Drive, La Jolla, California 92093, USA
*Corresponding author: nemmr@hotmail.com (N.E.M. Morales-Rodríguez)
Received: 13 December 2024; accepted: 23 October 2025
Abstract
El Cacahuate Lagoon, Tabasco, Mexico, is located 170 km from the current coastline and contains an inland mangrove ecosystem, surrounded by remnants of tropical forest. This inland mangrove is the habitat of an epiphyte community whose species composition and relationship with other epiphyte communities are unknown. This study describes the composition of vascular epiphytes associated with this inland mangrove, evaluates their similarity to epiphyte communities in coastal mangroves and adjacent vegetation within a 220 km radius of the study site, using the Jaccard Index and floristic turnover with β-diversity. We hypothesize that the similarity in the composition of the epiphytes of the inland mangrove should be greater with that of nearby forests than with coastal mangroves, since the seeds of forest epiphytes can easily reach the inland mangrove. The epiphyte community of the Cacahuate mangrove comprises 27 species and is more similar to the epiphyte communities of the coastal mangroves of Tabasco. This affinity could be related to the Pleistocene isolation of this mangrove, or to the long-distance dispersal of epiphytes from coastal mangroves.
Keywords: Biodiversity; Floristics; Red mangrove; Rhizophora mangle; Wanha’ Biosphere Reserve
Resumen
La laguna El Cacahuate, Tabasco, México, se localiza a 170 km de la línea costera actual y contiene un ecosistema de manglar interior, rodeado de remanentes de bosque tropical. Este manglar interior es el hábitat de una comunidad epífita cuya composición de especies y su relación con otras comunidades epífitas se desconocen. El presente estudio describe la composición de epífitas vasculares asociadas con este manglar interior, evalúa su similitud con las comunidades epífitas de los manglares costeros y vegetación adyacente en un radio de 220 km del lugar de estudio, utilizando el índice de Jaccard y su recambio florístico con la diversidad ß. Hipotetizamos que la similitud en la composición de las epífitas del manglar interior debería ser mayor con la de los bosques cercanos que con los manglares costeros, ya que las semillas de las epífitas de los bosques pueden llegar fácilmente al manglar interior. La comunidad epífita del manglar del Cacahuate comprende 27 especies y presenta una mayor similitud con las comunidades epífitas de los manglares costeros de Tabasco. Esta afinidad podría estar relacionada con el aislamiento pleistocénico de este manglar, o con la dispersión a larga distancia de las epífitas provenientes de los manglares costeros.
Palabras clave: Biodiversidad; Florística; Mangle rojo; Rhizophora mangle; Reserva de la Biosfera Wanha’
Introduction
Vascular epiphytic plants (henceforth epiphytes) constitute a functional group that develops on woody species. Unlike parasitic plants, epiphytes obtain water and nutrients from the atmosphere, precipitation, and decomposing organic material, rather than relying physiologically on the host (Krömer & Gradstein, 2016). Within epiphytes, 2 main categories are recognized: true epiphytes, which complete their entire life cycle on the host without producing roots that reach the soil, and hemiepiphytes, which in contrast, exhibit a mixed life cycle, since some germinate on the host and later develop roots that reach the ground (primary hemiepiphytes), whereas others begin their growth in the soil and, as they ascend, establish themselves on the host (secondary hemiepiphytes) (Granados-Sánchez et al., 2003; Zotz, 2016). Epiphytes comprise about 10% of the known plant species worldwide (Zotz et al., 2021). In the Neotropics, epiphytes significantly contribute to the overall diversity of the vegetation, as they can represent up to 50% of the vascular flora in some forests (Carmona-Higuita et al., 2025; Kelly et al., 2004; Taylor et al., 1986). Tree species in the low and mountain forests have different structural characteristics (e.g., greater height, dense canopy, and great diversity of bark textures), which combined with particular environmental conditions (e.g., high humidity), allow the presence of a high number of epiphytic species (Ceja-Romero et al., 2008; Flores-Palacios & García-Franco, 2006; Hietz & Hietz, 1995; Krömer et al., 2007, 2014; Martínez-Meléndez et al., 2011; Rzedowski, 1996). In contrast, mangrove forests generally show a low richness of epiphytes (Benzing, 1990; Carmona-Díaz et al., 2014; Zotz & Reuter, 2009; Zimmerman & Olmsted, 1992), which can be attributed to reduced tree diversity and consequently to low structural complexity of the habitat, but mainly due to environmental stress conditions such as the strong winds, waves, and high salinity present in coastal zones (Gómez & Winker, 1991; Jiménez-López et al., 2017; Zotz & Reuter, 2009). However, epiphytes are frequent in mangrove ecosystems, as several species have been recorded in different studies even though the objectives and sampling efforts are directed toward tree species, which are the dominant physiognomic elements (Aksornkoae, 1993; Cach-Pérez et al., 2013; Carmona-Díaz et al., 2014; De Sousa & Colpo, 2017; García-Luna et al 2024; Jiménez-López et al., 2018; Kupec, 2018; Noguera-Savelli et al., 2021; Rahman et al., 2015; Rohani et al., 2020).
Mangrove ecosystems are primarily distributed in tropical and subtropical regions, generally associated with the coast and brackish water bodies (Leal & Spalding, 2021). However, mangrove ecosystems also have been recorded in freshwater wetlands, away from marine influence, known as inland mangroves. The inland mangroves have been reported in many countries worldwide, such as in Australia, Antigua and Barbuda, the Bahamas, Indonesia, and Pakistan, located 15 to 50 km from the coast, and between 6 and 37 m above sea level (Lugo, 1981; Patel, 2014; Patel & Agoramoorthy, 2012; Stoddart et al., 1973; Taylor, 1986; Tripathi et al., 2013; Woodroffe, 1988).
Recently, in Mexico, individuals of Rhizophora mangle, along with another 112 coastal affinity species were reported along the banks of the San Pedro Mártir River and El Cacahuate Lagoon in Tabasco, the latter located almost on the border with Guatemala (Aburto-Oropeza et al., 2021). The presence of this inland mangrove is attributed to the global climatic phenomenon that occurred about 120,000 years ago when an increase in global temperatures caused a rise in sea levels, displacing the coastline inland (Aburto-Oropeza et al., 2021). As the planet cooled, the sea retreated to its current position, leaving mangrove trees dispersed along the riverbanks and creating an isolated mangrove ecosystem in the El Cacahuate Lagoon, 170 km from the Tabasco coast (Aburto-Oropeza et al., 2021).
Currently, the El Cacahuate Lagoon is surrounded by a matrix composed of remnants of evergreen tropical forest belonging to the “Cañón del Usumacinta” Flora and Fauna Protection Zone (Conanp, 2015), fragments of several types of tropical forest, and large areas of land used for livestock and agriculture activities. However, despite this mixed landscape, the hydrophytic vegetation surrounding the water body restricts access to the mangrove, which has resulted in minimal human impact. El Cacahuate mangrove is considered a relict area from the Pleistocene and a refuge for biodiversity due to its high degree of conservation (Aburto-Oropeza et al., 2021). This area together with the adjacent forest areas forms part of a core area of the recently designated Wanha’ Biosphere Reserve (Conanp, 2023a; Semarnat, 2023).
Preliminary surveys indicate the presence of several species of epiphytes along the upper San Pedro River (Aburto-Oropeza et al., 2021). However, in El Cacahuate mangrove the epiphyte species composition and its species similarities to the epiphytic communities of the surrounding vegetation or to the coastal mangroves of Gulf of Mexico are unknown. High epiphyte composition similarity with tropical forest would indicate processes of colonization after the sea retired, while high epiphyte composition with coastal mangroves communities should indicate that species arrived together with the mangrove during the interglacial period.
The aims of this study were to determine the richness and composition of the epiphyte community present in the inland mangrove of El Cacahuate Lagoon, and to assess the floristic similarity of the epiphytes of El Cacahuate with coastal mangroves and adjacent tropical forests. We expect that, given the inland Cacahuate mangrove’s location in a freshwater environment and its proximity to tropical forest areas, the similarity of the epiphyte community should be greater than that of the nearest terrestrial vegetation because the seeds of the epiphytes can likely reach the mangrove trees easily.
Materials and methods
The study was conducted in the El Cacahuate Lagoon, located in the “Sueños de Oro” rural community of the Tenosique Municipality, Tabasco State, Mexico (Fig. 1). It is a lentic water body associated with the sub-basin of the San Pedro Mártir River, situated 170 km in a straight line to the coast and over 300 km following the river’s course (17°17’58.949” N, 91°5’13.09” W, elevation 45 m asl). The region has a warm-humid climate with abundant rainfall in the summer (Aceves-Navarro & Rivera-Hernández, 2019). The average temperature is 27 °C, and the average annual precipitation is 2,264 mm, with September being the rainiest month (391 mm) and March the driest (54 mm) (Conanp, 2023a). The lagoon area is approximately 13.7 ha and reaches a depth of up to 24 m. The soils surrounding the lagoon are clayey loam and gleysol (Conanp, 2023a; Palma-López et al., 2017). In the mangrove, there is a surface layer of leaf litter 15 to 40 cm deep, followed by the mineralization of organic matter.
The vegetation present in the lagoon and around it consists of floodable grassland (60%), aquatic vegetation (15%), and mangrove (25%) (Conanp, 2023a). The floodable grasslands are primarily composed of Hydrocotyle umbellata L. (Araliaceae), Eleocharis interstincta (Vahl) Roem. & Schult. (Cyperaceae), Cladium jamaicense Crantz (Cyperaceae), Acrostichum danaeifolium Langsd. & Fisch. (Pteridaceae), and Bletia purpurea (Lam.) DC (Orchidaceae). The aquatic vegetation includes rooted hydrophytes with floating leaves such as H. umbellata and Nymphaea ampla (Salisb.) DC. (Nymphaeaceae), free-floating hydrophytes like Salvinia auriculata Aubl. (Salvinaceae), and submerged free-floating hydrophytes like Cabomba palaeformis Fassett (Cabombaceae), Utricularia foliosa L., and U. purpurea Walter (Lentibulariaceae). The mangrove is in the southwest part of the lagoon, comprising 7 ha of continuous vegetation. In the mangrove vegetation, R. mangle is the most abundant tree species, with rare occurrences of Bucida buceras L. (Combretaceae), Chrysobalanus icaco L. (Chrysobalanaceae), Haematoxylum campechianum L. (Fabaceae), and Pachira aquatica Aubl. (Malvaceae). Additionally, other species are rarely present such as Vanilla insignis (Orchidaceae), Acoelorraphe wrightii (Griseb. & H.Wendl.) H.Wendl. ex Becc. (Arecaceae), Anthurium schlechtendalii Kunth subsp. schlechtendalii (Araceae), Monstera tuberculata Lundell, and Philodendron radiatum Schott. (Araceae).
Fieldwork was conducted from April 2022 to February 2023. The recording of epiphytes in mangrove vegetation was carried out in 10 rectangular plots of 75 m long by 20 m wide. Plots were oriented perpendicular from the edge of the lagoon to inland spaced 40 m from each other (1,500 m² per plot, 15,000 m² in total; Fig. 1). This design aimed to include as much as possible of the total epiphytic composition present in the mangrove. As R. mangle is the dominant tree species in this mangrove, the recording of epiphyte species focused exclusively on it. Within the sampling plots, we recorded all epiphyte species present on each individual R. mangle tree, excluding hemiepiphytes and parasitic plants. Voucher specimens were collected following standardized methods by Lot and Chiang (1986) and Krömer and Gradstein (2016). For less abundant species, photographic records were obtained instead. All herbarium specimens were deposited in the UJAT herbarium (acronym following Thiers, 2021), and the photographs were stored in the photo archive of the Biological Sciences Division-UJAT.
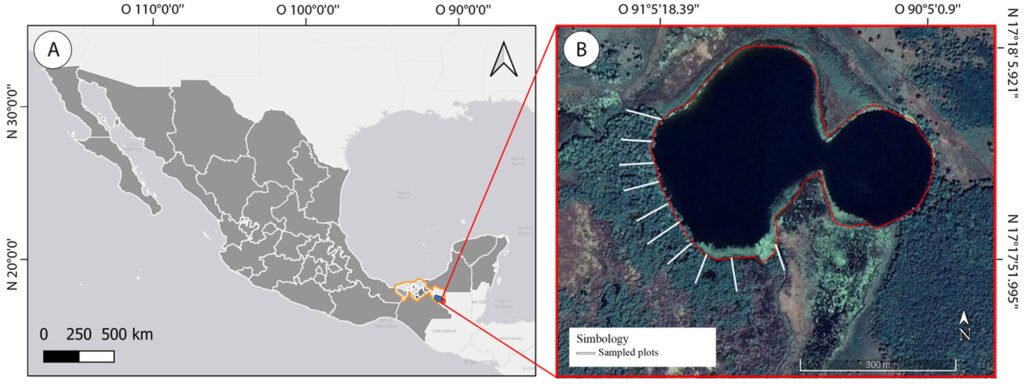
Figure 1. Location of the study area. A) Mexico, highlighting the state of Tabasco (orange) and the municipality of Tenosique (blue). B) El Cacahuate Lagoon (red outline) showing sampling transects (white lines) established in the mangrove vegetation.
The identification of the species was carried out with the support of taxonomic keys and specialized literature for the families Bromeliaceae, Cactaceae, and Orchidaceae (Alderete-Chávez & Capello-García, 1988; Ames & Correll, 1985; Beutelspacher, 2011; Campos-Díaz et al., 2020; Conap, 2010; Davidse et al., 1994; Espejo-Serna et al., 2004; Hágsater et al., 2005; Hietz & Hietz-Seifert, 1994; Martínez-Meléndez et al., 2011; Mondragón-Chaparro et al., 2011; Noguera-Savelli & Cetzal-Ix, 2014), as well as Aspleniaceae, Polypodiaceae, and Pteridaceae (Mickel & Smith, 2004). For species identification, original descriptions were reviewed in the Biodiversity Heritage Library (BHL, 2022). Additionally, to determine the conservation status of the recorded epiphyte species in the study area, we consulted the official Mexican environmental protection and species legislation NOM-059-Semarnat-2010 (Semarnat, 2010) and the Red List of Threatened Species by the International Union for Conservation of Nature (IUCN, 2021).
To determine similarities between the epiphyte species composition of the inland mangrove of El Cacahuate (IM-Cacahuate) and other epiphyte communities, available epiphyte floristic lists from various vegetation types were compiled within a 220 km radius starting from the study area, including sites in Mexico and Guatemala (Fig. 2). Radius length corresponds to the straight-lined distance between El Cacahuate Lagoon and the nearest coastal mangroves of Tabasco, Mexico. It is important to note that most of the studies reviewed were conducted with objectives and sampling efforts not specific to epiphytes, so only the epiphyte species reported were considered (excluding hemiepiphytes).
For the Mexican portion, the compiled lists correspond to coastal mangroves (CM-Paraíso: including localities of Cárdenas, Centla, Comalcalco, Paraíso, Tabasco; Díaz-Jiménez, 2007. CM-Centla: Centla, Tabasco; Jiménez-López et al., 2017, 2018. CM-Nohan: UMA Nohan, Cd. del Carmen, Campeche; Noguera-Savelli et al., 2021); high evergreen tropical forest (HETF-Tenosique: Tenosique, Tabasco, Hernández-Sastré et al., 2014); medium evergreen tropical forest (METF-Macuspana: Macuspana, Tabasco; López-Gómez, 2014; METF-Tenosique: Tenosique, Tabasco; Morales-Damián, 2012); medium subevergreen tropical forest (MSTF-Catazaja: Catazajá, Chiapas; Gutiérrez-Báez, 2004); and tropical dry forest (TDF-Tenosique: Tenosique, Tabasco; Morales-Damián, 2012). Additionally, the riparian flora list of the San Pedro Mártir River was included, which comprises epiphyte records in high, medium, and low evergreen tropical forests with the presence of R. mangle individuals in the riparian vegetation (TF&IM-San Pedro Mártir, Aburto-Oropeza et al., 2021). There are floristic lists that report the presence of epiphyte species for the Calakmul Biosphere Reserve, Campeche, (Conanp, 2023b; Martínez et al., 2001), which were not integrated into the database because this site belongs to the biotic province Yucatán Peninsula (Conanp, 2023b; García-Gil et al., 2002), which reports greater floristic affinity with the districts within the Yucatán Peninsula, Guatemala, and Belize (Duno-de Stefano et al., 2012) and the states of Chiapas, Oaxaca, and Veracruz (Ibarra-Manríquez et al., 2002; Morrone, 2019).
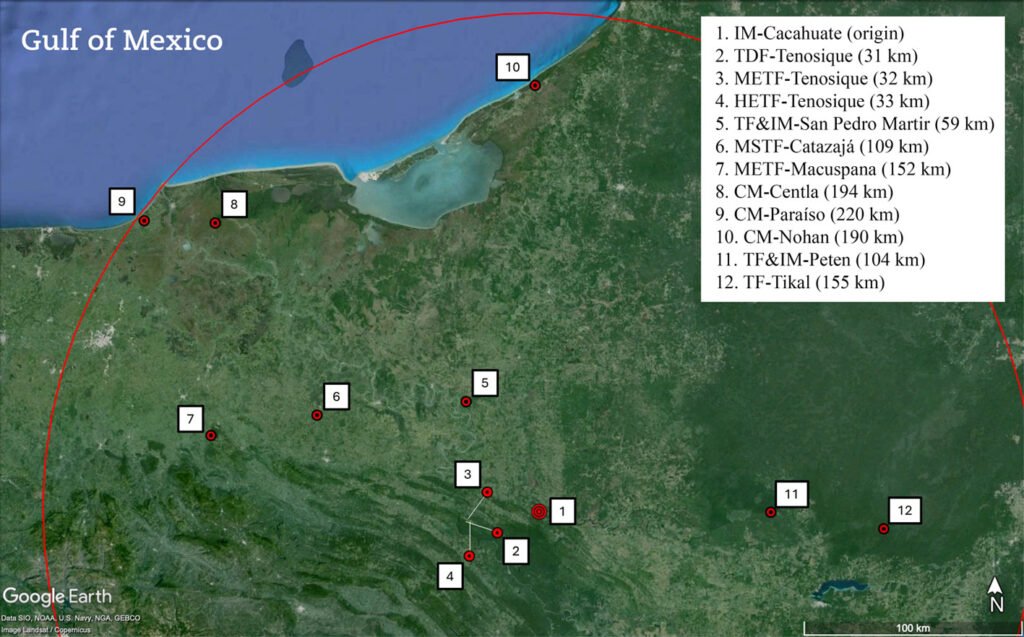
Figure 2. Location of El Cacahuate inland mangrove, the tropical forests, and coastal mangroves within a 220 km radius that include epiphytic floristic lists. The straight-line distance from each site to El Cacahuate is indicated in parentheses. Site acronyms are defined in the Materials and methods section.
In Guatemalan territory, only 2 floristic lists within the established distance for the analysis were obtained: one from a disturbed high evergreen tropical forest (TF-Tikal, Hellmuth & D’Angelo-Jerez, 2022), and the second from a high evergreen tropical forest combined with R. mangle from the El Petén area (TF&IM-Peten, Conap, 2010). Appendix presents the list of the recorded species in the present study and the species compiled from the reviewed studies. The classification of the types of vegetation is based on the proposal of Conabio (2013, 2019) for Chiapas and Tabasco, Mexico, and by Melgar (2003) and Conap (2010) for Tikal and El Petén, Guatemala.
A presence-absence data base was constructed, based on the epiphyte species recorded in our study area and the epiphyte species documented from the surrounding sites obtained of literature. The nomenclature was updated and standardized using the electronic database Tropicos (Tropicos, 2023). Subsequently, the similarity between epiphyte communities of the different vegetation types and locations was explored using the data matrix in a hierarchical clustering analysis (UPGMA) based on the Jaccard similarity index (JSI). This index was selected as it uses species presence-absence data, which was the type of data obtained from the reviewed studies. The measure of similarity ranges from 0% (not similar) to 100% (the most similar). Finally, a beta diversity analysis was performed using Cody’s index (Ci) to assess species turnover between communities. If a high index is obtained, it means that the sites are very different, while a low index means that they share most of their species. Both analyses were conducted using the program Past 4.12b (Hammer et al., 2001).
Results
A total of 6 families, 18 genera, and 27 species of epiphytes were recorded (Table 1)in El Cacahuate mangrove, with Orchidaceae and Bromeliaceae being the families with the highest number of species (11 and 10, respectively), followed by Cactaceae and Polypodiaceae (2 species each) and Aspleniaceae and Pteridaceae (1 species each; Fig. 3). The genera with the most species were Tillandsia (Bromeliaceae) with 8 species, and Epidendrum (Orchidaceae) with 3. Among the epiphyte species recorded in El Cacahuate, Asplenium serratum L. is listed as a threatened species under Mexican legislation (Semarnat, 2010), while Tillandsia fasciculata Sw. and Selenicereus grandiflorus (L.) Briton & Rose are cited on the Red List of Threatened Species by IUCN (2021) as species of least concern.
Table 1
List of epiphyte species associated with the mangrove of El Cacahuate Lagoon, Tenosique, Tabasco, Mexico. *New records for the Wanha’ Biosphere Reserve.
| Family | Species |
| Aspleniaceae | Asplenium serratum L.* |
| Bromeliaceae | Aechmea bracteata (Sw.) Griseb. |
| Catopsis morreniana Mez | |
| Tillandsia balbisiana Schult. & Schult.f. | |
| T. brachycaulos Schltdl. | |
| T. bulbosa Hook. | |
| T. dasyliriifolia Baker* | |
| T. fasciculata Sw. | |
| T. juncea (Ruiz & Pav.) Poir. | |
| T. schiedeana Steud. | |
| T. streptophylla Scheidw. Ex E.Morren | |
| Cactaceae | Deamia testudo (Karw. Ex Zucc.) Britton & Rose |
| Selenicereus grandiflorus (L.) Briton & Rose | |
| Orchidaceae | Catasetum integerrimum Hook. |
| Cohniella ascendens (Lindl.) Christenson | |
| Encyclia bractescens (Lindl.) Hoehne | |
| Epidendrum cardiophorum Schltr. | |
| E. diffusum Sw.* | |
| E. nocturnum Jacq. | |
| Myrmecophila tibicinis (Bateman) Rolfe | |
| Nidema boothi (Lindl.) Schltr.* | |
| Notylia barkerii Lindl.* | |
| Oncidium sphacelatum Lindl. | |
| Specklinia grobyi (Bateman ex Lindl.) F.Barros | |
| Polypodiaceae | Microgramma nítida (J. Sm.) A.R. Sm.* |
| Polypodium polypodioides (L.) Watt* | |
| Pteridaceae | Vittaria lineata (L.) Sm. |
Similarities between epiphyte communities. The present study and the review of published studies within the selected surrounding area allowed for the compilation of a total of 119 epiphyte species from sites with coastal mangrove, inland mangrove, tropical forests, and disturbed forests (Appendix). The sites with the highest number of epiphyte species were the high evergreen tropical forest of Tenosique (HETF-Teno, 53 species), the riparian vegetation of the San Pedro River (TF&IM-SPMR, 52 species), the medium evergreen tropical forest of Macuspana (METF-Macus, 45 species), the inland mangrove of El Cacahuate Lagoon (IM-Cac, 27 species), and the coastal mangrove of Centla (CM-Centla, 25 species). In the remaining sites, the number of epiphyte species ranged from 4 to 16, with 14 species recorded in the coastal mangroves of Nohan and Paraíso (CM-Nohan, CM-Paraíso; Appendix).
The clustering analysis (Fig. 4) clearly separated 4 groups (at a similarity level of 0.14). Two groups consisted of a single location: the first being the disturbed evergreen tropical forest of Tikal (TF-Tikal) and the second, the medium subevergreen tropical forest of Catazaja, Chiapas (MSTF-Catazaja). The other 2 groups contained several locations. The first group was made up of different forest types in Tabasco (HETF-Tenosique, METF-Macuspana, TDF-Tenosique, METF-Tenosique), while the second group included sites with coastal mangroves (CM-Nohan, CM-Paraíso, CM-Centla) and tropical forest and inland mangrove vegetation (TF&IM-San Pedro Mártir, TF&IM-Peten, IM-Cacahuate). In this latter group, the inland mangrove of El Cacahuate showed greater similarity in the epiphyte species with the coastal mangroves of Centla (JSI = 0.45, 16 shared species) and Paraíso (JSI = 0.38, 11 shared species), despite being 194 and 220 km away from El Cacahuate, respectively. The tropical forest and mangrove vegetation of the San Pedro River shared a greater number of species (19) with the El Cacahuate mangrove, but the similarity was slightly lower (JSI = 0.23), although the distance between these sites is lower, 59 km. The next ecosystem is the forest with inland mangrove of El Petén, sharing 8 species with El Cacahuate and showing a lower similarity than the previous groups (JSI = 0.18). At last, the coastal mangrove of Nohan, Campeche shared 7 species with El Cacahuate, with the lower similarity inner this group (JSI = 0.15).
Among all groups, the lowest similarity of the epiphyte community of El Cacahuate was with the medium subevergreen tropical forest of Catazaja (JSI = 0.05) and the disturbed evergreen tropical forest of Tikal (JSI = 0.03), with 2 and 1 species in common, respectively; both located over 100 km from El Cacahuate Lagoon. Among sites with mangrove vegetation, beta diversity analysis (Table 2) shows that the inland mangrove of El Cacahuate presents low species turnover with the coastal mangroves (Centla and Paraíso; Ci= 10 and 9.5, respectively). In contrast, species turnover is higher with the riparian forest and mangrove vegetation of the San Pedro Mártir River (Ci= 19).

Figure 3. Representative epiphyte species recorded in the inland mangrove of El Cacahuate Lagoon, Tenosique, Tabasco, Mexico. A) Microgramma nitida, B) Vittaria lineata, C) Asplenium serratum, D) Polypodium polypodioides, E) Deamia testudo, F) Selenicereus grandiflorus, G) Catopsis morreniana, H) Tillandsia balbisiana, I) T. bulbosa, J) T. streptophylla, K) T. dasyliriifolia, L) T. schiedeana, M) T. fasciculata, N) Epidendrum cardiophorum, O) E. nocturnum, P) E. diffusum, Q) Encyclia bractescens, R) Nidema boothi, S) Notylia barkerii, and T) Specklinia grobyi. Photographs by Jesús Rodríguez and Neil Morales.
Discussion
As far as we know, our study is the first to be conducted on epiphytic species of inland mangroves in El Cacahuate Lagoon. The epiphyte community of El Cacahuate Lagoon, with 27 records species, is among the richest ever reported in a mangrove ecosystem. Previous studies have reported the presence of fewer species in coastal mangroves (De Sousa & Colpo, 2017; Díaz-Jiménez, 2007; Jiménez-López et al., 2017, 2018; Menéndez-Liguori, 1976; Noguera-Savelli et al., 2021; Rohani et al., 2020). However, it is true that many of these studies did not involve extensive sampling, or they were not specifically focused on epiphyte species. Jiménez-López et al. (2017, 2018), in the mangrove of El Cometa Lagoon, Centla, Tabasco, were specifically focused on epiphytes, recording 25 species. In contrast, the tropical forest sites of Tenosique and Macuspana (Hernández-Sastré et al., 2014; López-Gómez, 2014; Morales-Damián, 2012), and the riparian forest and mangrove of San Pedro Mártir, Tabasco (Aburto-Oropeza et al., 2021), have a higher reported number of epiphyte species than the mangrove of El Cacahuate. However, these differences in the number of species reported are because the forests of Tenosique, Macuspana, and the San Pedro Mártir River were collected in larger areas of vegetation, a greater number of phorophyte species were sampled, and different sampling methods were used. For example: A) in the forests of Tenosique (Morales-Damián, 2012; Hernández-Sastré et al., 2014) sampled 25 × 25 m plots, recording the epiphytes associated with 9 species of phorophytes; B) in the Macuspana forest (López-Gómez, 2014), a representative sampling method of 1 ha was used, sampling epiphytes in 8 plots of 20 × 20 m (400 m²), considering both trees and shrubs present; and finally C) on the San Pedro Mártir River (Aburto-Oropeza et al., 2021), targeted sampling was carried out between 2014 and 2015 with sporadic collections until 2019, covering more than 25 linear km of river and sampling more than 30 species of phorophytes (Burelo-Ramos pers. com.). This shows that floristic lists targeting forests tend to have higher reports of epiphytes than mangrove ecosystems.
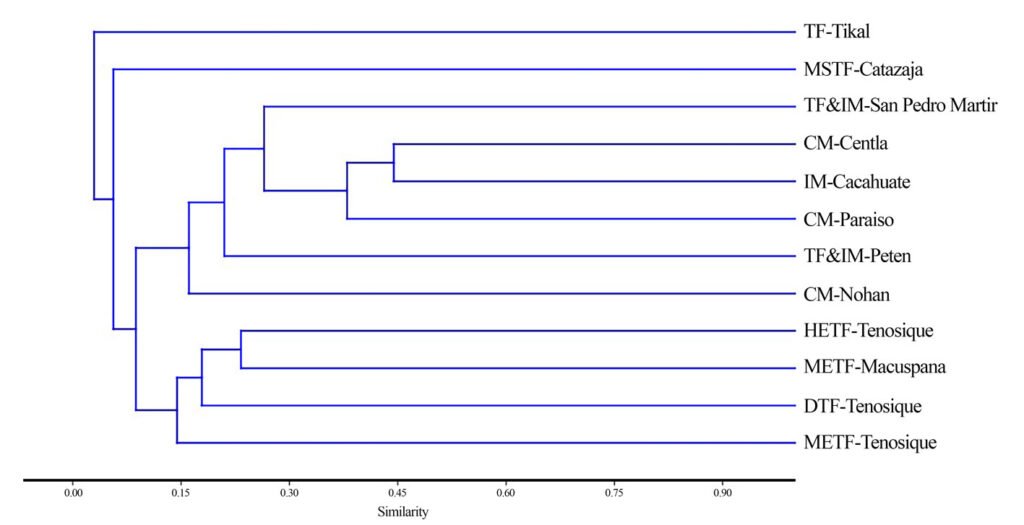
Figure 4. Dendrogram showing the similarity of epiphyte communities among the El Cacahuate Lagoon mangrove, coastal mangroves, and adjacent forest vegetation sites. Site acronyms are defined in the Materials and Methods section.
Table 2
Beta-diversity of epiphytes between the inland mangrove of El Cacahuate Lagoon and epiphyte floristic lists of surrounding tropical forest and coastal mangroves. Values above the diagonal represent Cody’s index for each pairwise comparison. Values below the diagonal indicate the number of shared species between sites. The top row displays the total species richness for each location. The first column lists the number of species unique to each site. Site acronyms are defined in the Materials and methods section.
| Total | 27 | 11 | 9 | 47 | 51 | 4 | 43 | 25 | 14 | 14 | 16 | 5 | |
| Unique | sites | IM-Cacahuate | TDF-Tenosique | METF-Tenosique | HETF-Tenosique | TF&IM-San Pedro | MSTF-Catazaja | METF-Macuspana | CM-Centla | CM-Paraíso | CM-Nohan | TF&IM-Peten | TF-Tikal |
| 0 | IM-Cacahuate | 12 | 15 | 23 | 19 | 13.5 | 29 | 10 | 9.5 | 13.5 | 13.5 | 15 | |
| 0 | TDF-Tenosique | 7 | 8 | 19 | 25 | 6.5 | 20 | 15 | 10.5 | 11.5 | 13.5 | 8 | |
| 0 | METF-Tenosique | 3 | 2 | 19 | 27 | 6.5 | 20 | 16 | 9.5 | 10.5 | 12.5 | 7 | |
| 11 | HETF-Tenosique | 14 | 10 | 9 | 32 | 24.5 | 28 | 29 | 24.5 | 26.5 | 28.5 | 24 | |
| 14 | TF&IM-SPMR | 20 | 6 | 3 | 17 | 24.5 | 32 | 23 | 21.5 | 26.5 | 24.5 | 25 | |
| 1 | MSTF-Catazaja | 2 | 1 | 0 | 1 | 3 | 21.5 | 11.5 | 8 | 7 | 10 | 4.5 | |
| 15 | METF-Macuspana | 5 | 6 | 6 | 17 | 15 | 2 | 28 | 26.5 | 26.5 | 29.5 | 24 | |
| 3 | CM-Cent | 16 | 3 | 1 | 7 | 15 | 3 | 6 | 8.5 | 11.5 | 13.5 | 14 | |
| 1 | CM-Para | 11 | 2 | 2 | 6 | 11 | 1 | 2 | 11 | 10 | 9 | 8.5 | |
| 5 | CM-Nohan | 7 | 1 | 1 | 4 | 6 | 2 | 2 | 7 | 4 | 13 | 9.5 | |
| 4 | TF&IM-Peten | 8 | 0 | 0 | 3 | 9 | 0 | 0 | 7 | 6 | 2 | 8.5 | |
| 1 | TF-Tikal | 1 | 0 | 0 | 2 | 3 | 0 | 0 | 1 | 1 | 0 | 2 |
The low richness of epiphytes in coastal mangroves has been associated with the adverse environmental conditions present, such as high temperatures, low precipitation, and excess salinity (Benzing, 1990; Carmona-Díaz et al., 2014; García-Luna et al., 2024; Gómez & Winker, 1991; Zimmerman & Olmsted, 1992; Zotz & Reuter, 2009). Mangrove trees eliminate excess salt through their leaves (Mikolaev et al., 2016; Zotz & Reuter, 2009), so large amounts of salt could be accumulating in some epiphytes (e.g., orchids and bromeliads) due to leaf litter and rainwater falling from the canopy, which is captured in the roots and the rosette leaves of these plants. Salt decreases the osmotic potential of epiphyte cells, reducing their survival by promoting tissue necrosis and leaf loss, especially for species that do not have adaptations to survive under extreme water stress conditions, v.gr., xerophytic habits (Benzing, 2000; Du & Hesp, 2020; Zotz & Reuter, 2009). However, Gómez and Winkler (1991) have pointed out a contrary position suggesting that the high precipitation in some coastal areas may dilute the high salinity that falls from the mangrove’s canopy, allowing for the existence of non-xerophytic epiphytes. Nevertheless, the low presence of epiphyte species in mangrove ecosystems suggests that the physiological limitations in many of these plants may be significant (Cach-Pérez et al., 2018). Considering the distribution of mangroves worldwide, more floristic studies are needed in this ecosystem to identify patterns in other epiphytes species numbers, physiological traits, and functional groups.
The mangrove of El Cacahuate Lagoon presents a less stressful environment compared to coastal mangroves, as it is in a freshwater body with high precipitation (annual average of 2,300 mm) and warm temperatures (annual average of 26 °C) (Aceves-Navarro & Rivera-Hernández, 2019). In coastal mangroves, which are characterized as saline ecosystems (Feller et al., 2010), R. mangle eliminates the excess of salt absorbed by its roots through lenticels on its leaves (Bento et al., 2024). However, although El Cacahuate Lagoon is a freshwater aquatic system (0.4 psu) compared to nearby coastal sites (22 psu), calcium carbonate from mountain runoff has been reported in the area (Aburto-Oropeza et al., 2021; Conanp, 2023a) and the sap salinity of the plants is like that of trees in coastal areas (J. López-Portillo pers. com.). This suggests that the R. mangle trees in El Cacahuate Lagoon still exude salt through the leaves. However, atmospheric humidity and high precipitation could dilute the concentration of salts reaching the epiphytes (J. López-Portillo pers. com.). These conditions seem to favor the presence of orchid, bromeliad, and fern species recorded at this site; it also suggests that the species shared between the inland mangrove and coastal mangroves have adapted to both environmental conditions. Specific ecophysiological studies of the shared and not shared epiphyte species between this inland mangrove and the coastal mangroves will provide a better understanding of the characteristics that limit and promote the distribution of epiphytes in these ecosystems.
Similarities between epiphyte communities. Given the proximity of the Tenosique mountain (ca. 2-32 km), we expected that the species composition of the study area showed greater similarity in epiphyte composition with the surrounding forests (HETF, METF, DTF). It has been recorded that the structure, anatomy, and seed dispersal mechanisms of many epiphytes permit frequent colonization and recolonization processes between nearby tropical sites (Chilpa-Galván et al., 2018; Toledo-Aceves et al., 2012). Nevertheless, we found the highest similarity of El Cacahuate mangrove’s epiphyte species with the epiphyte communities of the coastal mangroves of Centla and Paraíso, located 194-200 km away. These facts suggest 2 possible hypotheses which can help to explain these similarities: the first suggests that when the sea encroached 120,000 years ago, not only R. mangle individuals and other terrestrial coastal species established along the banks of the San Pedro River and El Cacahuate Lagoon, but this process was accompanied by the coastal-affinity epiphytes recorded in the study area; the second one points to contemporary dispersal processes as the reason for the presence of these epiphytes with a coastal distribution. It has been documented that after ca. 30 years, 30% of the original epiphyte species recorded in mangrove trees in the natural part of a canal, colonized juvenile mangroves planted for reforestation in the artificial part of the canal (Kupec, 2018). This suggests that the epiphyte species with coastal affinity may have arrived at the inland mangrove of El Cacahuate Lagoon by seed step-dispersal and colonization events throughout the mangrove established along the San Pedro River, or by long-distance dispersal events of minute seed from the coastal mangroves (Schurr et al., 2009). The other epiphyte species with extensive tropical forest distribution surely have arrived by both dispersal ways.
These 2 processes do not exclude each other, and their success can be highly dependent on the number of viables seeds and habitat availability (Rodríguez-Romero et al., 2011). However, successful establishment also depends on the morphological and anatomical characteristics of the seeds (Arditti & Ghani, 2000; Chilpa-Galván et al., 2018). For instance, some ferns families (e.g., Aspleniaceae, Polypodiaceae, and Pteridaceae) produce many very small spores that are easily dispersed by wind over long distances (Martínez-Salas & Ramos, 2014; Perrie et al., 2010; Shepherd et al., 2009). Similarly, orchid fruits (Orchidaceae) produce millions of tiny seeds, often just a few microns in size and weighing no more than 22 mcg (Arditti & Ghani, 2000; Menchaca-García & Moreno-Martínez, 2011), which are also adapted for long-distance wind dispersal. Many bromeliad species (Bromeliaceae, subfamily Tillandsoideae) have plumose seed appendages that facilitate their movement through the air and their attachment to rough bark surfaces (Einzmann & Zotz, 2017; Mondragón-Chaparro et al., 2011). Meanwhile, some species such as A. bracteata, which produce berry-like fruits, and cacti such as S. grandiflorus and D. testudo, which have pulpy fruits, are dispersed by birds. This suggests that short- or long-distance seed dispersal has contributed to structuring the epiphyte community in the inland mangrove of El Cacahuate Lagoon. Future genetic studies will reveal how closely related the populations of shared epiphyte species are between the inland mangroves of El Cacahuate Lagoon and the coastal mangroves of Tabasco, particularly for those species with a wide distribution range.
Our study clearly shows that the epiphyte community in the inland mangroves of El Cacahuate Lagoon is species-rich. The humid environmental conditions present apparently reduce the salinity concentration exuded by the mangrove trees, allowing the great epiphyte species richness recorded. Also, we have demonstrated that this epiphyte community has a high similarity in composition to the epiphyte communities of the coastal mangroves of Tabasco, suggesting a close genetic relationship between these populations. Future genetic studies will help elucidate the dispersal routes followed by the epiphytes to colonize this mangrove. These and other biological characteristics make this inland mangrove ecosystem one of great biodiversity and conservation value.
Acknowledgments
Special thanks to the UJAT herbarium for access to its facilities and the support provided during the cabinet phase. We are grateful to M. Ferrer, J. Rodríguez, and E. López for their help with the fieldwork. Special thanks to W. Miss and his family for their hospitality and for facilitating access to the study site. Thanks to S. Morales, C. Rodríguez, and I. Morales for providing transportation to the study site. We extend our gratitude to F. Jiménez H. and R. Adams for reviewing early drafts of this manuscript. This study is part of the requirements for obtaining a master’s degree for NEM. The work was supported by the Universidad Juárez Autónoma de Tabasco through the project “Biodiversity and conservation of the inland mangroves of the San Pedro Mártir River as elements for sustainable development in Balancán and Tenosique, Tabasco, Mexico” (No. 20220327), and the Centro para la Biodiversidad Marina y la Conservación, A.C. and Mares Mexicanos. The Consejo Nacional de Humanidades, Ciencia y Tecnología (Conahcyt) granted a scholarship to the first author for his master’s program.
Appendix. Checklist of vascular epiphytes recorded in the inland mangrove of El Cacahuate Lagoon, and compiled data from the floristic studies completed in a radius of 220 km from El Cacahuate Lagoon. Acronyms: IM-Cacahuate, inland mangrove of El Cacahuate, Tenosique, Tabasco; CM-Paraíso, coastal mangrove of Cárdenas, Centla, Comalcalco, Paraíso, Tabasco (Díaz-Jiménez, 2007); CM-Centla, coastal mangrove of Centla, Tabasco (Jiménez-López et al., 2017, 2018); CM-Nohan, coastal mangrove of the UMA Nohan, Cd. del Carmen, Campeche (Noguera-Savelli et al., 2021); HETF-Tenosique, high evergreen tropical forest of Tenosique, Tabasco (Hernández-Sastré et al., 2014); METF-Tenosique, medium evergreen tropical forest of Tenosique, Tabasco (Morales-Damián, 2012); METF-Macuspana: Macuspana, Tabasco; López-Gómez 2014); MSTF-Catazaja: medium subevergreen tropical forest of Catazajá, Chiapas (Gutiérrez-Báez, 2004); TDF-Tenosique: tropical dry forest of Tenosique, Tabasco (Morales-Damián, 2012); TF&IM-San Pedro Mártir: tropical forests with the presence of R. mangle individuals in the riparian vegetation of the San Pedro Mártir River (Aburto-Oropeza et al., 2021); TF-Tikal: disturbed high evergreen tropical forest (Hellmuth and D’Angelo-Jerez, 2022); TF&IM-Peten: high evergreen tropical forest combined with R. mangle from the El Petén area (Conap, 2010).
| Families | Species /sites | IM-Cacahuate | TDF-Tenosique | METF-Tenosique | HETF-Tenosique | TF&IM-San Pedro Mártir | MSTF-Catazaja | METF-Macuspana | CM-Centla | CM-Paraíso | CM-Nohan | TF&IM-Petén | TF-Tikal |
| Aspleniaceae | Asplenium auritum Sw. | x | |||||||||||
| Asplenium serratum L. | x | x | |||||||||||
| Bromeliaceae | Aechmea bracteata (Sw.) Griseb. | x | x | x | x | x | x | ||||||
| Aechmea mexicana Baker | x | ||||||||||||
| Aechmea tillandsoides (Mart. ex Schult. & Schult.f.) Baker | x | ||||||||||||
| Billbergia viridiflora H.Wendl. | x | ||||||||||||
| Catopsis morreniana Mez | x | x | |||||||||||
| Catopsis nutans (Sw.) Griseb. | x | ||||||||||||
| Tillandsia anceps G. Lodd. | x | ||||||||||||
| Tillandsia balbisiana Schult. & Schult.f. | x | x | x | x | x | x | |||||||
| Tillandsia brachycaulos Schltdl. | x | x | x | x | x | ||||||||
| Tillandsia bulbosa Hook. | x | x | x | x | x | ||||||||
| Tillandsia chlorophylla L. B. Smith | x | ||||||||||||
| Tillandsia dasyliriifolia Baker | x | x | x | x | |||||||||
| Tillandsia fasciculata Sw. | x | x | x | x | |||||||||
| Tillandsia festucoides Brongn. Ex Mez | x | x | |||||||||||
| Tillandsia filifolia Schldtl. & Cham. | x | x | |||||||||||
| Tillandsia ionantha Planch | x | ||||||||||||
| Tillandsia juncea (Ruiz & Pav.) Poir. | x | x | x | x | |||||||||
| Tillandsia recurvata (L.) L. | x | ||||||||||||
| Tillandsia polystachia (L.) L. | x | ||||||||||||
| Tillandsia pseudobaileyi C.S. Gardner | x | ||||||||||||
| Tillandsia pruinosa Sw. | x | ||||||||||||
| Tillandsia schiedeana Steud. | x | x | x | x | x | ||||||||
| Tillandsia streptophylla Scheidw. Ex E.Morren | x | x | x | x | x | x | |||||||
| Tillandsia usneoides (L.) L. | x | x | x | x | x | ||||||||
| Tillandsia valenzuelana A. Rich | x | ||||||||||||
| Cactaceae | Deamia testudo (Karw. Ex Zucc.) Britton & Rose | x | x | x | x | x | x | ||||||
| Pseudorhipsalis ramulosa (Salm-Dyck) Barthlott | x | ||||||||||||
| Epiphyllum crenatum (Lindl.) G.Don | x | ||||||||||||
| Epiphyllum hookeri Haw. | x | x | |||||||||||
| Epiphyllum phyllanthus (L.) Haw. | x | x | x | ||||||||||
| Rhipsalis baccifera (J.S. Muell.) Stearn | x | x | |||||||||||
| Selenicereus donkelaarii (Salm-Dick) Britton & Rose | x | ||||||||||||
| Selenicereus grandiflorus (L.) Briton & Rose | x | x | x | x | |||||||||
| Selenicereus undatus (Haw.) D.R. Hunt | x | ||||||||||||
| Gesneriaceae | Columnea schiedeana Schltdl. | x | |||||||||||
| Orchidaceae | Acianthera hondurensis (Ames) Pridgeon & M. W. Chase | x | x | ||||||||||
| Brassavola nodosa (L.) Lindl. | x | x | |||||||||||
| Brassia caudata (L.) Lindl. | x | x | |||||||||||
| Brassia maculata R. Br. | x | x | |||||||||||
| Camaridium pulchrum Schltr. | x | x | |||||||||||
| Campylocentrum micranthum (Lindl.) Maury | x | x | |||||||||||
| Catasetum integerrimum Hook. | x | x | x | x | x | x | x | x | |||||
| Chysis bractescens Lindl. | x | x | x | ||||||||||
| Coelia triptera (Sm.) G. Don ex Steud. | x | x | x | ||||||||||
| Cohniella ascendens (Lindl.) Christenson | x | x | x | x | x | x | x | ||||||
| Cohniella cosymbephorum (C. Morren) R. Jiménez & Carnevali | x | ||||||||||||
| Cohniella lindenii (Brongn.) M.W.Chase & N.H.Williams | x | x | |||||||||||
| Cohniella luridum (Lindl.) M.W. Chase & N.H. Williams | x | x | x | ||||||||||
| Cohniella Yucatánensis Cetzal & Carnevali | x | ||||||||||||
| Coryanthes picturata Rchb.f. | x | x | |||||||||||
| Cycnoches ventricosum Bateman | x | ||||||||||||
| Dichaea muricatoides Hamer & Garay | x | ||||||||||||
| Dichaea panamensis Lindl. | x | ||||||||||||
| Encyclia alata (Bateman) Schltr. | x | x | x | ||||||||||
| Encyclia bractescens (Lindl.) Hoehne | x | x | x | x | |||||||||
| Encyclia guatemalensis (Klotzsh) Dressler & G.E. Pollard | x | ||||||||||||
| Epidendrum anceps Jacq. | x | ||||||||||||
| Epidendrum cardiophorum Schltr. | x | x | x | ||||||||||
| Epidendrum difforme Jacq. | x | x | |||||||||||
| Epidendrum diffusum Sw. | x | x | x | ||||||||||
| Epidendrum flexuosum G. Mey. | x | x | |||||||||||
| Epidendrum nocturnum Jacq. | x | x | x | x | x | ||||||||
| Epidendrum stamfordianum Bateman | x | x | x | ||||||||||
| Gongora leucochila Lem. | x | x | |||||||||||
| Gongora unicolor Rolfe | x | ||||||||||||
| Isochilus carnosiflorus Lindl. | x | x | x | ||||||||||
| Lophiaris lindenii (Brongn.) Braem | x | x | |||||||||||
| Lophiaris oerstedii (Rchb. f.) R. Jiménez Carnevali & Dressler | x | x | x | x | x | ||||||||
| Lophiaris teaboana R. Jiménez, Carnevali & Tapia Muñoz | x | ||||||||||||
| Lycaste aromatica (Graham) Lindl. | x | ||||||||||||
| Maxillaria aciantha Rchb. f. | x | ||||||||||||
| Maxillaria crassifolia (Lindl.) Rchb. f. | x | x | x | x | |||||||||
| Maxillaria elatior (Rchb. f.) Rchb. f. | x | x | |||||||||||
| Maxillaria hedwigiae Hamer & Dodson | x | ||||||||||||
| Maxillaria tenuifolia Lindl. | x | x | |||||||||||
| Maxillaria uncata Lind. | x | ||||||||||||
| Maxillaria variabilis Bateman ex Lindl. | x | x | |||||||||||
| Myrmecophila christinae var. christinae Carnevali & Gómez-Juárez | x | ||||||||||||
| Myrmecophila tibicinis (Bateman) Rolfe | x | x | x | x | x | x | |||||||
| Nemaconia striata (Lindl.) Van den Berg, Salazar & Soto Arenas | x | x | |||||||||||
| Nidema boothi (Lindl.) Schltr. | x | x | x | x | x | x | |||||||
| Notylia barkeri Lindl. | x | x | x | x | x | x | |||||||
| Notylia orbicularis A.Rich. | x | ||||||||||||
| Oncidium sphacelatum Lindl. | x | x | x | x | x | ||||||||
| Ornithocephalus inflexus Lindl. | x | x | |||||||||||
| Platystele minimiflora (Schltr.) Garay | x | ||||||||||||
| Platythelys querceticola (Lindl.) Garay | x | ||||||||||||
| Platystele stenostachya (Rchb.f.) Garay | x | ||||||||||||
| Polystachya caracasana Rchb.f. | x | ||||||||||||
| Prosthechea cochleata (L.) W.E.Higgins | x | x | x | ||||||||||
| Prosthechea boothiana (Lindl.) W.E.Higgins | x | ||||||||||||
| Prosthechea livida (Lindl.) W.E.Higgins | x | ||||||||||||
| Prosthechea pygmaea (Hook.) W. E. Higgins | x | ||||||||||||
| Prosthechea radiata (Lindl.) W. E. Higgins | x | ||||||||||||
| Restrepiella ophiocephala (Lindl.) Garay & Dunst. | x | ||||||||||||
| Sarcinula brighamii (S. Watson) Luer | x | ||||||||||||
| Scaphyglottis confusa (Schltr.) Ames & Correll | x | x | |||||||||||
| Specklinia grobyi (Bateman ex Lindl.) F.Barros | x | x | |||||||||||
| Specklinia picta (Lindl.) Pridgeon & M.W. Chase | x | ||||||||||||
| Specklinia pisinna (Luer) Solano y Soto Arenas | x | ||||||||||||
| Stelis gracilis Ames | x | ||||||||||||
| Tribulago tribuloides (Sw.) Luer | x | ||||||||||||
| Trichosalpinx ciliaris (Lindl.) Luer | x | ||||||||||||
| Trigonidium egertonianum Bateman ex Lindl. | x | x | x | x | |||||||||
| Piperaceae | Peperomia angustata Kunth | x | |||||||||||
| Peperomia cobana C.DC. | x | ||||||||||||
| Peperomia glutinosa Millsp. | x | ||||||||||||
| Peperomia obtusifolia (L.) A.Dietr. | x | ||||||||||||
| Peperomia quadrifolia (L.) Kunth | x | ||||||||||||
| Peperomia rotundifolia (L.) Kunth | x | ||||||||||||
| Polypodiaceae | Campyloneurum angustifolium (Sw.) Fée | x | |||||||||||
| Neurodium lanceolatum (L.) Fée | x | ||||||||||||
| Microgramma nitida (J. Sm.) A.R. Sm. | x | x | |||||||||||
| Phlebodium decumanum (Willd.) J. Sm. | x | ||||||||||||
| Polypodium polypodioides (L.) Watt | x | x | x | ||||||||||
| Pteridaceae | Antrophyum ensiforme Hook. | x | |||||||||||
| Vittaria lineata (L.) Sm. | x | x | x |
References
Aburto-Oropeza, O., Burelo-Ramos, C. M., Ezcurra, E., Ezcurra, P., Henriquez, C. L., Vanderplank, S. et al. (2021). Relict inland mangrove ecosystem reveals Last Interglacial sea levels. Proceedings of the National Academy of Sciences, 118, 1–8. https://doi.org/10.1073/pnas.2024518118
Aceves-Navarro, L. A., & Rivera-Hernández, B. (2019). Clima. In A. Cruz Angón (Ed.), La biodiversidad del estado de Tabasco. Estudio de estado (pp. 123–132). México, D.F.: Comisión Nacional para el Conocimiento y Uso de la Biodiversidad.
Aksornkoae, S. (1993). Ecology and management of mangroves. Bangkok, Thailand: International Union for Conservation of Nature (IUCN). https://portals.iucn.org/library/sites/library/files/documents/WTL-024.pdf
Alderete-Chávez, A., & Capello-García, S. (1988). Orquídeas de Tabasco. Villahermosa: Instituto Nacional de Investigaciones sobre Recursos Bióticos/ Instituto de Cultura de Tabasco.
Ames, O., & Correll, D. S. (1985). Orchids of Guatemala and Belize. New York: Dover Publications.
Arditti, J., & Ghani, A. K. A. (2000). Numerical and physical properties of orchid seeds and their biological implications. New Phytologist, 145, 367–421. https://doi.org/10.1046/j.1469-8137.2000.00587.x
Bento, J. P. S. P., Victório, C. P., Plaça, L. F., Arruda, R. C. O. (2024). Salt extrusion, a new function of lenticels in red mangrove leaves. Environmental and Experimental Botany, 223, 05798. https://doi.org/10.1016/j.envexpbot.2024.105798
Benzing, D. H. (1990). Vascular epiphytes: general biology and related biota. Cambridge, UK: Cambridge University Press.
Benzing, D. H. (2000). Bromeliaceae: profile of an adaptative radiation. Cambridge, UK: Cambridge University Press.
Beutelspacher, C. R. (2011). Guía de orquídeas de Chiapas. Tuxtla Gutiérrez, Chiapas: Universidad de Ciencias y Artes de Chiapas. Chiapas
BHL (Biodiversity Heritage Library) (2022). Available at: https://www.biodiversitylibrary.org
Cach-Pérez, M. J., Andrade, J. L., Chilpa-Galván, N., Tamayo-Chim, M., Orellana, R., & Reyes-García, C. (2013). Climatic and structural factors influencing epiphytic bromeliad community assemblage along a gradient of water-limited environments in the Yucatán Peninsula, Mexico. Tropical Conservation Science, 6, 283–302. https://doi.org/10.1177/194008291300600209
Campos-Díaz, M. J., Burelo-Ramos, C. M., & Arias, S. (2020). La familia Cactaceae en Tabasco, México. Acta Botanica Mexicana, 127, e1635. https://doi.org/10.21829/abm127.2020.1635
Carmona-Díaz, G., Hernández-Carmona, S., Morales-Mávil, J. E., Hernández-Salazar, L. T., & García-Orduña, F. (2014). Contribución al conocimiento ecológico de la epífita vascular Aechmea bracteata (Bromeliaceae) en el manglar de Sontecomapan, Catemaco, Veracruz, México. Revista Científica Biológico Agropecuario, Tuxpan, 2, 816–821.
Carmona-Higuita, M. J., Mendieta-Leiva, G., Gómez-Díaz, J. A., Villalobos, F., Ramos, F. N., Elias, J. P. C. et al. (2025). Endemism centres of the five richest vascular epiphyte families in the Neotropics. Journal of Biogeography, 52, 80–91. https://doi.org/10.1111/jbi.15016
Ceja-Romero, J., Espejo-Serna, A., López-Ferrari, A. R., García-Cruz, J., Mendoza-Ruiz, A., & Pérez-García, B. (2008). Las plantas epífitas, su diversidad e importancia. Ciencias, 1, 34–41.
Chilpa-Galván, N., Márquez-Guzmán, J., Zotz, G., Echevarría-Machado, I., Andrade, J. L., Espadas-Manrique, C. et al. (2018). Seed traits favouring dispersal and establishment of six epiphytic Tillandsia (Bromeliaceae) species. Seed Science Research, 28, 349–359. https://doi.org/10.1017/S0960258518000247
Conabio (Comisión Nacional para el Conocimiento y Uso de la Biodiversidad) (2013). La biodiversidad en Chiapas: Estudio de Estado. México: Conabio/ Gobierno del Estado de Chiapas.
Conabio (Comisión Nacional para el Conocimiento y Uso de la Biodiversidad) (2019). La biodiversidad en Tabasco. Estudio de Estado. México: Conabio. https://bioteca.biodiversidad.gob.mx/janium/Documentos/14868.pdf
Conap (Consejo Nacional de Áreas Protegidas) (2010). Guía de reconocimiento del género Tillandsiade Guatemala. Guatemala. https://www.caftadr-environment.org/wp-cont
ent/uploads/2016/04/guia-de-Reconocimiento-de-Tillandsia-2.pdf
Conanp (Comisión Nacional de Áreas Naturales Protegidas) (2015). Área de protección de flora y fauna Cañon del Usumacinta. México: Semarnat/ Conanp. https://www.
conanp.gob.mx/que_hacemos/pdf/programas_manejo/2015/Canon_del_Usumacinta.pdf
Conanp (Comisión Nacional de Áreas Naturales Protegidas) (2023a). Estudio previo justificativo para el establecimiento del Área Natural Protegida Reserva de la Biósfera Wanha’.Tabasco, México. https://www.conanp.gob.mx/pdf/separata/EPJ-RB-Wanha.pdf
Conanp (Comisión Nacional de Áreas Naturales Protegidas) (2023b). Estudio previo justificativo para la modificación de la declaratoria del Área Natural Protegida Reserva de la Biosfera Calakmul, Campeche, México. https://www.conanp.gob.mx/pdf/separata/EPJ-Modificacion-RB-Calakmul.pdf
Davidse, G., Sousa-Sánchez, M., & Chater, A. O. (1994). Alismataceae a Cyperaceae. In G. Davidse (Ed.), Flora mesoamericana (pp. 1–543). México D.F.: Universidad Nacional Autónoma de México.
De Sousa, M. M., & Colpo, K. D. (2017). Diversity and distribution of epiphytic bromeliads in a Brazilian subtropical mangrove. Anais da Academia Brasileira de Ciências, 89, 1085–1093. https://doi.org/10.1590/0001-3765201720160702
Díaz-Jiménez, P. (2007). Flora epífita de los manglares de Tabasco, México. Kuxulkab, 13, 11–17.
Du, J., & Hesp, P. A. (2020). Salt spray distribution and its impact on vegetation zonation on coastal dunes: a review. Estuaries and Coasts, 43, 1885–1907. https://doi.org/10.1007/s12237-020-00820-2
Duno-de Stefano, R., Can-Itza, L. L., Rivera-Ruiz, A., & Calvo-Irabién, L. M. (2012). Regionalización y relaciones biogeográficas de la península de Yucatán con base en los patrones de distribución de la familia Leguminosae. Revista Mexicana de Biodiversidad, 83, 1053–1072. https://doi.org/10.7550/rmb.27824
Einzmann, H. J., & Zotz, G. (2017). Dispersal and establishment of vascular epiphytes in human-modified landscapes. AoB Plants, 9. https://doi.org/10.1093/aobpla/plx052
Espejo-Serna, A., López-Ferrari, A. R., Ramírez-Morillo, I., Holst, B., Luther, H., & Till, W. (2004). Checklist of Mexican Bromeliaceae with notes on species distribution and levels of endemism. Selbyana, 25, 33–86. https://doi.org/10.2307/41760147
Feller, I. C., Lovelock, C. E., Berger, U., McKee, K. L., Joye, S. B., & Ball, M. C. (2010). Biocomplexity in Mangrove Ecosystems. Annual Review of Marine Science, 2, 395–417. https://doi.org/10.1146/annurev.marine.010908.163809
Flores-Palacios, A., & García-Franco, J. G. (2006). The relationship between tree size and epiphyte richness: testing four different hypotheses. Journal of Biogeography, 33, 323–330. https://doi.org/10.1111/j.1365-2699.2005.01382.x
García-Gil, G., Palacio-Prieto, J. L., & Ortiz-Pérez, M. A. (2002). Reconocimiento geomorfológico e hidrográfico de la Reserva de la Biosfera Calakmul, México. Investigaciones Geográficas, 48, 7–23. https://doi.org/10.14350/rig.59170
García-Luna, C., Carrillo-Bastos, A., Cutz-Pool, L. Q., & González-Salvatierra, C. (2024). Composición de las epífitas vasculares asociadas al manglar ribereño en la comunidad de Ramonal, Quintana Roo, México. Madera y Bosques, 30, e3042616. https://doi.org/10.21829/myb.2024.3042616
Gutiérrez-Baéz, C., & Zamora-Crescencio, P. (2022). Listado florístico de la zona arqueológica de Calakmul, Campeche, México. Foresta Veracruzana, 24, 1–6.
Gómez, M. A., & Winkler, S. (1991). Bromelias en manglares del Pacífico de Guatemala. Revista de Biología Tropical, 39, 207–214.
Granados-Sánchez, D., López-Ríos, G. F., Hernández-García, M. Á., & Sánchez-González, A. (2003). Ecología de las plantas epífitas. Revista Chapingo. Serie Ciencias Forestales y del Ambiente, 9, 101–111.
Gutiérrez-Báez, C. (2004). Listado florístico del norte de Chiapas: Catazajá y límites con Palenque. Polibotánica, 17, 107–124.
Hágsater, E., Soto-Arenas, G. A., Salazar, G. A., Jiménez-Machorro, R., López-Rosas, M. A., & Dressler, R. L. (2005). Orchids of Mexico. México: Instituto Chinoin.
Hammer, O., Harper, D. A. T., & Ryan, P. D. (2001). PAST: Paleontological statistics software package for education and data analysis. Paleontología Electrónica, 4, 9.
Hellmuth, N., & D’Angelo-Jerez, S. (2022). Epífitas en una de las ceibas gigantes más populares de Tikal Plantas que crecen en las ramas y extremidades de los árboles. Guatemala: Flaar Mesoamérica. https://flaar-mesoamerica.org/wp-content/uploads/2023/01/PANAT-epiphytes-giant-Ceiba-Jul-20-2022-SJ-Sep-2022-espanol-PM-Nov-2022.pdf
Hernández-Sastré, J., Jiménez-Pérez, N. C., & Guadarrama-Olivera, M. A. (2014). Diversidad y estructura vertical de la comunidad de epífitas vasculares del Ejido Niños Héroes de Chapultepec, Tenosique, Tabasco (Bachelor´s Thesis). México. Universidad Juárez Autónoma de Tabasco.
Hietz, P., & Hietz-Seifert, U. (1994). Epífitas de Veracruz: guía ilustrada para las regiones de Xalapa y Los Tuxtlas. México: Instituto de Ecología A. C.
Hietz, P., & Hietz-Seifert, U. (1995). Composition and ecology of vascular epiphyte communities along an altitudinal gradient in central Veracruz, Mexico. Journal of Vegetation Science, 6, 487–498. https://doi.org/10.2307/3236347
Ibarra-Manríquez, G., Villaseñor, J. L., Durán, R., & Meave, J. (2002). Biogeographical analysis of the tree flora of the Yucatán Peninsula. Journal of Biogeography, 29, 17–29. https://doi.org/10.1046/j.1365-2699.2002.00648.x
IUCN (International Union for Conservation of Nature) (2021). The IUCN Red List of Threatened Species. Version 2021-3. Available at: https://www.iucnredlist.org.
Jiménez-López D. A., Roblero-Velasco R. de J., Martínez-Meléndez N., Ocampo G., & Gallardo-Cruz J.A. (2017). Relación entre variables del forófito y la riqueza de epífitas vasculares en los Pantanos de Centla, Tabasco, México. Acta Botanica Mexicana, 121, 125–137. https://doi.org/10.21829/abm121.2017.1179
Jiménez-López, D. A., Peralta-Carreta, C., Solórzano, J. V., Cervantes-Jiménez, G. L., & Domínguez-Vázquez, M. A. (2018). A checklist of vascular epiphytes of El Cometa Lagoon, Pantanos de Centla Biosphere Reserve, México. Journal of Threatened Taxa, 10, 12589–12597. https://doi.org/10.11609/jot.3794.10.11.12589-12597
Kelly, D. L., O’Donovan, G., Feehan, J., Murphy, S., Drangeid, S. O., & Marcano-Berti, L. (2004). The epiphyte communities of a montane rain forest in the Andes of Venezuela: patterns in the distribution of the flora. Journal of Tropical Ecology, 20, 643–666. https://doi.org/10.1017/S0266467404001671
Krömer, T., García-Franco, J. G., & Toledo-Aceves, T. (2014). Epífitas vasculares como bioindicadores de la calidad forestal: impacto antrópico sobre su diversidad y composición. In: C. A González Zuarth, A. Vallarino, JC Pérez Jiménez & AM Low (Eds.), Bioindicadores: guardianes de nuestro futuro ambiental (pp. 605–624). México: Ecosur/ Inecc.
Krömer, T., Gradstein, S. R., & Acebey, A. (2007). Diversidad y ecología de epífitas vasculares en bosques montanos primarios y secundarios de Bolivia. Ecología en Bolivia, 42, 23–33.
Krömer, T., & Gradstein, S. R. (2016). Vascular epiphytes. In T. H. Larsen (Ed.), Core standardized methods for rapid biological field assessment (pp. 25–36). USA: Conservation International.
Kupec, A. (2018). The comparison of vascular epiphytes diversity related to their occurrence in natural and artificial mangrove channels, Greenfields, eastern coast of Nicaragua. In S. Sharma (Ed.), Mangrove Ecosystem Ecology and Function (pp. 41–52). Malaysia: IntechOpen. https://doi.org/10.5772/intechopen.79133
Leal, M., & Spalding, M. D. (2021). The state of the World’s Mangroves 2021. Global Mangrove Alliance. https://www.mangrovealliance.org/wp-content/uploads/2022/09/The-State-of-the-Worlds-Mangroves-Report_2022.pdf
López-Gómez, D. (2014). Efecto de borde sobre la biodiversidad de orquídeas epifitas en el Parque Estatal de Agua Blanca, Macuspana, Tabasco, México (Bachelor’s Thesis). Universidad Juárez Autónoma de Tabasco. Tabasco, México.
Lot, A., & Chiang, F. (1986). Manual de Herbario, administración y manejo de colecciones, técnicas de recolección y preparación de ejemplares botánicos. México D.F.: Consejo Nacional de la Flora de México.
Lugo, A. E. (1981). The inland mangroves of Inagua. Journal of Natural History, 15, 845–852. https://doi.org/10.1080/00222938100770611
Martínez, E., Sousa, M., & Ramos-Álvarez, C. H. (2001). Listados florísticos de México XXII. Región de Calakmul, Campeche. México D.F.: Instituto de Biología, Universidad Nacional Autónoma de México.
Martínez-Meléndez, N., Martínez-Camilo, R., Pérez-Farrera, M. A., & Martínez-Meléndez, J. (2011). Las epífitas de la Reserva El Triunfo, Chiapas. Guía ilustrada de las especies más notables. Tuxtla Gutiérrez, Chiapas: UNICACH.
Martínez-Salas, E., & Ramos, C. H. (2014). Biodiversidad de Pteridophyta en México. Revista Mexicana de Biodiversidad, 85 (Suppl.), S110-S113. https://doi.org/10.7550/rmb.31827
Melgar, W. (2003). Estado de la diversidad biológica de los árboles y bosques de Guatemala. Documentos de Trabajo: Recursos Genéticos Forestales. Roma, Italia: Dirección de Recursos Forestales, FAO.
Menchaca-García, R. A., & Morano-Martínez, D. (2011). Manual para la propagación de orquídeas. Jalisco, México: Comisión Nacional Forestal/ Semarnat. http://www.conafor.gob.mx:8080/biblioteca/ver.aspx?articulo=462
Menéndez-Liguori, F. (1976). Los manglares de la laguna de Sontecomapan los Tuxltas, Veracruz: estudio florístico-ecológico (Bachelor’s Thesis). Universidad Nacional Autónoma de México. México.
Mickel J. T., & Smith A. R. (2004). The Pteridophytes of Mexico. New York: Botanical Garden Press.
Mikolaev, Y. O., Bernal, E. S., Baños, J. B., Escobar, M. O., & Hernández, J. G. (2016). Saline year-gradients in the lagoon “La Salina,” Tonameca, Oaxaca, Mexico: Effects on growth of mangrove biotope. Water, Air, & Soil Pollution, 227, 426. https://doi.org/10.1007/s11270-016-3133-3
Mondragón-Chaparro, D. M., Ramírez, I. M., Flores, M. M., & García, J. G. (2011). La Familia Bromeliaceae en México. Texcoco, Estado de México: Universidad Autónoma de Chapingo.
Morales-Damián, A. (2012). Inventario de orquídeas del ejido Niños Héroes de Chapultepec, Tenosique, Tabasco (Bachelor’s Thesis). Villahermosa: Universidad Juárez Autónoma de Tabasco.
Morrone, J. J. (2019). Regionalización biogeográfica y evolución biótica de México: encrucijada de la biodiversidad del Nuevo Mundo. Revista Mexicana de Biodiversidad, 90, e902980. https://doi.org/10.22201/ib.20078706e.2019.90.2980
Noguera-Savelli, E., Pacheco-Pizano, J., & Cetzal-Ix, W. (2021). Epífitas del fragmento de manglar de la UMA Nohan, Carmen, Campeche, México. Desde el Herbario CICY 13, 136–142.
Noguera-Savelli, E., & Cetzal-Ix, W. (2014). Revisión e integración del conocimiento de las Orchidaceae de Tabasco, México. Botanical Sciences, 92, 519–540. https://doi.org/10.17129/botsci.117
Palma-López, D. J., Jiménez Ramírez, R., Zavala-Cruz, J., Bautista-Zúñiga, F., Gavi-Reyes, F., & Palma-Cancino, D. Y. (2017). Actualización de la clasificación de suelos de Tabasco, México. Agroproductividad, 10, 29–35.
Patel, P. (2014). Inland mangroves of the world. Current Science, 107, 1094.
Patel, P., & Agoramoorthy, G. (2012). India’s rare inland mangroves deserve protection. Environmental Science & Technology, 46, 4261–4262. https://doi.org/10.1021/es301062v
Perrie, L. R., Ohlsen, D. J., Shepherd, L. D., Garrett, M., Brownsey, P. J., & Bayly, M. J. (2010). Tasmanian and Victorian populations of the fern Asplenium hookerianum result from independent dispersals from New Zealand. Australian Systematic Botany, 23, 387–392. https://doi.org/10.1071/SB10028
Rahman, M. S., Hossain, G. M., Khan, S. A., & Uddin, S. N. (2015). An annotated checklist of the vascular plants of Sundarban Mangrove Forest of Bangladesh. Bangladesh Journal Plant Taxon, 22, 17–41. https://doi.org/10.3329/bjpt.v22i1.23862
Rodríguez-Romero, M. L., Zavala-Hurtado, J. A., & Pacheco, L. (2011). Presencia, abundancia y estrategias reproductivas de helechos en áreas alteradas de la Sierra Nevada, México. Revista de Biología Tropical, 59, 417–433.
Rohani, S., Lee, F. L., & Yusoff, A. S. (2020). Diversity and vertical distribution of vascular epiphytes on a Malaysian mangrove island. Biotropia, 27, 134–142. https://doi.org/10.11598/btb.2020.27.2.1182
Rzedowski, J. (1996). Análisis preliminar de la flora vascular de los bosques mesófilos de montaña de México. Acta Botanica Mexicana, 35, 25–44. https://doi.org/10.21829/abm35.1996.955
Schurr, F. M., Spiegel, O., Steinitz, O., Trakhtenbrot, A., Tsoar, A., & Nathan, R. (2009). Long-distance seed dispersal. Annual Plant Reviews, 38, 204–237. https://doi.org/10.1002/9781444314557.ch6
Semarnat (Secretaría del Medio Ambiente y Recursos Naturales) (2010). Norma Oficial Mexicana NOM-059-Semarnat-2010, Protección ambiental – Especies nativas de México de flora y fauna silvestres – Categorías de riesgo y especificaciones para su inclusión, exclusión o cambio – Lista de especies en riesgo. Diario Oficial de la Federación. 2da Sección, 30 de diciembre de 2010.
Semarnat (Secretaría del Medio Ambiente y Recursos Naturales) (2023). Edicto: Reserva de la Biosfera la región denominada Wanha’, ubicada en los municipios de Balancán y Tenosique, en el estado de Tabasco, México. Diario Oficial de la Federación. 9 de agosto de 2023. https://dof.gob.mx/nota_detalle.php?codigo=5698495&fecha=15/08/2023#gsc.tab=0
Shepherd, L. D., De Lange, P. J., & Perrie, L. R. (2009). Multiple colonizations of a remote oceanic archipelago by one species: how common is long-distance dispersal? Journal of Biogeography, 36, 1972–1977. https://doi.org/10.1111/j.1365-2699.2009.02120.x
Sousa, M. M. D., & Colpo, K. D. (2017). Diversity and distri-
bution of epiphytic bromeliads in a Brazilian subtropical mangrove. Anais da Academia Brasileira de Ciências, 89, 1085–1093. https://doi.org/10.1590/0001-3765201720160702
Stoddart, D. R., Bryan, G. W., & Gibbs, P. E. (1973). Inland mangroves and water chemistry, Barbuda, West Indies. Journal of Natural History, 7, 33–46. https://doi.org/10.1080/00222937300770031
Taylor, F. J. (1986). Mangroves in freshwater. Blumea: Biodiversity, Evolution and Biogeography of Plants, 31, 271–272.
Thiers. B. (2021). Index Herbariorum: a global directory of public herbaria and associated staff. New York Botanical Garden’s Virtual Herbarium. Retrieved March 2024, from http://sweetgum.nybg.org/ih
Toledo-Aceves, T., García-Franco, J. G., Hernández-Rojas, A., & MacMillan, K. (2012). Recolonization of vascular epiphytes in a shaded coffee agroecosystem. Applied Vegetation Science, 15, 99–107. https://doi.org/10.1111/j.1654-109X.2011.01140.x
Tripathi, N., Singh, R. S., Bakhori, B., Dalal, C., Parmar, D. & Mishra, B. (2013). The World’s only inland mangrove in sacred grove of Kachchh, India, is at risk. Current Science, 105, 1053–1055.
Tropicos.org. (2023) Available at: https://tropicos.org
Woodroffe, C. D. (1988). Relict mangrove stand on last interglacial terrace, Christmas Island, Indian Ocean. Journal of Tropical Ecology, 4, 1–17. https://doi.org/10.1017/S0266467400002431
Zimmerman, J. K., & Olmsted, I. C. (1992). Host tree utilization by vascular epiphytes in a seasonally inundated forest (Tintal) in Mexico. Biotropica, 24, 402–407. https://doi.org/10.2307/2388610
Zotz, G. (2016). Physiological ecology. In G. Zotz (Ed.), Plants on plants: the biology of vascular epiphytes (pp. 95–137). Switzerland: Springer Cham. https://doi.org/10.1007/978-3-319-39237-0
Zotz, G., & Reuter, N. (2009). The effect of exposure to sea water on germination and vegetative growth of an
epiphytic bromeliad. Journal of Tropical Ecology, 25, 311–319. https://doi.org/10.1017/S0266467409006014
Zotz, G., Weigelt, P., Kessler, M., Kreft, H., & Taylor, A. (2021). EpiList 1.0: a global checklist of vascular
epiphytes. Ecology, 102, e03326. https://doi.org/10.1002/ecy.3326