Primer registro de Axianassa darrylfelderi (Crustacea: Decapoda: Axianassidae) en México y descripción de su primer estadio de zoea
Ignacio Cáceres a, b, José Salgado-Barragán c, Brenda Díaz-Cárdenas b, Eduardo Ríos-Jara a, Manuel Ayón-Parente a, *
a Universidad de Guadalajara, Centro Universitario de Ciencias Biológicas y Agropecuarias, Departamento de Ecología, Laboratorio de Ecología Molecular, Microbiología y Taxonomía, Camino Ramón Padilla Sánchez No. 2100, 45110 Nextipac, Zapopan, Jalisco, Mexico
b Universidad de Guadalajara, Centro Universitario de Ciencias Biológicas y Agropecuarias, Departamento de Biología Celular y Molecular, Laboratorio de Marcadores Moleculares en Biomedicina y Ecología, Camino Ramón Padilla Sánchez No. 2100, 45110 Nextipac, Zapopan, Jalisco, Mexico
c Universidad Nacional Autónoma de México, Unidad Académica Mazatlán, Instituto de Ciencias del Mar y Limnología, Laboratorio de Invertebrados Bentónicos, PO Box 811, 82000 Mazatlán, Sinaloa, Mexico
*Corresponding author: manuel.ayon@academicos.udg.mx (M. Ayón-Parente)
Received: 21 May 2024; accepted: 11 September 2025
Abstract
Axianassa darrylfelderi, previously known only from the Pacific coast of Colombia and Panama, is reported for the first time in Mexico and its first larval stage is described. Morphological characters of adults and the first zoea, in addition to a phylogenetic analysis were used to review the taxonomic status of the species. Our results show that A. darrylfelderi and A. australis are indeed related, but both species can be considered as different.
Keywords: Mud shrimps; Decapod; New records; Central Mexican Pacific
Resumen
Axianassa darrylfelderi, anteriormente conocida solo de la costa pacífica de Colombia y Panamá, se reporta por primera vez para México y también se describe su primer estadio larvario. Se utilizaron caracteres morfológicos de adultos y de la primera zoea, además de análisis filogenéticos, para revisar el estatus taxonómico de ésta. Los resultados muestran que A. darrylfelderi y A. australis efectivamente están relacionadas, pero ambas especies pueden considerarse diferentes.
Palabras clave: Camarones de lodo; Decápodo; Nuevo registro; Pacífico central mexicano
Introduction
As part of a research project on the inventory of decapod crustaceans from the bay of Chamela, Jalisco, Mexico, in the Mexican Pacific coast, 65 specimens of the mud shrimp Axianassa darrylfelderi Anker & Lazarus, 2015 were collected in mud bottoms of the Pérula Estuary. According to the World Register of Marine Species (WoRMS) consulted in February 2024, the genus Axianassa is composed of 15 species distributed in tropical and subtropical waters of the western Atlantic (A. intermedia Schmitt, 1924, A. arenaria Kensley & Heard, 1990, A. jamaicensis Kensley & Heard, 1990, A. australis Rodrigues & Shimizu, 1992, A. ferrazeae Blanco-Rambla & Kensley, 1998, and A. ovis Blanco-Rambla & Kensley, 1998), the eastern Pacific (A. mineri Boone, 1931, A. canalis Kensley & Heard, 1990, A. darrylfelderi Anker & Lazarus, 2015, A. christi Anker & Pachele, 2016, and A. linda Anker & Pachele, 2016), and the Indo-Pacific (A. ngochoae Anker, 2010, A. sinica Liu & Liu, 2010, A. planioculus Komai & Fujita, 2019, and A. microlepis Komai, Watanabe, Matsui & Tamego, 2020). Of the species recorded in the eastern Pacific, only A. mineri has been recorded in Mexico, Hernández-Aguilera (1998) reported 2 specimens from the intertidal of Isla Isabel and the Islas Marías.
Axianassa darrylfelderi was described from material collected in Bahía Málaga, Pacific coast of Colombia. The authors mentioned that it is very similar to A. australis from the coasts of Brazil and indicated that both species could be differentiated by the presence of several teeth on the lateral margin of the uropodal exopodite of A. darrylfelderi and absent in A. australis, the relative length of the antennal acicle, in relation to the 4th antennal segment is longer in A. darrylfelderi than in A. australis, and the armature of the cutting edge of the polex of the minor cheliped is armed with 3 strong (bicuspid) and numerous small teeth in A. darrylfelderi versus 1 proximal bi or tricuspid tooth and 2 simple distal teeth in A. australis. On the other hand, Anker and Pachelle (2016) reported A. darrylfelderi on the Pacific coast of Panama and observed that the species have more morphological variations. Due to the above, this species can be differentiated from A. australis solely by differences in the proportion of the relative length of the antennal acicle in relation to the 4th antennal peduncle (0.35 in A. darrylfelderi vs. 0.25 in A. australis) and other minor differences, e.g., 3rd to 5th abdominal pleurae with moderate pubescence in A. darrylfelderi vs. strongly pubescent in A. australis. Therefore, the authors considered A. darrylfelderi as a valid species, although with reservations (see the species identification key in Anker and Pachelle [2016]: 122) and suggested that a molecular analysis would be useful to confirm A. darrylfelderi as a distinct species.
It is known that the larval development of invertebrates provides important information about the biology of the species and their phylogenetic relationships; however, information on the larval stages of many invertebrates, including decapod crustaceans, is still scarce. Within the genus Axianassa, the larval development of A. australis is the only one known. Rodrigues and Shimizu (1992) described A. australis as a new species and included the description of its first zoeal stage (ZI). On the other hand, Ngoc-Ho (1981) included the complete larval development of Axianassa sp. (presumably A. australis) and Strasser and Felder (2005) presented the complete larval development of this same species, including a comparative analysis of its ZI, and those described in previous works.
Molecular analyses have become a useful tool to solve problems about the status of species (Mantelatto et al., 2006, 2009; Matzen da Silva et al., 2011) or to determine phylogenetic relationships among species or groups of species (Felder & Robles, 2009; Henmi et al., 2022; Morrison et al., 2002; Robles et al., 2009, 2020). Given the close morphological resemblance between A. darrylfelderi and A. australis and the uncertainty of the validity of A. darrylfelderi as a distinct species, the importance of combining morphological traits with molecular evidence to elucidate this problem is evident. The objective of this work is to present a new distribution record for A. darrylfelderi in the Pacific coast of Mexico, as well as to contribute information on the validity of the species through a molecular analysis and the description of its ZI.
Materials and methods
The Pérula Estuary is in the northwest portion of the town of Pérula, in Chamela Bay, Jalisco, Mexico (Fig. 1). The estuary is 2.5 km long with a width ranging between 16 and 65 m and an average depth of 0.8 m. The surrounding vegetation is classified as low deciduous forest type and the estuary is partially surrounded by mangroves (Avicennia germinans, Conocarpus erectus, Laguncularia racemosa, and Rhizophora mangle), those species are within the category of non-endemic threatened species in the Mexican Official Norm (NOM-059-SEMARNAT-2010).
The organisms were collected using a “yabby” suction pump in muddy substrates, at depths of 0-0.5 m. Once collected, specimens of A. darrylfelderi were kept alive for several hours for observation. One of the ovigerous females collected in April 2018 was separated and kept until it released the zoea larvae. Unfortunately, the larvae only survived for a few hours and were collected and fixed in 70% ethanol. Several larvae were selected for dissection and description of the larval stage. Carapace length (CL) was measured along the dorsal midline from the tip of the rostral projection to the posterior margin of the carapace and total length (TL) was measured from the tip of the rostrum to the posterior midpoint of the telson. All measurements are in millimeters. In the description of larval appendages, the arrangement of the setae was listed sequentially from the proximal to the distal ones, as suggested by Konishi (1989), Nates et al. (1997), and Strasser and Felder (2005). In the listings of the groups of setae, those on the different segments were separated by a comma and those on the same segment or on different lobes of the same segment, article or endite, were separated with a plus sign. Roman numerals were used to describe the pattern of processes on the posterior margin of the telson. The dissection of the larvae was performed with the aid of a Leica M125 stereoscopic microscope. Ten larvae were dissected for their description and both appendages (right and left) were used for counting the setae. The illustrations were made using a Leica DM1000 microscope equipped with a camera lucida. Adults, juveniles, and larvae of Axianassa were preserved in 70% ethanol and deposited, with an accession number, in the Crustacean Collection of the Laboratory of Marine Ecosystems and Aquaculture (LEMA-CR) of the University Center for Biological and Agricultural Sciences (CUCBA) in the University of Guadalajara (U de G).
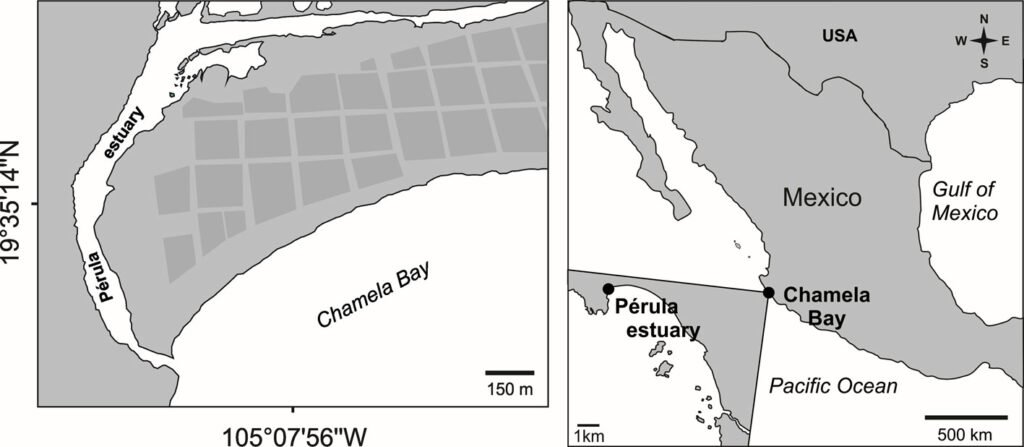
Figure 1. Geographical location of the study area.
Phylogenetic analysis, genetic distances and haplotype networks
Total genomic DNA of an individual of A. darrylfelderi was extracted from muscle tissue of the walking legs. Partial DNA sequences were obtained from 2 genes, 16S and COI. DNA was extracted using the Quick-DNA TM Tissue/Insect Microprep kit, Catalog No. D6015 of ZYMO RESEARCH. The 16S region was amplified with the primer “CCT GTT TAN CAA AA CAT and AGA AAA CCA ACC TGG” (Crandal & Fitzpatrick Jr., 1996), and the following thermal cycler conditions: 5 min at 95 °C; annealing for 35 cycles: 1 min at 95 °C, 1 min at 54 °C, 1 min at 72 °C; final extension 10 min at 72 °C. For the COI region we used the primer “TAA ACT TCA GGG TGA CCA AAA AAT CA and GGT CCA CAA ATC ATA AAG ATA TTG G” (Folmer et al., 1994), and the following thermal cycler program: 5 min at 95 °C; annealing for 30 cycles: 1 min at 95 °C, 1 min at 40 °C, 1 min at 72 °C; final extension 10 min at 72 °C. The PCR took place in a final volume of 25 μL consisting of 12.1 μL of H2O, 2 μL of reaction buffer, 0.2 μL of dNTPs, 0.08 μL of each primer, 1.5 μL of MgCl2 0.1 μL of Taq Platinum (Invitrogen), and 20 ng of DNA. Amplification of the regions was confirmed by agarose gel electrophoresis at 1%. The amplified products were sent to the National Laboratory for Plant Identification and Characterization (LANIVEG) at CUCBA, UdeG, for sequencing. The generated sequences were deposited in GenBank under the accession numbers PV52069 for the COI region and PV521869 for 16S region.
The COI and 16S sequences obtained were edited with Sequencher v 4.1.4 (Gene Codes Corp., Ann Arbor, Michigan). The BLAST tool from Genbank (https://blast.ncbi.nlm.nih.gov/Blast.cgi) was used to determine the percentage of similarity between A. darrylfelderi and A. australis. To test the divergence between the collected individual and individuals of A. australis, we analyzed the genetic distances under the Tamura-Nei 93 (TN93) model in the ape package (Paradis & Schliep, 2019) in R (R Core Team, 2021). To perform a phylogenetic analysis, we obtained the following sequences of A. australis from GenBank: MF490135, OQ108484 and OQ108485 for the COI region and MF490232, EU874948, OQ110581 and OQ110582 for the 16S region. Following Teles & Mantelatto (2023) we used Upogebia brasiliensis (COI: MF490136, 16S: MF490236), U. noronhensis (COI: MF490138, 16S: MF490234) and U. paraffinis (COI: MF490137, 16S: MF490235) to root the tree. Regarding the 16S tree, we also add A. planioculus (LC489001) as outgroup. The sequences were visually aligned in the PhyDe v 0.9971 program (Müller et al., 2010).
A phylogenetic analysis using maximum likelihood was performed in RAxML GUI 2.0 (Edler et al., 2020) for each region with the HKY + I + G evolutionary model for the COI region and the HKY + G model for the 16S region, both models were inferred in RAxML based on the Bayesian information criterion. To assess nodal support a resampling of 10,000 rapid bootstrap inferences was done. To corroborate the distance between A. australis and A. darrylfelderi sequences,we generated a haplotype network in the R package ape (Paradis & Schliep, 2019; R Core Team, 2021).
Results
Family Laomediidae Borradaile, 1903
Genus Axianassa Schmitt, 1924
Axianassa darrylfelderi Anker and Lazarus, 2015
(Fig. 2A-C)
Pérula Estuary: 6 males (CL 4.5-5.9 mm), 3 females (CL 4.5-8.0 mm), 2 ovigerous females (CL 6.3-7.5 mm), Jan 15, 2015 (LEMA-CR 697). Three females (CL 5.4- 5.7 mm), Jul 30, 2015 (LEMA-CR 698). Four juveniles (CL 2.6-3.3 mm), Oct 18, 2015 (LEMA-CR 699). Fifteen juveniles (CL 3.6-6.2 mm), Nov 15, 2015 (LEMA-CR 700). Seven males (CL 10.4-10.6 mm), 5 females (CL 9.7-10.4 mm), 9 ovigerous females (CL 10.4-10.6 mm), Apr 28, 2018 (LEMA-CR 701). Seven males (CL 6.2-12.0 mm), 4 ovigerous females (CL 10.7-11.5 mm), Sep 4, 2018 (LEMACR-CR 702).
The discovery of A. darrylfelderi on the coast of Jalisco, represents the third record of the species in the Eastern Tropical Pacific. The 2 previous records correspond to the type locality, Bahía Málaga, Colombia, and Playa El Agallito, Panama (Anker & Lazarus, 2015; Anker & Pachele, 2016). The specimens collected in the Pérula Estuary represent the first record of the species for Mexico and thus extends its geographical distribution about 3,000 km to the north.
According to Anker and Pachele (2016), specimens of A. darrylfelderi from Colombia and Panama have morphological variations between individuals from the same populations and between males and females. We were also able to observe morphological variations among the specimens from the Pérula Estuary. The most evident variations were: the number of teeth on the ischium of the third maxillipeds varied from 13 to 18, with an average of 14; the number of teeth on the ischium of the largest cheliped varied from 1 to 6 in males and from 3 to 5 in females, while the number of teeth on the ischium of the minor cheliped ranged from 3 to 5 in males and from 3 to 4 in females. Uropods in all the specimens always carried a spiniform seta and from 0 to 5 fixed teeth on the external margin. Most of the individuals presented a rounded and unarmed rostrum (Fig. 2A, C), and only 2 specimens had a rostrum ending in a spine (Fig. 2B). The majority of the specimens had both antennal acicles armed with a mesial tooth (Fig. 2A) but 3 specimens had just 1 armed acicle (Fig. 2B) and 1 specimen had unarmed antennal acicles (Fig. 2C). The dorsomesial surface of the coxa of the third maxilliped bears only 1 large tooth in most of the specimens and only 2 specimens had 2 strong adjacent teeth.
Description of zoea I
(Fig. 3A-K)
Size. CL = 0.63 ± 0.038 mm, range 0.57-0.67 mm; TL = 1.56 ± 0.075 mm, range 1.48-1.63 mm. Carapace (Fig. 3A) about the same length as abdomen; rostrum elongate, rounded in cross-section, smooth; eyes fused to carapace. Abdomen (Fig. 3A) somite 5 with posterolateral projections about length of somite; lacking dorsal spines. Pleopods not developed.
Telson (Fig. 3A) narrow, anteriorly not differentiated from abdominal segment 6, triangular posterior with medial cleft, anal spine absent; 7 + 7 processes on posterior margin arranged as (I. II, III, II, I) where I is outermost naked seta, II is very thin (anomuran hair), III is 5 plumodenticulate setae.
Antennule (Fig. 3B) elongate, exopodal and endopodal lobes not distinct; 5 terminal aesthetascs and 1 seta; 1 long plumose seta on future endopodal lobe. Antenna (Fig. 3C) protopod with 1 distal spine between rami; endopod with 3 long, plumose setae; scaphocerite (exopod) armed with 1 strong distolateral spine, 10 plumose setae on inner margin. Mandibles (Fig. 3D, E) asymmetrical; right mandible with 2 prominent teeth at base, lower plate with numerous small teeth, distal end pointed but not as strongly produced as on left mandible; left mandible sickle-shaped, distal end pointed with 2 teeth, inner surface of base with several teeth. Left lobe of paragnath (Fig. 3F) sickle-shaped. Maxillule (Fig. 3G) coxal endite with 4 marginal setae; basal endite with 2 setae and 2 large dentate spines; endopodal lobe disctinct from protopod, with 3 setea on distal margin; propotopod without setae. Maxilla (Fig. 3H) coxal endite bilobed, 1 seta on proximal lobe, and 3-4 setae on distal lobe; basal endite bilobed with 4 setae on proximal lobe, 3-5 setae on distal lobe; endopod small, unsegmented with 2 terminal setae; scaphognathite with 5 plumose setae. Maxilliped 1 (Fig. 3I) coxa without setae; basis with 1 + 2 + 1 setae; endopod 4-segmented, with 1, 1, 2, 5 setae; exopod unsegmented, 4 plumose setae on distal margin. Maxilliped 2 (Fig. 3J) coxa and basis without setae; endopod 4-segmented, 1, 1, 2, 5 setae, exopod unsegmented, 4 plumose setae on distal margin. Maxilliped 3 (Fig. 3K) 2-segmented, without setae.
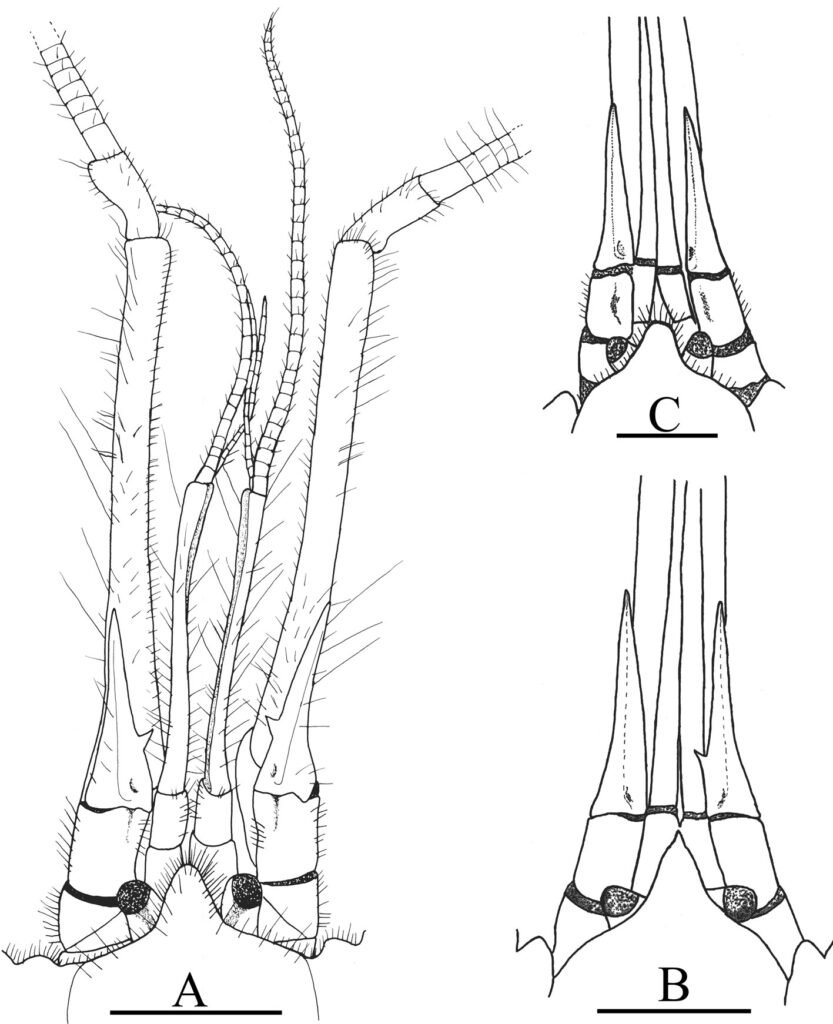
Figure 2. Axianassa darryfelderi from Pérula Estuary, Jalisco. A, Frontal region in dorsal view of a male (CL = 10.2 mm); B, rostrum, eyestalks and antennal acicles in dorsal view of a female (CL = 9.6 mm); C, rostrum, eyestalks and antennal acicles in dorsal view of a male (CL = 11.4 mm). Scale = 2 mm.
Table 1
Percentage identity (similarity) and genetic distances between Axianassa darrylfelderi and A. australis according to COI and 16S mitochondrial regions.
| Percentage identity | Genetic distance | |
| COI | 89 | 0.124 |
| 16S | 97 | 0.047 |
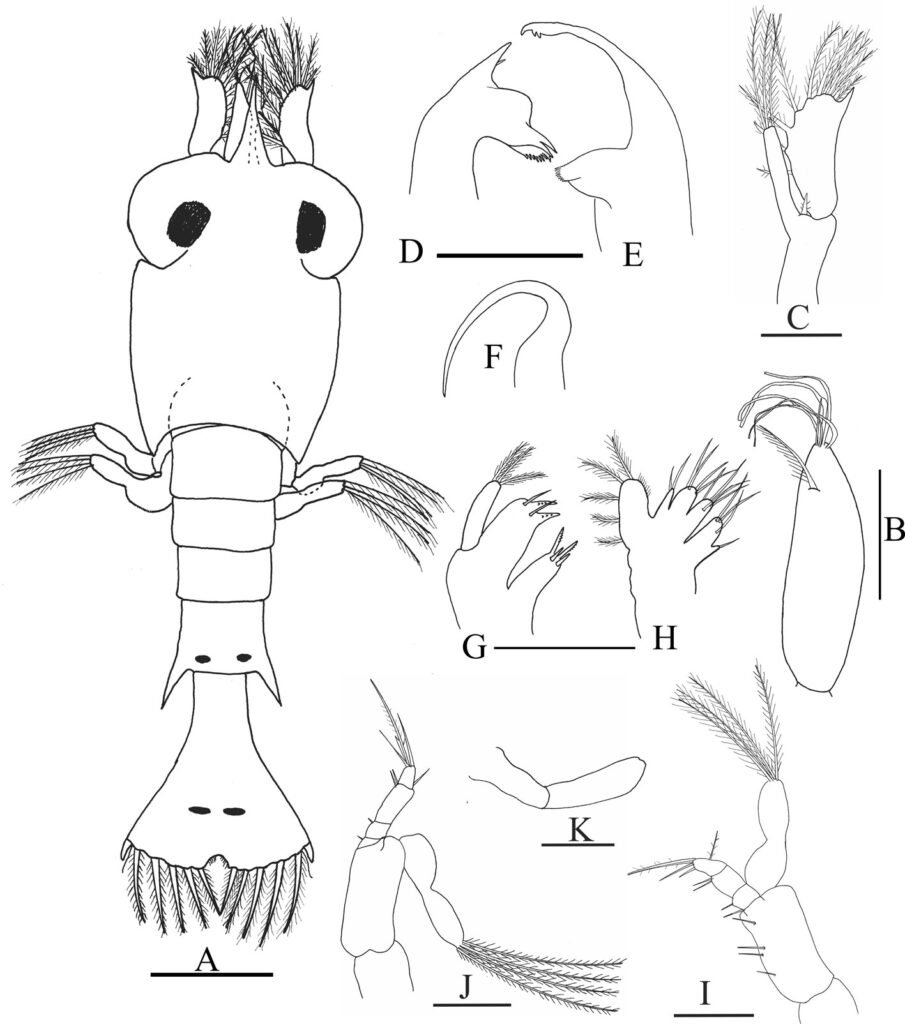
Figure 3. Axianassa darrylfelderi, zoea I. A, Dorsal view; B, antennule; C, antenna; D, right mandible; E, left mandible; F, left lobe of paragnath; G, maxillule; H, maxilla; I, maxilliped I; J, maxilliped 2; K, maxilliped 3. Scale bars = O.1 mm.
Pereopods. Not developed.
Sequences of 644 bp and 492 bp were obtained for COI and 16S, respectively. BLAST analyses showed 89% similarity to the A. australis COI region and 97% similarity to the A. australis 16S region (Table 1). According to the genetic distances, A. darrylfelderi was 12.4% (COI) and 4.7% different from A. australis (Table 1). The ML phylogenetic hypothesis of the COI region, groups A. darrylfelderi and A. australis with low support (49) and the 16S hypothesis indicates that A. darrylfelderi and A. australis are sister groups witha boostrap support of 96 (Fig. 4). The haplotype network showed a distance of 44 mutational steps in the COI network and 12 mutational steps in the 16S network between A. darrylfelderi and A. australis (Fig. 5). The genetic analyses presented in this study are a partial contribution to establishing that, based on the specimens collected in the Pérula Estuary, the genetic sequences of A. darrylfelderi are close, but different to those of A. australis compiled from GenBank, which would allow us to establish that both species can be considered as valid.
Discussion
The use of complementary tools for taxonomic analysis has allowed us to clarify the phylogenetic status of several species of decapod crustaceans. The great morphological similarity between A. darrylfelderi and A. australis led Anker and Pachelle (2016) to question whether the former is a valid species or to consider if it is a synonym of the latter. We believe that the application of molecular analyses (partial 16S rDNA and COI sequences) and a morphological review of adults and the Z1 allowed us to solve this problem, and to consider A. darrylfelderi as a valid species.
Anker and Pachelle (2016) modified the criteria used by Anker and Lazarus (2015) to differentiate specimens of A. darrylfelderi and A. australis (presence or absence of teeth on the distolateral margin of the uropodal exopodite, the proportional length of the antennal acicle and the armature of the cutting edges of the pollex in the male´s minor cheliped) because they observed that the specimens of A. darrylfelderi collected in Panama showed a wide morphological variation that overlapped with the variations observed in A. australis, so they suggested that only the relative length of the antennal acicle could be considered as a valid difference between both species (0.35 of the length of the 4th article of the antennal peduncle in A. darrylfelderi vs. 0.25 of the length of the 4th article of the antennal peduncle in A. australis). Furthermore, they added that abdominal pleurae 3-5 of A. australis are densely pubescent, obscuring their margin, while those of A. darrylfelderi are moderately pubescent and that the dorsomesial surface of the coxa of the third maxilliped of A. darrylfelteri has 2 small teeth (occasionally 1) vs. 1 tooth in A. australis.
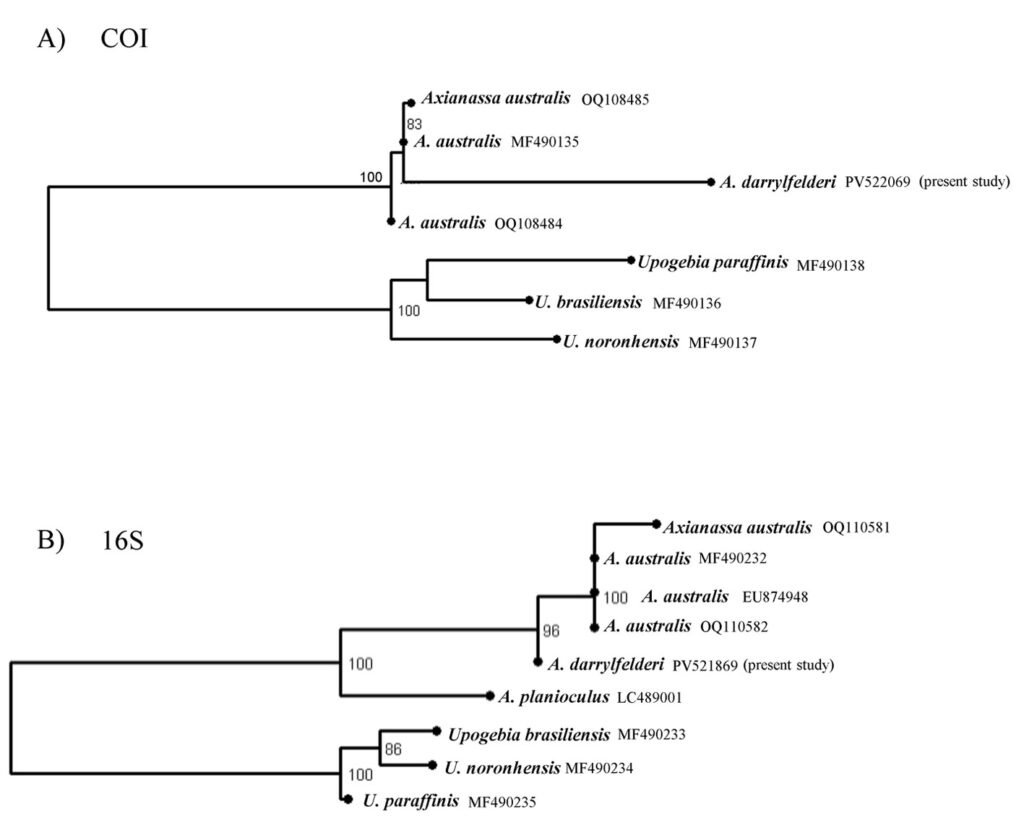
Figure 4. Phylogenetic hypotheses of Axianassa darrylfelderi obtained by the maximum likelihood method with the COI (A) and 16S (B) regions. Node numbers indicate bootstrap support values above 70. GenBank accession numbers are shown in the tips of the tree.
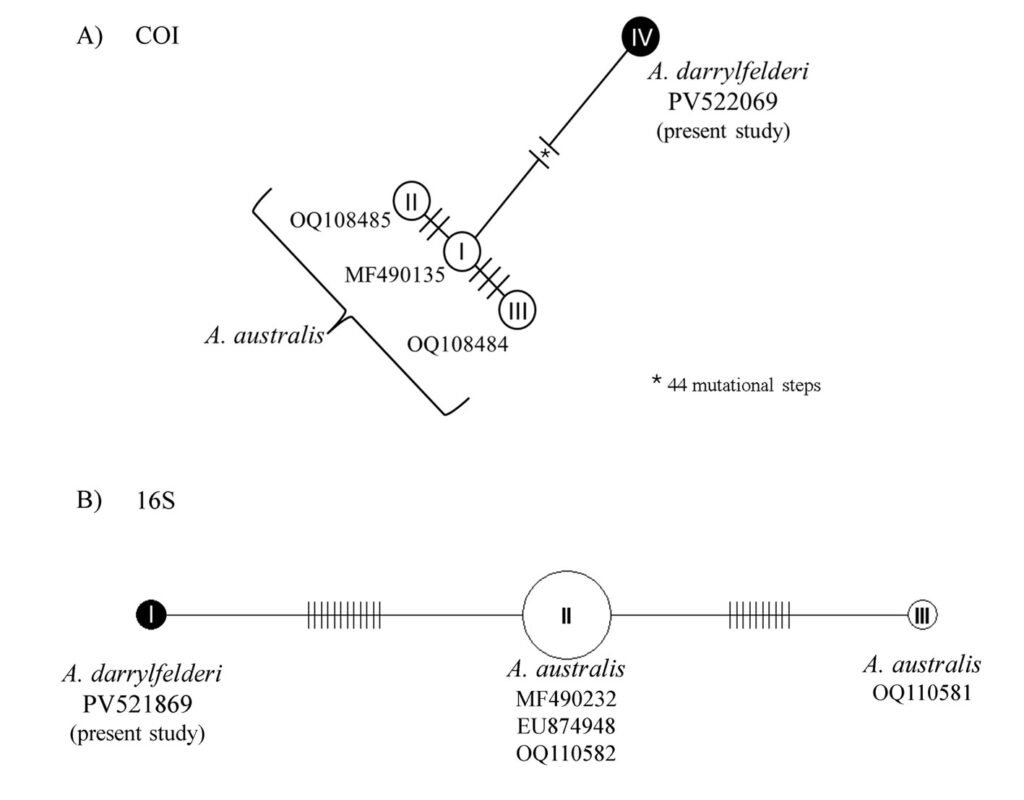
Figure 5. Haplotype networks of Axianassa darrylfelderi and A. australis. The lines between haplotypes represent mutational steps. The numbers indicate GenBank accession numbers.
The specimens collected in Pérula Estuary could be identified as A. darrylfelderi because their antennal acicles are 0.35 of the length of the 4th article of the antennal peduncle and the abdominal pleurae 3-5 are scarcely pubescent. However, unlike what was established by Anker and Pachelle (2016), the majority of our specimens had only 1 tooth on the dorsomesial surface of the coxa of the third maxilliped, and only 2 specimens had 2 teeth; so, we consider that this characteristic cannot be used to separate the 2 species. In addition, we observed in A. darrylfelderi other morphological variations, for example, most of our specimens have an unarmed rostrum (as mentioned in the original description of the species) but 2 of them have a terminal small spine or papilla and other specimens have antennal acicles without a ventromesial spine. This is important to highlight because those characters are used to distinguish other species within the genus Axianassa. Kensley and Heard (1990) pointed out that A. arenaria has a narrow to rounded rostrum ending in a small papilla (Fig. 5B, p. 565) similar to that observed in our specimens. On the other hand, A. arenaria, A. christyi, A. ngochoae, and A. sinica have an unarmed antennal acicle as pointed out by Anker and Pachelle (2016).
The morphology of the Z1 of A. darrylfelderi from the the Pérula Estuary has a close resemblance to the ZI of A. australis analyzed by Ngoc-Ho (1981), Rodrigues and Shimizu (1992) and Strasser and Felder (2005) (Table 2). Perhaps the greatest differences can be observed with those described by Rodrigues and Shimizu (1992), however, this work should be considered with caution since there are some inconsistencies in its illustrations. For example, figures 25 and 26 correspond to the left mandible and left lobe paragnath, respectively, and not to right and left mandibles as they mentioned, so it is possible that the larvae observed by Rodrigues and Shimizu (1992) were not so different from those described in the other works, and they did not illustrate other structures (e.g., subterminal plumose seta on the antennula or a spine on the antennal peduncle), thus we could conclude that the Z1 of species of Axianassa are relatively similar and the differentiation among species can be made only in advanced stages, as seen in other mud shrimp (e.g., De Oliveira et al., 2014; Miyabe et al., 1998; Pohle et al., 2011; Strasser & Felder, 1999) or even in other decapods as hermit or panopeid crabs (Salgado-Barragán & Ruiz-Guerero, 2005; Siddiqui et al., 1993). Concerning the differences in the ZI shown in table 2, the only reliable character that allowed us to clearly distinguish the ZI of A. darrylfelderi from that of A. australis is the presence of first pereopods as small bilobed buds in A. australis that are absent in the ZI of A. darrylfelderi. Miyabe et al. (1998) pointed out that the Zl of Callianassa japonica (= Neotrypaea japonica) and C. petalura (= N. petalura) could be differentiated solely by the presence (in the former) or the absence (in the latter) of a coxal seta present in the second maxilliped. We observed that the morphological characters used to separate A. darrylfelderi and A. australis are very variable and significantly hinder the differentiation between these species.
COI and 16S regions have been widely used to delimit species in crustaceans (Costa et al., 2007; Lefébure et al., 2006; Magalhães et al., 2016). According to our results, the divergence between A. darrylfelderi and A. australis is above the lower interspecific distances reported in the literature (Costa et al., 2007; Lefébure et al., 2006; Magalhães et al., 2016), which supports our hypothesis. Phylogenetic analysis showed confusing results. While phylogenetic inference with 16S region recovers both species as sister groups, the COI hypothesis groups both species in the same clade but with a low support. This could be due either to the lack of genetic information regarding Axianassa genus or to a close relationship between A. darrylfelderi and A. australis. Nonetheless, the haplotype networks revealed a great genetic distance between both species, supporting our hypothesis.
Table 2
Comparison between larva zoea I of A. australis and A. darrylfelderi. Rodrigues and Shimizu, 1992 (1); Ngoc-Ho, 1981 (2); Strasser and Felder, 2005 (3).
| Axianassa australis (1) | Axianassa australis (2) | Axianassa australis (3) | Axianassa darrylfelderi (Present study) | |
| Rostrum | As long as antennae | As long as antennae | As long as antennae | As long as antennae |
| Eyes | Round, sessile | Round, sessile | Round, sessile | Round, sessile |
| Abdomen | Somite 5 with lateral spines Somite 6 fused with telson, | Somite 5 with lateral spines Somite 6 fused with telson, | Somite 5 with lateral spines Somite 6 fused with telson | Somite 5 with lateral spines Somite 6 fused with telson |
| Antennule | Unsegmented | Unsegmented | Unsegmented | Unsegmented |
| Distal setae | 3 aesthetascs + 2 setae | 3 aesthetascs + 2 setae | 6 aesthetascs | 5 aesthestascs + 1 seta |
| Subterminal plumose | Absent | Present | Present | Present |
| Antenna: Spine on peduncle Exopod Endopod | Absent Spine + 10 setae 3 apical setae | Present Spine + 10 setae 3 apical setae | Present Spine + 8 – 10 setae 3 apical setae | Present Spine + 10 setae 3 apical setae |
| Mandibles | Asymmetrical | Asymmetrical | Asymmetrical | Asymmetrical |
| Maxillule: Endopod Coxal endite Basal endite | Unsegmented, 3 apical setae 4 setae 4 setae | Unsegmented, 3 apical setae 4 setae 4 setae | Unsegmented 2-3 apical setae 3-4 setae 2 setae, 2 spines | Unsegmented, 3 apical 4 setae 2 setae, 2 spines |
| Maxilla: Scaphognathite Endopod Coxal endite Basal endite | 5 setae Unsegmented, 2 setae 4 setae 8 setae | 5 setae Unsegmented, 2 setae 5 setae 8 setae | 3-5 setae Unsegmented, 2 setae 3-5 setae 7-10 setae | 5 setae Unsegmented, 2 setae 3-5 setae 7-9 setae |
| Maxilliped I: Exopod Endopod Basis | 2-segmented, 4 apical setae 4-segmented. 1,0,1,4 setae 3 mesial, 2 lateral | 2-segmented, 4 apical setae 4-segmented, 1,1,2,5 setae 1+2+1 setae | Unsegmented, 4 apical setae 4-segmented, 1,1,2,4-5 setae 1+2+1 setae | Unsegmented, 4 apical setae 4 segmented. 1,1,2,5 1+2+1 setae |
| Maxilliped 2: Exopod Endopod Basis | 2-segmented, 4 apical setae, 3 lateral setae 4-segmented. 0,0,3,5 setae No setae | 2-segmented, 4 apical setae 4-segmented, 0,0,2,4 setae No setae | Unsegmented, 4 apical setae 4-segmented, 0-1,0-1,0-2,4-5 setae No setae | Unsegmented, 4 apical setae 4-segmented. 1,1,2,5 setae No setae |
| Maxilliped 3: Exopod Endopod Pereopods | 2-segmented, no setae Absent Absent | 2-segmented, no setae Absent Pereopods 1 and 2 present as small buds | 2-segmented, no setae Absent Pereopod 1 as small bilobed bud | 2-segmented, no setae Absent Absent |
| Telson processes Medial spine Anomuran hair | 7 +7 Absent Present | 7 +7 Absent Present | 7 + 7 Absent Present | 7 + 7 Absent Present |
Acknowledgments
Special thanks to the biology students from Universidad de Guadalajara who supported the sampling. Thanks also to the field station of the Instituto de Biología (Universidad Nacional Autónoma de México) that offered important support in its facilities in Chamela. We are grateful to two anonymous reviewers whose comments improved the manuscript significantly.
References
Anker, A. (2010). The mud-shrimp genus Axianassa Schmitt, 1924 (Crustacea, Decapoda, Axianassidae) in the Indo-West Pacific, with description of a new species from French Polynesia. Zootaxa, 2557, 49–59. https://doi.org/10.11646/zootaxa.2557.1.5
Anker, A., & Lazarus, J. F. (2015). Description of two new associated infaunal decapod crustaceans (Axianassidae and Alpheidae) from the tropical eastern Pacific. Papéis Avulsos de Zoologia, 55, 115–129. https://doi.org/10.1590/0031-1049.2015.55.08
Anker, A., & Pachelle, P. P. G. (2016). Mud-shrimps of the genus Axianassa Schmitt, 1924 from Panama, with description of two new species (Decapoda: Gebiidea: Laomediidae). Zootaxa, 4111, 101–125. https://doi.org/10.11646/zootaxa.4111.2.1
Blanco-Rambla, J. P., & Kensley, B. (1998). Dos nuevas especies de camarones laomedidos del género Axianassa Schmitt, 1924 (Crustacea: Decapoda: Thalassinidea) para el Caribe sur. In XLVIII Convención Anual AsoVAC, Maracaibo, 9-13 de noviembre de 1998. Acta Científica Venezolana, 49, 349.
Boone, L. (1931). Anomuran, macruran Crustacea from the Bay of Panama and the freshwaters of the Canal zone. Bulletin of the American Museum of Natural History, 61, 137–189.
Costa, F. O., deWaard, J. R., Boutillier, J., Ratnasingham, S., Dooh, R. T., Hajibabaei, M. et al. (2007). Biological identifications through DNA barcodes: the case of the Crustacea. Canadian Journal of Fisheries and Aquatic Sciences, 64, 272–295. https://doi.org/10.1139/F07-008
Crandal, K. A., & Fitspatrick Jr., J. F. (1996). Crayfish molecular systematics: Using a combination of procedures to estimate phylogeny. Systematic Biology, 45, 1–26. https://doi.org/10.1093/sysbio/45.1.1
De Oliveira, D. B., Martinelle-Lemos, J. M., & Araújo-Abrunhosa, F. (2014). The complete larval development of the mud shrimp Upogebia vasquezi (Gebiidea: Upogebiidae) reared in the laboratory. Zootaxa, 3826, 517–543. https://doi.org/10.11646/zootaxa.3826.3.5
Edler, D., Klein, J., Antonelli, A., & Silvestro, D. (2020). raxmlGUI 2.0: A graphical interface and toolkit for phylogenetic analyses using RAxML. Methods in Ecology and Evolution, 12, 373–377. https://doi.org/10.1111/2041-210X.13512
Felder, D. L., & Robles, R. (2009). Molecular phylogeny of the family Callianassidae based on preliminary analyses of two mitochondrial genes. In J. W. Martin, K. A. Crandall, & D. L. Felder (Eds.), Decapod crustacean phylogenetics. Crustacean Issues. Koenemann, S. (Series ed.) Vol. 18 (pp. 327–342). Boca Raton, London, New York: CRC Press, Taylor & Francis Group.
Folmer, O., Black, M., Hoeh, W., Lutz, R., & Vrijenhoek, R. (1994). DNA primers for amplification of mitochondrial cytochrome c oxidase subunit I from diverse metazoan invertebrates. Molecular Marine Biology and Biotechnology, 3, 294–299.
Henmi, Y., Itani, G., Osawa, M., & Komai, T. (2022). A new species of the ghost shrimp genus Callianassa Leach, 1814 (Decapoda: Axiidea: Callianassidae) from Wakasa Bay, western Japan: the first representative of the genus from the Pacific region. Zootaxa, 5182, 465–478. https://doi.org/10.11646/zootaxa.5182.5.4
Hernández-Aguilera, J. L. (1998) Sobre una colección de talasínidos (Crustacea: Decapoda) de la costa del Pacífico de México, con la descripción de una especie nueva del género Biffarius. Ciencias Marinas, 24, 303–312. http://dx.doi.org/10.7773/cm.v24i3.754
Kensley, B., & Heard, R. (1990). The genus Axianassa (Crustacea: Decapoda: Thalassinidea) in the Americas. Proceedings of the Biological Society of Washington, 103, 558–572.
Komai, T., & Fujita, Y. (2019). A new species of the mud shrimp genus Axianassa Schmitt, 1924 (Decapoda: Gebiidea: Laomediidae) from the Ryukyu Islands, southern Japan. Zootaxa, 4658, 452–460. https://doi.org/10.11646/zootaxa.4658.3.2
Komai, T., Watanabe, T., Matsui, S., & Tamego, T. (2020). A new species of the mud shrimp genus Axianassa Schmitt, 1924 (Decapoda: Gebiidea: Laomeiidae) from Japon. Zootaxa, 4852, 461–474. https://doi.org/10.11646/zootaxa.4852.4.4
Konishi, K. (1989). Larval development of the mud shrimp Upogebia (Upogebia) major (De Hann) (Crustacea: Thalassinidea: Upogebiidae) under laboratory conditions, with comments on larval characters of thalassinid families. Bulletin of the National Research Institute of Aquaculture, 15, 1–17.
Lefébure, T., Douady, C. J., Gouy, M., & Gilbert, J. (2006). Relationship between morphological taxonomy and molecular divergence within Crustacea: proposal of a molecular threshold to help species delimitation. Molecular Phylogenetics and Evolution, 40, 435–447. https://doi.org/10.1016/j.ympev.2006.03.014
Liu, W., & Liu, R. (2010). First record of the gebiidean genus Axianassa Schmitt, 1924 (Crustacea: Decapoda: Gebiidea: Axianassidae) in the West Pacific, with description of a new species from the South China Sea. Raffles Bulletin of Zoology, 58, 193–197. http://dx.doi.org/10.5281/zenodo.5342684
Magalhães, T., Robles, R., Felder, D. L., & Mantelatto, F. L. (2016). Integrative taxonomic study of the purse crab genus Persephona Leach, 1817 (Brachyura: Leucosiidae): combining morphology and molecular data. Plos One, 11, e0152627. https://doi.org/10.1371/journal.pone.0152627
Mantelatto, F. L., Robles, R., Biagi, R., & Felder, D. L. (2006). Molecular analysis of the taxonomic and distributional status for the hermit crab genera Loxopagurus Forest, 1964, and Isocheles Stimpson, 1858 (Decapoda, Anomura, Diogenidae). Zoosystema, 28, 495–506.
Mantelatto F. L., Robles, R., Schubart, C. D., & Felder, D. L. (2009). Molecular phylogeny of the genus Cronius Stimpson, 1860, with reassignment of C. tumidulus and several American species of Portunus to the genus Achelous De Haan, 1833 (Brachyura: Portunidae). In J. W. Martin, K. A. Crandall, & D. L. Felder (Eds.), Crustacean issues: Decapod Crustacean Phylogenetics (pp. 567–579). Boca Raton: Taylor and Francis/ CRC Press.
Matzen da Silva, J., Creer, S., Dos Santos, A., Costa, A. C., Cunha, M. R., Costa, F. O. et al. (2011). Systematic and evolutionary insights derived from mtDNA COI barcode diversity in the decapoda (Crustacea: Malacostraca).
Plos One, 6, e19449. http://dx.doi.org/10.1371/journal.pone.0019449
Miyabe, S., Konishi, K., Fukuda, Y., & Tamaki, A. (1998). The complete larval development of the ghost shrimp, Callianassa japonica Ortmann, 1891 (Decapoda: Thalassinidea: Callianassidae), reared in the laboratory. Crustacean Research, 27, 101–121. https://doi.org/10.18353/crustacea.27.0_101
Morrison, C. L., Harvey, A. W., Lavery, S., Tieu, K., Huang, Y., & Cunningham, C. W. (2002). Mitochondrial gene rearrangements confirm the parallel evolution of the crab-like form. Proceedings of the Royal Society of London, Series B, 269, 345–350. https://doi.org/10.1098/rspb.2001.1886
Müller, J., Müller, K., Neinhuis, C., & Quandt, D. (2010). PhyDE-Phylogenetic Data Editor, versión 0.9971. Program distributed by the autor: http://www.phyde.de
Nates, S. F., Felder, D. L., & Lemaitre, R. (1997). Comparative larval development in two species of the burrowing ghost shrimp genus Lepidophthalmus (Decapoda: Callianassidae). Journal of Crustacean Biology, 17, 497–519. https://doi.org/10.2307/1549444
Ngoc-Ho, N. (1981). A taxonomic study of the larvae of four thalassinid species (Decapoda, Thalassinidea) from the Gulf of Mexico. Bulletin of the British Museum (Natural History) Zoology, 40, 237–273.
Paradis, E., & Schliep, K. (2019). ape 5.0: an environment for modern phylogenetics and evolutionary analyses in R. Bioinformatics, 35, 526–528. https://doi.org/10.1093/bioinformatics/bty633
Pohle, G., Santana, W., Jansen, G., & Greenlaw, M. (2011). Plankton-caught zoeal stages and megalopa of the lobster shrimp Axius serratus (Decapoda: Axiidae) from the Bay of Fundy, Canada, with a summary of axiidean and gebiidean literature on larval descriptions. Journal of Crustacean Biology, 31, 82–99. http://dx.doi.org/10.1651/10-3321.1
R Core Team. (2021). R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. URL https://www.R-project.org/
Robles, R., Dworschak, P. C., Felder, D. L., Poore, G. C. B., & Mantelatto, F. L. (2020). A molecular phylogeny of Callianassidae and related families (Crustacea: Decapoda: Axiidea) with morphological support. Invertebrate Systematics, 34, 113–132. https://doi.org.10.1071/IS9021
Robles, R., Tudge, C. C., Dworschak, P. C., Poore, G. C. B., & Felder, D. L. (2009). Molecular phylogeny of the Thalassinidea based on nuclear and mitochondrial genes. In J. W. Martin, K. A. Crandall, & D. L. Felder (Eds.), Crustacean Issues 18: Crustacean issues: Decapod Crustacean Phylogenetics (pp. 309–326). Boca Raton: Taylor and Francis/ CRC Press.
Rodrigues, S. A., & Shimizu, R. M. (1992). Description of a new Axianassa (Crustacea: Decapoda: Thalassinidea) from Brazil, and its first larval stage. Biological Society of Washington, 105, 317–323.
Salgado-Barragán, J., & Ruiz-Guerrero, M. (2005). Larval development of the eastern Pacific mud crab Acantholobulus mirafloresensis (Abele and Kim, 1989) (Decapoda: Brachyura: Panopeidae) described from laboratory-reared material. Invertebrate Reproduction & Development, 47, 133–145. https://doi.org/10.1080/07924259.2005.9652154
Schmitt, W. L. (1924). The macruran, anomuran and stomato-
pod Crustacea. Bijdragen tot der kennis der fauna van Curaçao. Resultaden eener reis von Dr. C. J. van der Horst in 1920. Bijdragen tot de Dierkunde, 23, 9–82. https://doi.org/10.1163/26660644-02301005
Semarnat (Secretaría del Medio Ambiente y Recursos Naturales). (2010). Norma Oficial Mexicana NOM-059-SEMARNAT-2010, Protección ambiental – Especies nativas de México de flora y fauna silvestres – Categorías de riesgo y especificaciones para su inclusión, exclusión o cambio – Lista de especies en riesgo. Diario Oficial de la Federación. 30 de diciembre de 2010, Segunda Sección, México.
Siddiqui, E. A., McLaughlin, P. A., & Crain, J. A. (1993). Larval development of the hermit crab Clibanarius albidigitus (Crustacea: Anomura: Diogenidae) reared under laboratory conditions. Marine Biology, 116, 603–613. https://doi.org/10.1007/BF00355479
Strasser, K. M., & Felder, D. L. (1999). Larval development in two populations of the ghost shrimp Callichirus major (Decapoda: Thalassinidea) under laboratory conditions. Journal of Crustacean Biology, 19, 844–878. http://www.jstor.org/stable/1549168
Strasser, M. K., & Felder, L. D. (2005). Larval development of the mud shrimp Axianassa australis (Decapoda: Thalassinidea) under laboratory conditions. Journal of Natural
History, 39, 2289–2306. https://doi.org/10.1080/00222930500061320
Teles, J. N., & Mantelatto, F. L. (2023). New records of the shrimp-like decapods Alpheus angulosus, Alpheus buckupi, and Axianassa australis from Maranhão, Brazil. Nauplius, 31, e2023022. https://doi.org/10.1590/2358-2936e2023022
WoRMS Editorial Board. (2024). World register of marine species. https://doi.org/10.14284/170