Densidad poblacional y madurez sexual de Ambystoma lermaense, una especie amenazada en el centro de México
Gerardo Legorreta-Balbuena a, Carlos Omar Becerra-Soria b, Susana Hernández-Monasterio b, Gabriela Parra-Olea b, *
a Universidad Nacional Autónoma de México, Instituto de Investigaciones Biomédicas, Laboratorio de Biología Integrativa, Tercer Circuito s/n, Ciudad Universitaria, Col. Copilco Universidad, 04510 Ciudad de México, Mexico
b Universidad Nacional Autónoma de México, Instituto de Biología, Departamento de Zoología, Tercer Circuito s/n, Ciudad Universitaria, Col. Copilco Universidad, 04510 Ciudad de México, Mexico
*Corresponding author: gparra@ib.unam.mx (G. Parra-Olea)
Received: 21 January 2025; accepted: 14 November 2025
Abstract
Ambystoma lermaense, an endangered salamander species endemic to central Mexico, faces significant threats from habitat degradation, pollution, and human activities. This study evaluates the population density, structure, and sexual maturity of A. lermaense in Capilla Vieja, Estado de México, over 3 time periods: 2009, 2010, and 2020. Using visual encounter surveys along a 730-m stream transect, we recorded population densities of 0.021 individuals/m² in 2009, 0.070 individuals/m² in 2010, and 0.075 individuals/m² in 2020, reflecting relative stability despite anthropogenic pressures. Juveniles accounted for 87% of the population, with sexual maturity observed at a snout-vent length of 60 mm, supported by histological evidence of vitellogenic oocytes in females and mature sperm in males. Our study provides important insights into the population dynamics and sexual maturity of A. lermaense in the sampling locality, where this species had not been previously studied.
Keywords: Ambystomatidae; Population structure; Paedomorphosis; Vitellogenic oocytes; Mature sperm; Decline
Resumen
Ambystoma lermaense, una especie de salamandra en peligro de extinción endémica del centro de México, enfrenta amenazas significativas debido a la degradación del hábitat, la contaminación y las actividades humanas. Este estudio evalúa la densidad poblacional, estructura y madurez sexual de A. lermaense en Capilla Vieja, Estado de México, durante 3 períodos: 2009, 2010 y 2020. Utilizando muestreos visuales a lo largo de un transecto de 730 m en un arroyo, registramos densidades poblacionales de 0.021 individuos/m² en 2009, 0.070 individuos/m² en 2010 y 0.075 individuos/m² en 2020, lo cual refleja una estabilidad relativa a pesar de las presiones antropogénicas. Los juveniles representaron 87% de la población y se observó madurez sexual a un tamaño de 60 mm de longitud hocico-cloaca, sustentado por evidencia histológica de ovocitos vitelogénicos en hembras y espermatozoides maduros en machos. Nuestro estudio proporciona información importante sobre la dinámica poblacional y la madurez sexual de A. lermaense en la localidad de muestreo, donde esta especie no había sido estudiada previamente.
Palabras clave: Ambystomatidae; Estructura poblacional; Paedomorfosis; Ovocitos vitelogénicos; Espermatozoides maduros; Declive
Introduction
Amphibians worldwide are experiencing unprecedented declines, driven by a combination of habitat loss, pollution, climate change, invasive species, and disease (Grant et al., 2016; Stuart et al., 2004). These declines are particularly concerning for high-altitude amphibians, which often have restricted ranges and specialized habitat requirements (Green, 2003; Pounds, 2001; Stuart et al., 2004). As indicators of ecosystem health, amphibians play critical roles in maintaining ecological balance, yet they are disproportionately affected by anthropogenic pressures (Blaustein & Wake, 1990). Among these, Ambystoma lermaense, an endangered salamander species endemic to central Mexico, exemplifies the challenges faced by amphibians in fragmented and degraded habitats (IUCN SSC Amphibian Specialist Group, 2015).
Ambystoma lermaense, inhabits high-altitude streams at elevations ranging from 2,500 to 3,000 m asl (IUCN SSC Amphibian Specialist Group, 2015). This facultatively paedomorphic species is found in aquatic habitats such as lakes, ponds, marshes, and occasionally streams (Everson et al., 2021; IUCN SSC Amphibian Specialist Group, 2015), which are increasingly impacted by human activities (Reilly & Brandon, 1994). Agricultural expansion, livestock grazing, pollution, and urban development threaten the quality and connectivity of these aquatic habitats (Egea-Serrano et al., 2012; Robles-Mendoza et al., 2009). These pressures, combined with seasonal hydrological fluctuations and climate variability, pose significant risks to the long-term viability of A. lermaense populations (Dirzo et al., 2022; Huacuz-Elías, 2003).
Understanding population dynamics is a cornerstone of effective conservation planning. Accurate data on population density, structure, and trends are essential for identifying populations at risk and prioritizing interventions (Pechmann et al., 1991). Previous studies on species of the genus Ambystoma have revealed a wide spectrum of population densities, ranging from critically low levels in urbanized environments to exceptionally high densities in protected areas. For instance, the Mexican axolotl (A. mexicanum) exhibits densities as low as 0.001 individuals per square meter in the urban canals of Xochimilco (Contreras et al., 2009), while A. rivulare densities reach 2.3 individuals per square meter in the protected natural area Reserva de la Biósfera Mariposa Monarca (Huacuz-Elías, 2003). These disparities underscore the profound impact of habitat quality and protection on amphibian populations.
Despite its resilience in suboptimal conditions, A. lermaense remains highly vulnerable to habitat degradation and fragmentation (Jacinto-Maldonado et al., 2023). In the Capilla Vieja region of Estado de México, semi-intensive livestock activities and seasonal stream drying compound the challenges faced by this species (previously recognized in this locality as A. granulosum; Everson et al., 2021). As part of a natural protected area, Capilla Vieja offers a unique opportunity to study how A. lermaense populations persist in a landscape that combines elements of protection and human impact. The site’s connectivity to a broader watercourse system also provides insights into the role of habitat corridors in sustaining population viability.
This study examines the population density, structure, and sexual maturity of A. lermaense in Capilla Vieja over 3 time periods: 2009, 2010, and 2020. By situating these findings within the broader context of Ambystoma conservation, we aim to draw comparisons with other populations and species to better understand the factors driving population trends. The research also highlights the critical role of protected areas and localized conservation actions in addressing the multifaceted threats to amphibians. This work contributes to the growing body of knowledge needed to inform strategies for safeguarding A. lermaense and other endangered amphibians in Mexico’s diverse ecosystems.
Materials and methods
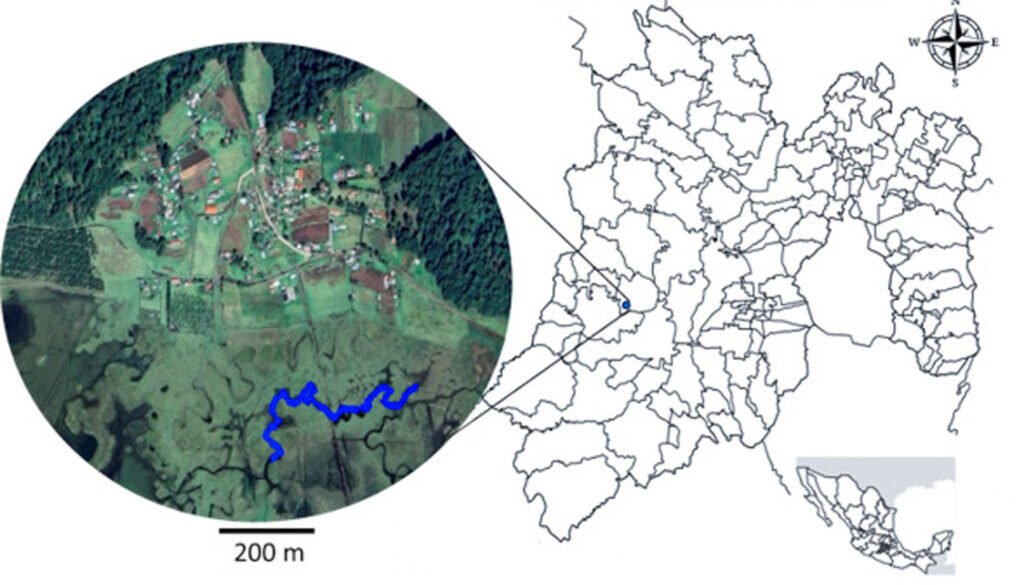
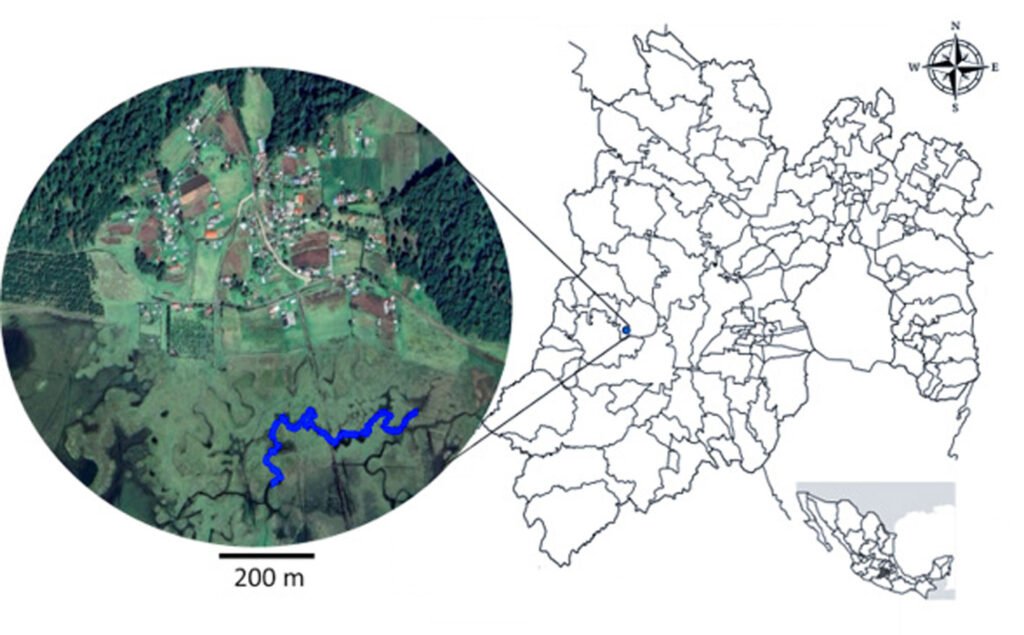
The study was conducted in a section of a stream located in Capilla Vieja, municipality of Amanalco de Becerra, Estado de México, at coordinates 19º13’22” N, 99º59’12” W, with an elevation ranging from 2,790 to 2,870 m asl (Fig. 1). The predominant vegetation in this area is pine-oak-fir forest. The climate is temperate subhumid, with summer rains, an average annual temperature of 12 °C, and temperature extremes ranging from 0.5 °C to 29.7 °C (Rentería et al., 2005). Capilla Vieja is part of the sustainable ecosystem management subzone for agricultural areas within the Natural Protected Area known as Área de Protección de Recursos Naturales de las Cuencas de los Ríos Valle de Bravo, Malacatepec, Tilostoc y Temascaltepec (Semarnat, 2018).
The study stream crosses an extensive grassland plain that is used for semi-intensive livestock activities, including sheep, cattle, and horses. The stream during the rainy season features shallow areas with nearly stagnant water. In certain sections, the bottom is extremely soft, with mud depths ranging from 15 to 25 cm and exceeding 50 cm in some points. The stream width varies between 2 and 5 m, expanding during the rainy season and shrinking or disappearing during the dry season. This stream is part of a connected watercourse system that flows in a northeast-southwest direction, contributing to the water supply of the Capilla Vieja lagoon (Comisión Nacional de Áreas Naturales Protegidas, 2018).
Samplings were conducted once a month from July to October during 3 periods corresponding to the years 2009, 2010 and 2020. Each month, a 730 m transect with a width of 3 m along the stream was surveyed (starting coordinates: 19°13’10.81” N, 99°59’4.95” W; ending coordinates: 19°13’3.73” N, 99°59’15.13” W). Sampling was carried out using the visual encounter survey method between 09:00 and 14:00 hours (Heyer et al., 1994; Lips et al., 1999), recording the total number of individuals. Each individual was captured using hand nets and placed in 20-liter plastic containers filled with water from the stream.
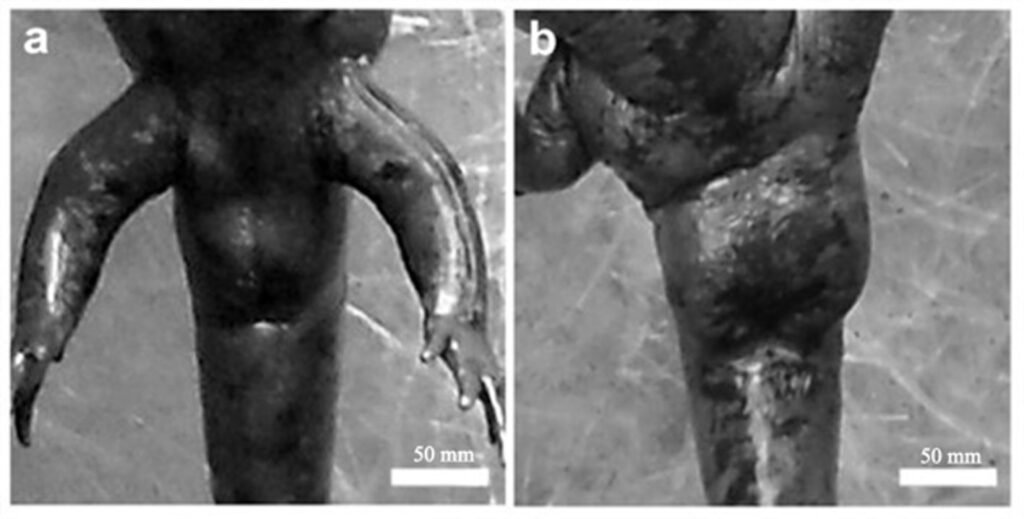
Morphometric measurements were recorded for each specimen: snout-vent length (SVL), total length (TL), and weight (W). Individuals were classified into 2 categories: adult metamorphic (males and females) and larvae (juveniles), a category that included individuals with gills for whose sex could not be determined. Population density was calculated by dividing the total number of individuals (N) by the sampled area of the stream (N/m²) (Williams et al., 2002), and it was estimated for each sampling period. Males and females were identified based on the size and shape of the cloaca (Aguilar-Miguel et al., 2009; Taylor, 1940). In females, the cloaca is flat and smaller (Fig. 2a), whereas in males it is enlarged due to high glandular activity (Fig. 2b).
To estimate the size at sexual maturity, a sample of 52 individuals was collected from random points along the stream during the 2009 and 2010 sampling periods. The sample included individuals ranging in size from 50 to 115 mm in SVL. An ANOVA was used to assess the relationship between body size and sexual maturity.
To euthanize the specimens, they were anesthetized using a 0.1% solution of MS-222 (tricaine methanesulfonate) for 20 min (Mitchell, 2009; Rubio-Limonta & Silveira-Coffigny, 2009). A longitudinal ventral incision was made to expose the ovaries and testes. The gonads were fixed in 4% paraformaldehyde for 24 h at 4 °C. Standard histological techniques for light microscopy were applied: dehydration in graded alcohols, clearing with xylene, embedding in paraplast, sectioning at 5 µm, and Hematoxylin-Eosin staining (Aguilar-Miguel et al., 2009). Digital images of spermatogenesis and oogenesis stages were captured using a Nikon Coolpix 4300 (4 MP) digital camera mounted on a Nikon Optiphot-2 upright microscope and calibrated with ImageJ software.
Results
A total of 366 individuals were recorded across the 2,190 m² of the sampled stream during the 3 periods (Table 1). The period with the lowest abundance was 2009, with 46 individuals, while the highest was 2020, with 165 individuals. The population density of A. lermaense increased over the sampling periods. In 2009, the density was 0.021 ind/m², in 2010, it increased to 0.070 ind/m², and finally, in 2020, it reached 0.075 ind/m².
Table 1
Total number of individuals recorded during the sampling periods.
| Period | July | August | September | October | Total |
| 2009 | 4 | 23 | 14 | 5 | 46 |
| 2010 | 4 | 56 | 70 | 25 | 155 |
| 2020 | 7 | 130 | 23 | 5 | 165 |
| Total | 15 | 209 | 107 | 35 | 366 |
Population structure. Of the total number of individuals found across the 3 periods, 87% were juveniles, 6% were metamorphic females, and 7% were metamorphic males. Throughout the 3 sampling periods, the months with the highest number of larvae were August and September, for metamorphic females, July and September, and for metamorphic males, August and September (Fig. 3).
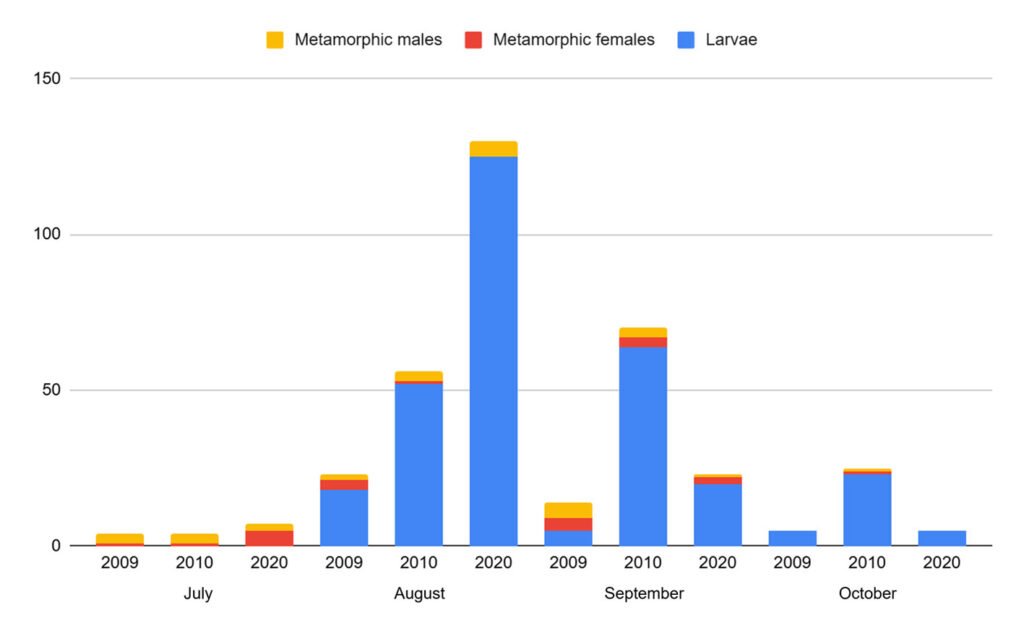
Table 2
Morphometric data (average, minimum, and maximum) of individuals analyzed histologically to determine sexual maturity. SVL = Snout-vent length, TL = total length (mm), and weight (g).
| Category | Metamorphic females | Metamorphic males | Larvae |
| Avg. SVL | 81.4 | 90.8 | 62.5 |
| Min. SVL | 60 | 60 | 50 |
| Max. SVL | 100 | 115 | 85 |
| Avg. TL | 156.1 | 167.9 | 114.6 |
| Min. TL | 130 | 110 | 85 |
| Max. TL | 180 | 220 | 155 |
| Avg. weight | 23.53 | 20.13 | 9.57 |
| Min. weight | 10 | 8 | 4 |
| Max. weight | 42 | 36 | 20 |
| Gonadal development (Yes/No) | Yes | Yes | No |
| Number of individuals | 14 | 24 | 14 |
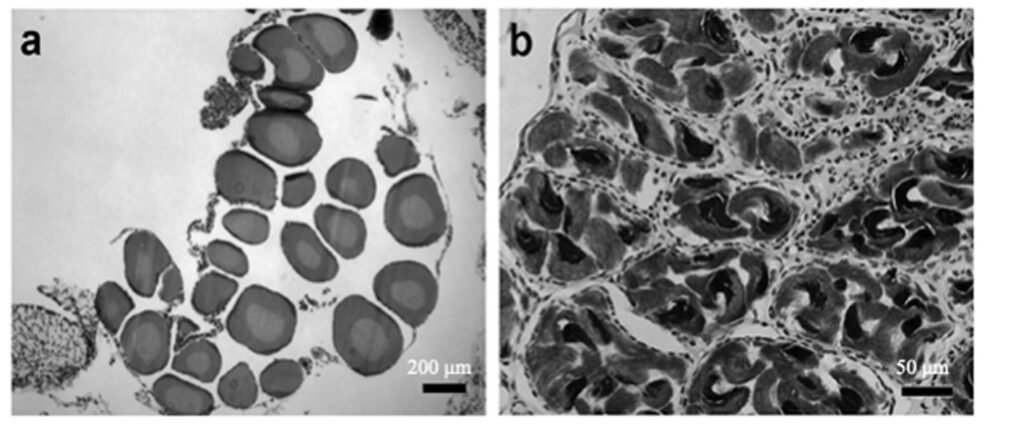
Sexual maturity. Of the 52 dissected individuals, 24 were males, 14 females and 14 larvae. Males had an SVL ranging from 60 to 115 mm, females from 60 to 100 mm, and larvae from 50 to 85 mm (Table 2). According to the ANOVA, a significant association was found between size and the different age categories (p < 0.05). Through histological analysis, 38 individuals exhibited sexual development (Table 2). We determined that sexual maturity occurs at an SVL of 60 mm, as vitellogenic oocytes in females and mature sperm in males were observed at this size (Fig. 4).
Discussion
Our study on Ambystoma lermaense in Capilla Vieja provides critical insights into its population dynamics, sexual maturity, and conservation status. By situating these findings within a broader ecological and conservation context, we can deepen our understanding of the factors influencing the persistence of this species and its relatives. The gradual increase in population density from 0.021 individuals/m² in 2009 to 0.075 individuals/m² in 2020 is a positive indicator of stability. However, these densities remain substantially lower than those reported by Huacuz (2003) in the Reserva de la Biosfera Mariposa Monarca (2.3 individuals/m²). This stark contrast underscores the influence of habitat quality and protection on population viability. The Capilla Vieja site, despite being part of a protected natural area, is exposed to semi-intensive livestock activities and seasonal hydrological fluctuations, both of which could limit carrying capacity for A. lermaense.
Examining densities and threats across 3 Ambystoma species provides valuable comparative insights: Ambystoma mexicanum, critically low densities in Xochimilco lake (0.001 individuals/m²) highlight severe impacts from urbanization and habitat degradation (Contreras et al., 2009). Ambystoma altamirani, densities ranging from 0.016 to 0.027 individuals/m² in trout-free streams in Estado de México suggest that invasive species exacerbate population declines (Lemos-Espinal et al., 1999). Ambystoma ordinarium, densities of 0.030-0.050 individuals/m² in Michoacán State highlight the challenges posed by deforestation and agricultural expansion (Aguilar-Miguel, 2005). These comparisons reinforce the role of protected areas in supporting higher population densities and the importance of mitigating threats in unprotected habitats.
The discovery that sexual maturity in A. lermaense occurs at a SVL of 60 mm aligns with findings for other facultatively paedomorphic Ambystoma species. Paedomorphosis, the retention of larval traits in adults, allows these salamanders to reproduce in aquatic habitats, potentially enhancing resilience in fragmented or fluctuating environments. However, the high proportion of juveniles (87%) in Capilla Vieja suggests a reliance on consistent recruitment, which could be disrupted by environmental changes.
The Capilla Vieja site is part of a connected watercourse system, but its fragmentation due to agricultural activities and seasonal stream drying limits habitat availability. Maintaining connectivity between breeding and non-breeding habitats is critical for sustaining gene flow and reducing local extinction risks. Lessons from the Reserva de la Biosfera Mariposa Monarca, where robust habitat connectivity supports high densities of this species, should guide management strategies for fragmented landscapes.
High-altitude amphibians like A. lermaense are particularly vulnerable to climate change. Rising temperatures and altered precipitation patterns could shrink suitable habitat ranges and exacerbate seasonal drying of streams. Proactive measures, such as restoring vegetation cover and monitoring hydrological changes, are necessary to mitigate these effects. While protected areas like the Reserva de la Biosfera Mariposa Monarca demonstrate the effectiveness of large-scale conservation, localized efforts are equally vital. Capilla Vieja’s inclusion in a protected area offers an opportunity to implement targeted interventions, such as: enhancing water quality through pollution control; reducing the impact of livestock on stream habitats; and introducing community-based conservation programs to engage local stakeholders.
The findings on A. lermaense reflect broader trends in amphibian conservation. Amphibians worldwide face similar threats of habitat degradation, invasive species, and climate change. This study contributes to the growing body of evidence that localized conservation actions, combined with broader protections, can stabilize and even enhance populations of threatened species. Future studies could explore: genetic diversity, assessing genetic variation in A. lermaense populations to evaluate the effects of fragmentation on genetic health. Behavioral ecology, investigating breeding behavior, dispersal patterns, and predator-prey dynamics to inform conservation strategies. Impact of invasive species, examining whether invasive species, such as trout, are encroaching on nearby A. lermaense habitats.
The relatively stable population density of Ambystoma lermaense in Capilla Vieja reflects its resilience but highlights the challenges posed by habitat degradation and fragmentation. Comparisons with other Ambystoma species and populations in protected areas underscore the importance of habitat quality and connectivity. To ensure the long-term survival of A. lermaense and its relatives, conservation efforts must integrate habitat restoration, community engagement, and climate adaptation strategies. As amphibians are critical indicators of ecosystem health, their protection benefits biodiversity and ecological integrity on a broader scale.
Acknowledgments
We gratefully acknowledge Conahcyt for the scholarship awarded to GL-B during his PhD studies in Biological Sciences at UNAM (2009–2011; registration number 161331). We also thank Gabriel Gutiérrez-Ospina for funding provided through GlaxoSmithKline, as well as the annual budget allocated to the Integrative Biology Laboratory by the Administrative Secretariat of the Institute of Biomedical Research, UNAM, which partially supported this project. Additional support was provided by DGAPA-UNAM (PAPIIT-UNAM, IN208024) and Secihti(CBF-2025-G-418) to GP-O. We are grateful to Fernando Arana Magallón, coordinator of the Biological and Aquaculture Research Center of Cuemanco (CIBAC-UAM-X), for his assistance with fieldwork, laboratory work, and specimen collection. We also thank Miguel Tapia-Rodríguez, head of the Microscopy Unit at IIB-UNAM, for his support in figure design. We acknowledge Semarnat for the permits granted for this study (Nos. SGPA/DGVS/00417/09, SGPA/DGVS/02729/10, and SGPA/DGVS/03197/20). Finally, we thank the two anonymous reviewers and the section editor for their valuable comments, which improved the manuscript.
References
Aguilar-Miguel, X. (2005). Ambystoma ordinarium. Algunas especies de anfibios y reptiles contenidos en el Proyecto de Norma Oficial Mexicana PROY-NOM-059-ECOL-2000. Facultad de Ciencias, Centro de Investigación en Recursos Bióticos, Universidad Autónoma del Estado de México.
Aguilar-Miguel, X., Legorreta-Balbuena, G., & Casas-Andreu, G. (2009). Reproducción ex situ en Ambystoma granulosum y Ambystoma lermaense (Amphibia: Ambystomatidae). Acta Zoológica Mexicana, 25, 443–454. https://doi.org/10.21829/azm.2009.253652
Blaustein, A. R., & Wake, D. B. (1990). Declining amphibian populations: a global phenomenon? Trends in Ecology & Evolution, 5, 203–204. https://doi.org/10.1016/0169-5347(90)90129-2
Semarnat (Secretaría de Medio Ambiente y Recursos Naturales). (2018). Acuerdo por el que se da a conocer el resumen del Programa de Manejo del Área Natural Protegida con la categoría de área de protección de recursos naturales cuencas de los ríos Valle de Bravo, Malacatepec, Tilostoc y Temascaltepec, Estado de México. Diario Oficial de la Federación. Normateca Ambiental. https://normatecambiental.org/2018/12/05/acuerdo-por-el-que-se-da-a-conocer-el-programa-de-manejo-del-area-natural-protegida-de-cuencas-de-los-rios-valle-de-bravo-malacatepec-tilostoc-y-temascaltepec-estado-de-mexico/
Contreras, V., Martínez-Meyer, E., Valiente, E., & Zambrano, L. (2009). Recent decline and potential distribution in the last known wild populations of the critically endangered Mexican Axolotl. Biological Conservation, 142, 2881–2885. https://doi.org/10.1016/j.biocon.2009.07.008
Dirzo, R., Ceballos, G., & Ehrlich, P. R. (2022). Circling the drain: the extinction crisis and the future of humanity. Philosophical Transactions of the Royal Society B, 377, 20210378. https://doi.org/10.1098/rstb.2021.0378
Egea-Serrano, A., Relyea, R. A., Tejedo, M., & Torralva, M. (2012). Understanding the impact of chemicals on amphibians: a meta-analytic review. Ecology and Evolution, 2, 1382–1397. https://doi.org/10.1002/ece3.249
Everson, K. M., Gray, L. N., Jones, A. G., Lawrence, N. M., Foley, M. E., Sovacool, K. L. et al. (2021). Geography is more important than life history in the recent diversification of the tiger salamander complex. Proceedings of the National Academy of Sciences, 118, e2014719118. https://doi.org/10.1073/pnas.2014719118
Grant, E. H. C., Miller, D. A. W., Schmidt, B. R., Adams, M. J., Amburgey, S. M., Chambert, T. et al. (2016). Quantitative evidence for the effects of multiple drivers on continental-scale amphibian declines. Scientific Reports, 6, 25625. https://doi.org/10.1038/srep25625
Green, D. M. (2003). The ecology of extinction: population fluctuation and decline in amphibians. Biological Conservation, 111, 331–343. https://doi.org/10.1016/S0006-3207(02)00302-6
Heyer, W. R., Donnelly, M. A., McDiarmid, R. W., Hayek, A. C., & Foster, M. S. (1994). Measuring and monitoring biological diversity: standard methods for amphibians. Washington D.C.: Smithsonian Institution Press.
Huacuz-Elías, D. C. (2003). Estado de conservación de Ambystoma lermaense Taylor, (1940) en el Santuario Sierra Chincua de la Reserva de la Biosfera “Mariposa Monarca”. Revista de la Facultad de Biología de la UMSNH, 5, 51–63.
IUCN SSC Amphibian Specialist Group. (2015). Ambystoma lermaense (errata version published in 2020). The IUCN Red List of Threatened Species 2015: e.T1093A176771946. https://dx.doi.org/10.2305/IUCN.UK.2015-4.RLTS.T1093A176771946.en
Jacinto-Maldonado, M., González-Salazar, C., Basanta, M. D., García-Peña, G. E., Saucedo, B., Lesbarrères, D. et al. (2023). Water pollution increases the risk of chytridiomycosis in Mexican amphibians. EcoHealth, 20, 74–83. https://doi.org/10.1007/s10393-023-01631-0
Lemos-Espinal, J. A., Smith, G. R., Ballinger, R. E., & Ramírez-Bautista, A. (1999). Status of protected endemic salamanders (Ambystoma: Ambystomatidae: Caudata) in the transvolcanic belt of Mexico. British Herpetological Society Bulletin, 68, 1–4.
Lips, K. R., Reaser., J. K., & Young, B. (1999). Monitoreo de anfibios en América Latina: manual de protocolos. Herpetological Circular No. 30. https://amphibiaweb.org/resources/Anfibios.pdf
Mitchell, M. A. (2009). Anesthetic considerations for amphibians. Journal of Exotic Pet Medicine, 18, 40–49. https://doi.org/10.1053/j.jepm.2008.11.006
Pechmann, J. H., Scott, D. E., Semlitsch, R. D., Caldwell, J. P., Vitt, L. J., & Gibbons, J. W. (1991). Declining amphibian populations: the problem of separating human impacts from natural fluctuations. Science, 253, 892–895. https://doi.org/10.1126/science.253.5022.892
Pounds, J. (2001). Climate and amphibian declines. Nature, 410, 639–640. https://doi.org/10.1038/35070683
Reilly, S. M., & Brandon, R. A. (1994). Partial paedomorphosis in the Mexican stream ambystomatids and the taxonomic status of the genus Rhyacosiredon Dunn. Copeia, 1994, 656–662. https://doi.org/10.2307/1447181
Rentería, G., Cota, R. G., & Ortega, E. (2005). Descripción del medio natural de la cuenca del Valle de Bravo, Estado de México. Jiutepec, Morelos: Instituto Mexicano de Tecnología del Agua.
Robles-Mendoza, C., García-Basilio, C., Cram-Heydrich, S., Hernández-Quiroz, M., & Vanegas-Pérez, C. (2009). Organophosphorus pesticides effect on early stages of the axolotl Ambystoma mexicanum (Amphibia: Caudata). Chemosphere, 74, 703–710. https://doi.org/10.1016/j.chemosphere.2008.09.087
Rubio-Limonta, M., & Silveira-Coffigny, R. (2009). Bienestar de los animales acuáticos, con fines de control sanitario. REDVET. Revista Electrónica de Veterinaria, 10. https://www.redalyc.org/pdf/636/63617143009.pdf
Stuart, S. N., Chanson, J. S., Cox, N. A., Young, B. E., Rodrigues, A. S., Fischman, D. L. et al. (2004). Status and trends of amphibian declines and extinctions worldwide. Science, 306, 1783–1786. https://doi.org/10.1126/science.1103538
Taylor, E. H. (1940). A new Rhyacosiredon (Caudata) from western México. Herpetologica, 1, 171–176.
Williams, L. R., Crosswhite, D. L., & William, M. G. (2002). Short-term effect of riparian disturbance on Desmog-
nathus brimleyorum (Plethodontidae) at natural spring in Oklahoma, U.S.A. The Southwestern Naturalist, 47,
611–613. https://doi.org/10.2307/3672667