Contribución a la diversidad y distribución del género Scleroderma (Boletales: Sclerodermataceae) en México
Erika Cecilia Pérez-Ovando a, Bernardo Águila b, Wilfredo A. Matamoros a, Olivia Ayala-Vásquez c, d, Roberto Garibay-Orijel b, *
a Universidad Autónoma de Ciencias y Artes de Chiapas, Instituto de Ciencias Biológicas, Programa de Doctorado en Ciencias en Biodiversidad y Conservación de Ecosistemas Tropicales, Libramiento Norte Poniente, Col. Lajas Maciel, 29039 Tuxtla Gutiérrez, Chiapas, Mexico
b Universidad Nacional Autónoma de México, Instituto de Biología, Departamento de Botánica, Tercer Circuito s/n, Ciudad Universitaria, Coyoacán, 04510 Ciudad de México, Mexico
c Secretaría de Ciencia, Humanidades, Tecnología e Innovación, Av. Insurgentes Sur 1582, Col. Crédito Constructor, Benito Juárez, 03940 Ciudad de México, Mexico
d Colegio de Postgraduados, Campus Montecillo, Departamento de Edafología, Laboratorio de Micorrizas, 56230 Montecillo, Estado de México, Mexico
* Corresponding author: rgaribay@ib.unam.mx (R. Garibay-Orijel)
Received: 14 March 2025; accepted: 04 November 2025
Abstract
The order Boletales is one of the most diverse and widespread groups of macromycetes globally, with the genus Scleroderma being a significant representative. Despite the ecological, medical, and biocultural importance of this genus, studies on its systematics and genetic diversity in Mexico are scarce. Here, we studied the diversity and distribution of Scleroderma in Mexico using all available information (voucher registers, voucher DNA, and environmental DNA). A total of 120 nrITS DNA sequences from different species were analyzed, including 27 from environmental samples, while the remaining sequences were obtained from public databases. In Mexico, 15 Scleroderma species have been cited by morphological taxonomy, while molecular evidence suggest around 24 taxa. Phylogenetic analyses divided the genus into 4 main clades. Clade A corresponds to section Scleroderma and includes S. albidum s.l., S. areolatum, and S. cepa/laeve; clade B includes species of section Sclerangium and S. anomalosporum; clade C includes only S. dictyosporum; and clade D corresponds to section Reticulatae and includes S. bovista, S. citrinum s.l., and S. meridionale. The presence of S. capeverdeanum s.l. and S. venenatum is reported for the first time in Mexico.
Keywords: Environmental DNA; Soil; Macromycetes; Biogeography
Resumen
El orden Boletales constituye uno de los grupos de macromicetos más diversos y ampliamente distribuidos, con el género Scleroderma como un representante significativo. A pesar de su relevancia, los estudios sobre su sistemática y diversidad en México son escasos. En este trabajo analizamos la diversidad y distribución de Scleroderma en México a partir de toda la información disponible (registros de especímenes, DNA de esporomas y DNA ambiental). Se evaluaron 120 secuencias de DNA nrITS, de las cuales 27 corresponden a muestras ambientales; las restantes se obtuvieron de bases de datos públicas. Mientras que por taxonomía morfológica se han citado 15 especies de Scleroderma en el país, nuestros resultados sugieren aproximadamente 24 especies. Los análisis filogenéticos revelaron 4 clados principales: el clado A corresponde a la sección Scleroderma e incluye a S. albidum s.l., S. areolatum y S. cepa/laeve; el clado B incluye las especies de la sección Sclerangium y a S. anomalosporum; el clado C integra únicamente a S. dictyosporum; y el D corresponde a la sección Reticulatae, e incluye a S. bovista, S. citrinum s.l. y S. meridionale. Asimismo, se reporta por primera vez en México la presencia de S. capeverdeanum y S. venenatum.
Palabras clave: DNA ambiental; Suelo; Macromicetos; Biogeografía
Introduction
The estimation of the global diversity of fungi is still under debate, conservative authors propose a range between 1.5 and 3.8 million species (Hawksworth, 1991; Hawksworth & Lücking, 2017). However, current knowledge about fungal diversity is still insufficient, as many of the species have not been described or detected through their sporomes (Tedersoo et al., 2014). Furthermore, in Mexico the complex topography, strong habitat variability, and convergence of Nearctic and Neotropical biota contribute to a high fungal diversity characterized by many cryptic lineages (Cifuentes-Blanco et al., 1997; Sánchez-Ramírez, 2015). Consequently, morphological traits of fungi are insufficient in many taxa to detect hidden diversity.
In recent decades, tools such as metabarcoding have been used to improve species detection, particularly from environmental samples such as air, water, and soil (Heeger et al., 2018; Korpelainen et al., 2017; Tedersoo et al., 2014, 2022). Regarding macromycetes, these techniques have enhanced our understanding of the diversity of genera with medical, edible, economic, or cultural importance, such as Suillus and Tuber (Bonito et al., 2010; Tao et al., 2024) and have clarified ecological relationships between Boletus and the soil microbiota (Santolamazza-Carbone et al., 2023). However, the diversity of other groups such as Scleroderma remains underestimated.
The genus Scleroderma described by Persoon (1801) belongs to the family Sclerodermataceae within the Boletales (Binder & Bresinsky, 2002; Kirk et al., 2008). Species in this genus develop ectomycorrhizal associations with temperate and tropical plants such as Coccoloba, Eucalyptus, Fagus, Pinus, and Quercus, among others (Guzmán et al., 2013; Lu et al., 1998; Rincón et al., 2001; Sims et al., 1995). It has been estimated that the Sclerodermatineae originated 66 million years ago, at the end of the Cretaceous, while its main genera diversified around the middle Cenozoic. Consequently, Scleroderma is presumed to have originated approximately 38.37 million years ago, at the end of the Eocene (Wilson et al., 2012).
Several species of Scleroderma are considered edible, toxic, or medicinal. For instance, S. citrinum, S. flavidum,and S. yunnanense stand out as edible mushrooms in China (Wang et al., 2004; Zhang et al., 2013), while S. citrinum, S. dictyosporum,and S. polyrhizum are consumed in India (Karun et al., 2022). The edibility of S. laeve and S. texense has been documented for Mexico (Cortés-Pérez et al., 2021; De Ávila et al., 1980). Moreover, toxicity assays using Artemia franciscana larvae confirmed that Mexican specimens of S. texense do not exhibit toxic effects, supporting their local use as edible fungi (Ruiz-González et al., 2017). On the other hand, S. areolatum, S. bovista, S. cepa, S. citrinum, S. flavidum, S. polyrhizum,and S. verrucosum are considered medicinal in China (Dai & Yang, 2008). In Mexico, S. areolatum, S. nitidum, and S. verrucosum are used to stop bleeding from wounds (Guzmán et al., 2013). Conversely, some authors have classified species like S. albidum, S. areolatum, S. cepa, and S. venenatum as toxic (Guzmán, 1970; Sato et al., 2020; Yao et al., 2023; Zhang et al., 2020; Zhong et al., 2021).
In soil restoration projects, Scleroderma plays a significant role in maintaining and establishing ectomycorrhizal hosts. Its species improve the establishment, growth, and functional traits of their hosts (Bullaín-Galardis et al., 2024). For this reason, species such as S. citrinum, S. polyrhizum, S. texense, and S. verrucosum have been used to inoculate native trees during revegetation efforts in abandoned agricultural lands or arid soils (Pera & Parladé, 2005; Valdés-Ramírez et al., 2010). Even tropical species such as S. bermudense has been inoculated in Coccoloba uvifera to alleviate salinity stress and restore coastal dunes (Bandou et al., 2006; Bullaín-Galardis et al., 2024).
From a morphological perspective, Scleroderma was divided into 3 sections by Guzmán (1967, 1970) based on microscopic diagnostic characters such as basidiospore ornamentation and the structure of generative hyphae: section Aculeatispora characterized by the presence of echinulate basidiospores and the absence of clamp connections; section Sclerangium with subreticulate, subechinulate or echinulate basidiospores and abundant clamp connections; finally, section Scleroderma with reticulate basidiospores and few clamp connections. However, a change in the name of the sections Aculeatispora and Scleroderma to sections Scleroderma and Reticulatae, respectively, was later proposed (Guzmán et al., 2013). Some molecular studies partially agreed with this classification and reported new species based on some DNA regions such as nrITS (Nouhra et al., 2012; Phosri et al., 2009; Rusevska et al., 2014; Wu et al., 2023).
Scleroderma has around 100 species names registered in Index Fungorum (https://www.indexfungorum.org) and MycoBank (https://www.mycobank.org/). In Scleroderma, species complexes are not exclusively composed of cryptic taxa, while some species are morphologically indistinguishable and can only be separated using molecular data, many others can be reliably distinguished based on macroscopic and microscopic characters. However, some species such as S. meridionale, S. polyrhizum, and S. septentrionale represent species complexes that are being delimited through phylogenetic studies (Ortíz-Rivero et al., 2021). This highlights the importance of integrating morphological and molecular evidence to achieve accurate species delimitation within the genus.
Although phylogenetic and molecular studies for the genus exist worldwide (Nouhra et al., 2012; Phosri et al., 2009; Rusevska et al., 2014; Yang et al., 2025), only morphological studies have been carried out in Mexico, where 15 morphospecies have been reported: S. albidum, S. areolatum, S. bermudense, S. bovista, S. cepa, S. citrinum, S. hypogaeum, S. mexicana, S. michiganense, S. nitidum, S. polyrhizum, S. pseudostipitatum, S. texense, and S. verrucosum (Guzmán, 1970; Guzmán et al., 2013; Guzmán-Dávalos & Guzmán, 1985). Additionally, S. guzmanii was described by Ortíz-Rivero et al. (2021), and S. cepa s.l. was recently reported through phylogenetic analysis associated with Carya illinoinensis (Sánchez-Ledesma et al., 2023).
Due to the uncertainty regarding the diversity of Scleroderma species in Mexico, we conducted a comprehensive study integrating all genetic and geographic information available for the genus in the country. This approach is key to obtaining a more complete view of Scleroderma diversity in Mexico.
Materials and methods
Soil environmental DNA (eDNA) sampling was carried out in the northeast, center, south, and southeast of Mexico (Fig. 1C) from 2019 to 2021, as part of the Global Soil Mycobiome Consortium Project (Tedersoo et al., 2021). A total of 73 locations in the states of Aguascalientes, Campeche, Chiapas, Coahuila, Hidalgo, Jalisco, Mexico City, Michoacán, Morelos, Nuevo León, Oaxaca, Puebla, Quintana Roo, Estado de Mexico, Tabasco, Tlaxcala, Veracruz, and Yucatán, were sampled. In each locality, we collected 40 soil cores around an area of 2,500 m2. Each soil core was retrieved with a polyvinyl tube (5 cm diameter and 10 cm depth) to take the organic horizon and the first part of the mineral horizon, avoiding litter. Afterwards, these were combined and homogenized to form a composite sample (~ 1 kg). Composite samples were placed in Ziploc bags (~ 250 g), dried and stored at 5 °C.
DNA extraction was performed as described in Tedersoo et al. (2021). Polymerase chain reactions (PCR) were performed to amplify the internal transcribed spacer region of the nuclear ribosomal DNA (nrITS) with the universal eukaryotic primers ITS9mun and ITS4ngsUni, according to the methods and conditions in Tedersoo and Anslan (2019). PCR libraries were constructed and sequenced in the PacBio Sequel II platform at the University of Oslo. The resulting sequences were demultiplexed, quality filtered chimeric sequences were removed, OTUs clustered at 98% similarity, and taxonomically annotated as described in Tedersoo et al. (2021). The most abundant sequence of each OTU was selected as its representative sequence and used for further analysis.
The resulting Scleroderma DNA sequences from soil were complemented with sequences retrieved from GenBank (Sayers et al., 2024) derived from voucher specimens and ectomycorrhizae. Our final dataset contained 120 nrITS sequences of which 27 correspond to eDNA and voucher specimens and 93 were retrieved from GenBank (Table 1). We used 2 sequences of Tremellogaster surinamensis (Wilson et al., 2011) as an outgroup to root the Scleroderma phylogeny. We aligned the DNA sequences with MAFFT with the FFT-NS-i algorithm with 1,000 repetitions implemented in Geneious Prime. The resulting alignment was manually edited and trimmed. The final alignment had 768 sites, including gaps.
The best nucleotide substitution model for our alignment (HKY + I + Gama) was selected based on the corrected Akaike information criterion (AICc; Burnham & Anderson, 2002) inferred in jModelTest (Darriba et al., 2012). We inferred phylogenetic relationships based on Bayesian and Maximum likelihood approaches. Both analyses were performed in MrBayes 6.1 and PHYML implemented in Geneious Prime. Maximum likelihood support was estimated with 1,000 bootstrap pseudo-replicates (MLB) (Felsenstein, 1985), leaving the rest of parameters as default. For the Bayesian inference, we ran 4 simultaneous Monte Carlo Markov Chains (MCMC) for 10 M generations, sampling every 1,000 trees.
Table 1
nrITS DNA sequences obtained from environmental soil samples and GenBank used for phylogenetic analysis.
| Species | GenBank access code | Country | Reference |
| Scleroderma albidum | PP582969 | India | Unpublished** |
| Scleroderma albidum | OR656734 | Spain | Unpublished** |
| Scleroderma albidum | KJ676532 | Brazil | Montagner et al., 2015 |
| Scleroderma albidum s.l.* | PV232154 | Mexico | This study |
| Scleroderma albidum s.l.* | PV232155 | Mexico | This study |
| Scleroderma albidum s.l.* | PV232156 | Mexico | This study |
| Scleroderma anomalosporum Type | NR147662 | Brazil | Baseia et al., 2016 |
| Scleroderma areolatum | FM213352 | USA | Phosri et al., 2009 |
| Scleroderma areolatum | EU784407 | Brock et al., 2009 | |
| Scleroderma areolatum | HF933231 | Macedonia | Rusevska et al., 2014 |
| Scleroderma areolatum | KC152224 | Mexico | Unpublished** |
| Scleroderma areolatum | KY574379 | Mexico | Unpublished** |
| Scleroderma areolatum | PV211138 | Mexico | Unpublished** |
| Scleroderma areolatum* | PV232141 | Mexico | This study |
| Scleroderma bermudense* | PV232132 | Mexico | This study |
| Scleroderma bermudense* | PV232133 | Mexico | This study |
| Scleroderma bermudense* | PV232142 | Mexico | This study |
| Scleroderma bermudense* | PV232143 | Mexico | This study |
| Scleroderma bermudense* | PV232144 | Mexico | This study |
| Scleroderma bermudense | OQ351731 | Cuba | Unpublished** |
| Scleroderma bermudense | OQ351725 | Cuba | Unpublished** |
| Scleroderma bermudense | OQ351721 | Cuba | Unpublished** |
| Scleroderma bermudense | KJ209672 | Senegal | Unpublished** |
| Scleroderma bermudense | KJ209674 | France | Unpublished** |
| Scleroderma aff. bermudense sp. 8* | PV232136 | Mexico | This study |
| Scleroderma aff. bermudense sp. 8* | PV232137 | Mexico | This study |
| Scleroderma aff. bermudense sp. 8* | PV232138 | Mexico | This study |
| Scleroderma aff. bermudense sp. 9* | PV232134 | Mexico | This study |
| Scleroderma aff. bermudense sp. 10* | PV232135 | Mexico | This study |
| Table 1. Continued | |||
| Species | GenBank access code | Country | Reference |
| Scleroderma bovista | KU739419 | Spain | Fernández-Miranda, 2014 |
| Scleroderma bovista | FM213340 | Spain | Phosri et al., 2009 |
| Scleroderma bovista | HF933235 | Macedonia | Rusevska et al., 2014 |
| Scleroderma bovista | HF933242 | Serbia | Rusevska et al., 2014 |
| Scleroderma cf. bovista | FM213339 | USA | Phosri et al., 2009 |
| Scleroderma camassuense Type | NR147663 | Brazil | Baseia et al., 2016 |
| Scleroderma capeverdeanum Type | NR164545 | Cape verde | Crous et al., 2016 |
| Scleroderma capeverdeanum | KU747111 | Cape verde | Crous et al., 2016 |
| Scleroderma capeverdeanum s.l.* | PV232145 | Mexico | This study |
| Scleroderma capeverdeanum s.l.* | PV232157 | Mexico | This study |
| Scleroderma cepa | OR882638 | USA | Unpublished** |
| Scleroderma cepa | EU784411 | Brock et al., 2009 | |
| Scleroderma cepa | OP743680 | USA | Unpublished** |
| Scleroderma cepa s.l. | MZ092921 | Mexico | Sánchez-Ledesma et al., 2023 |
| Scleroderma citrinum | FM213344 | USA | Phosri et al., 2009 |
| Scleroderma citrinum | KJ679575 | Brazil | Montagner et al., 2015 |
| Scleroderma citrinum | KJ679576 | Brazil | Montagner et al., 2015 |
| Scleroderma aff. citrinum* | PV232139 | Mexico | This study |
| Scleroderma columnare | AB459519 | Thailand | Ruankaew-Disyatat et al., 2016 |
| Scleroderma columnare | AB459512 | Thailand | Ruankaew-Disyatat et al., 2016 |
| Scleroderma dictyosporum | FJ840444 | Burkina Faso | Sanon et al., 2009 |
| Scleroderma dictyosporum | FJ840442 | Burkina Faso | Sanon et al., 2009 |
| Scleroderma dictyosporum | FJ840443 | Burkina Faso | Sanon et al., 2009 |
| Scleroderma dictyosporum | FJ840449 | Burkina Faso | Sanon et al., 2009 |
| Scleroderma duckei | KX792086 | Brazil | Baseia et al., 2016 |
| Scleroderma duckei Type | NR147664 | Brazil | Baseia et al., 2016 |
| Scleroderma dunensis Type | NR147646 | Brazil | Crous et al., 2016 |
| Scleroderma dunensis | KU747112 | Brazil | Crous et al., 2016 |
| Scleroderma dunensis | KU747114 | Brazil | Crous et al., 2016 |
| Scleroderma erubescens Type | OQ554978 | China | Wu et al., 2023 |
| Scleroderma erubescens | OQ554976 | China | Wu et al., 2023 |
| Scleroderma geaster | MT270642 | Spain | Ortiz-Rivero et al., 2021 |
| Scleroderma geaster | MT270640 | Portugal | Ortiz-Rivero et al., 2021 |
| Scleroderma guzmanii | MT270647 | Mexico | Ortiz-Rivero et al., 2021 |
| Scleroderma guzmanii Type | NR176725 | USA | Ortiz-Rivero et al., 2021 |
| Scleroderma furfuraceum Type | NR186993 | Russia | Rebriev & Zvyagina, 2022 |
| Scleroderma laeve | EU718117 | Wilson et al., 2011 | |
| Scleroderma laeve | ON212388 | South Korea | Cho et al., 2022 |
| Scleroderma meridionale | HF933238 | Macedonia | Rusevska et al., 2014 |
| Table 1. Continued | |||
| Species | GenBank access code | Country | Reference |
| Scleroderma meridionale | MG367369 | Italy | Leonardi et al., 2018 |
| Scleroderma meridionale | MG264160 | Italy | Leonardi et al., 2018 |
| Scleroderma meridionale | EU718121 | USA | Unpublished** |
| Scleroderma michiganense | FM213348 | USA | Phosri et al., 2009 |
| Scleroderma michiganense | FM213347 | USA | Phosri et al., 2009 |
| Scleroderma michiganense | FM213346 | USA | Phosri et al., 2009 |
| Scleroderma nastii | ON212391 | South Korea | Cho et al., 2022 |
| Scleroderma nitidum | KU759906 | Brazil | Baseia et al., 2016 |
| Scleroderma nitidum | KU759908 | Brazil | Baseia et al., 2016 |
| Scleroderma patagonicum | KY462453 | Argentina | Truong et al., 2017 |
| Scleroderma patagonicum | HQ688789 | Argentina | Noura et al., 2012 |
| Scleroderma polyrhizum | MT270661 | USA | Ortiz-Rivero et al., 2021 |
| Scleroderma polyrhizum | MT270662 | Spain | Ortiz-Rivero et al., 2021 |
| Scleroderma separatum | OQ554975 | China | Wu et al., 2023 |
| Scleroderma separatum | OQ554973 | China | Wu et al., 2023 |
| Scleroderma septentrionale | FM213338 | USA | Phosri et al., 2009 |
| Scleroderma septentrionale | FM213336 | Sweden | Phosri et al., 2009 |
| Scleroderma cf. septentrionale | FM213337 | USA | Phosri et al., 2009 |
| Scleroderma sinnamariense | FM213358 | Thailand | Phosri et al., 2009 |
| Scleroderma sinnamariense | AB908177 | India | Unpublished** |
| Scleroderma sinnamariense | HQ687222 | Thailand | Unpublished** |
| Scleroderma squamulosum | OQ554980 | China | Wu et al., 2023 |
| Scleroderma squamulosum Type | OQ554979 | China | Wu et al., 2023 |
| Scleroderma suthepense Type | NR132871 | Thailand | Kumla et al., 2013 |
| Scleroderma texense | MT270650 | USA | Ortiz-Rivero et al., 2021 |
| Scleroderma texense | MT270649 | USA | Ortiz-Rivero et al., 2021 |
| Scleroderma texense* | PV232140 | Mexico | This study |
| Scleroderma venenatum | OQ750238 | China | Unpublished** |
| Scleroderma venenatum | JF273540 | China | Zhang et al., 2020 |
| Scleroderma venenatum | KC52225 | Mexico | Unpublished** |
| Scleroderma venenatum | MH513631 | China | Zhang et al., 2020 |
| Scleroderma venenatum | OM874611 | Russia | Rebriev & Zvyagina, 2022 |
| Scleroderma venenatum | OM874613 | Russia | Rebriev & Zvyagina, 2022 |
| Scleroderma venenatum | MH513632 | China | Zhang et al., 2020 |
| Scleroderma verrucosum | EU784415 | England | Brock et al., 2009 |
| Scleroderma verrucosum | HF933241 | Macedonia | Rusevska et al., 2014 |
| Scleroderma vinaceum | OQ554986 | China | Wu et al., 2023 |
| Scleroderma vinaceum Type | OQ554987 | China | Wu et al., 2023 |
| Scleroderma yunnanense | JQ639046 | China | Wu et al., 2023 |
| Table 1. Continued | |||
| Species | GenBank access code | Country | Reference |
| Scleroderma yunnanense | JQ639045 | China | Wu et al., 2023 |
| Scleroderma yunnanense | JQ639041 | China | Wu et al., 2023 |
| Scleroderma sp. 1* | PV232153 | Mexico | This study |
| Scleroderma sp. 2 | PV211140 | Mexico | Unpublished ** |
| Scleroderma sp. 3* | PV232158 | Mexico | This study |
| Scleroderma sp. 4* | PV232149 | Mexico | This study |
| Scleroderma sp. 4* | PV232150 | Mexico | This study |
| Scleroderma sp. 4* | PV232151 | Mexico | This study |
| Scleroderma sp. 4* | PV232152 | Mexico | This study |
| Scleroderma sp. 5* | PV232146 | Mexico | This study |
| Scleroderma sp. 6* | PV232147 | Mexico | This study |
| Scleroderma sp. 7* | PV232148 | Mexico | This study |
| Tremellogaster surinamensis | KT724170 | Colombia | Unpublished ** |
| Tremellogaster surinamensis | EU718127 | Wilson et al., 2011 |
* Sequences obtained from environmental soil samples. ** Unpublished GenBank sequences.
We visually assessed sampling stationarity in Tracer ver. 1.7 (Rambaut et al., 2018) and by the observed average standard deviation of split frequencies (< 0.01). Finally, we discarded 20% of the sampled trees as burn-in, resulting in a collection of 7,510 trees. A majority rule consensus tree and Bayesian posterior probabilities (BPP) were calculated with the post-burn-in trees. We visually compared topological congruence between replicates and inference methods and edited the final tree in FigTree ver 1.4.4 (Rambaut, 2018).
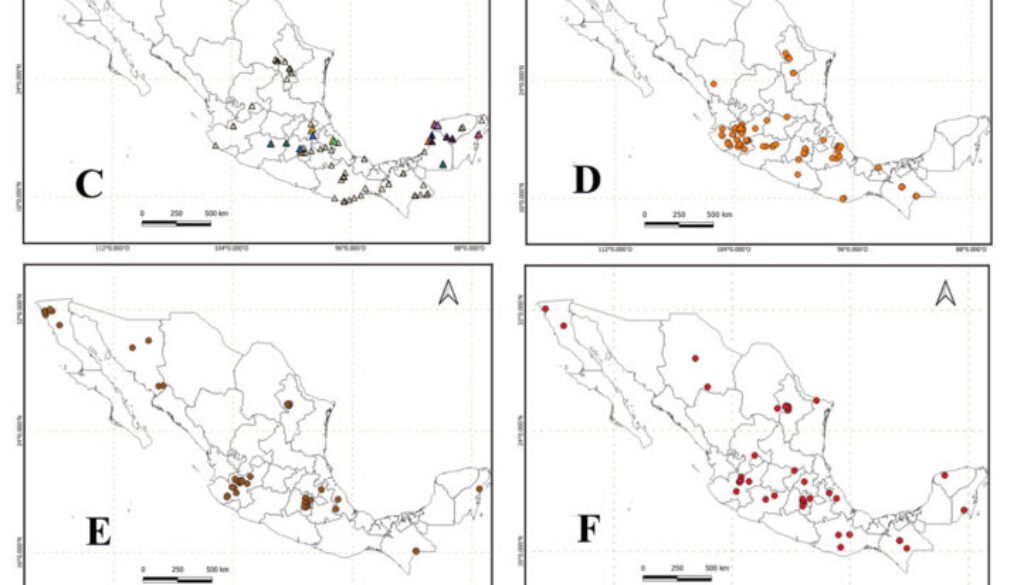
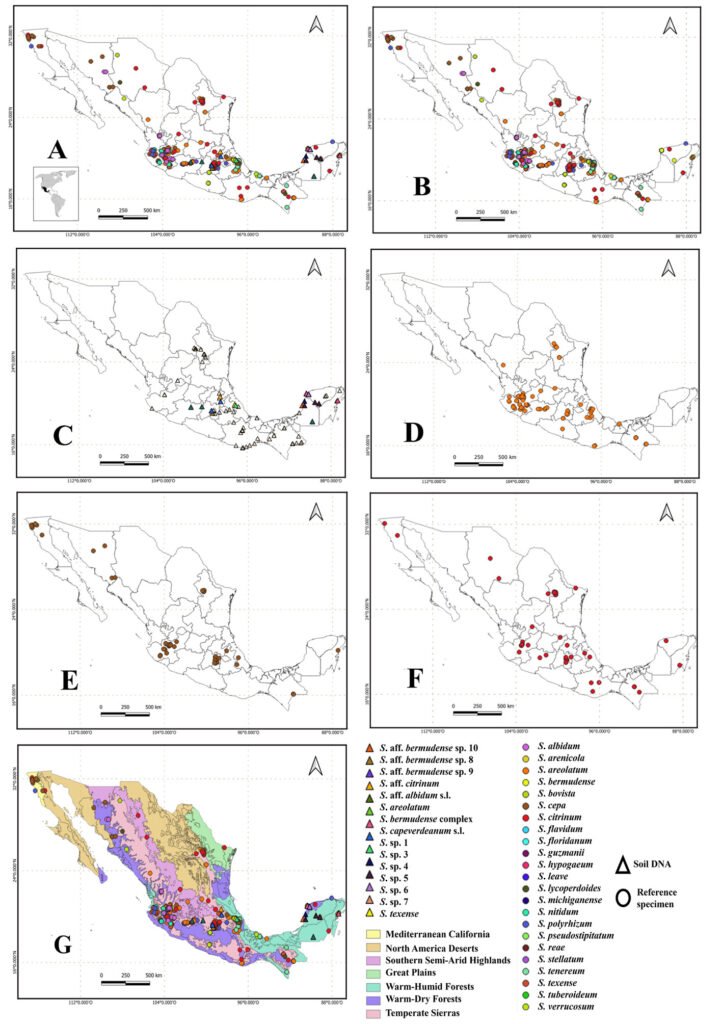
To determine the distribution and species richness of Scleroderma in Mexico, we used the occurrence of the clades in the 73 sampling sites (Fig. 1C). This information was complemented with the 2,349 records of Scleroderma for Mexico in the GBIF public database (GBIF.org, August 27, 2024) (Fig. 1B). Records at the genus level were excluded. Since the political division of the country is not suitable for understanding patterns and processes of biodiversity, a distribution map of the genus was created using the level IV ecoregions of Mexico (Conabio, 2008).
Results
The consensus tree shows that Scleroderma is divided into 4 main clades (A, B, C, and D) with medium to high MLB and BPP support values. The topology of the inferred phylogenetic trees was similar in both analyses. The Mexican species were distributed in clades A, B, and D (Fig. 2). Morphologically, 15 species reported in the literature are recognized for Mexico, 9 from GBIF records only. Scleroderma capeverdeanum s.l.and S. venenatum complex are recorded for the first time along with several unnamed clades from soil samples. Of the total number of species, the clades corresponding to S. areolatum, the S. bermudense complex and the S. capeverdeanum s.l. are recognized from specimen vouchers and eDNA sampling, which leads us to consider that the number of species for Mexico is around 24.
Clade A (MLB: 92% and BPP: 1) contains species such as S. areolatum, S. cepa/laeve, and S. verrucosum. Several species from soil samples are also grouped together. Among the species sequenced from specimen vouchers, mycorrhizae, and eDNA from Mexico is S. areolatum. The grouping of Mexican sequences within the S. capeverdeanum s.l. clade (MLB: 75% and BPP:0.70) stands out, as well as sequences within S. venenatum (MLB: 100% and BPP: 1). Clade B (MLB: 62%, BPP: 0.99) includes species such as S. guzmanii and S. polyrhizum. Its most diverse subclade comprises the S. bermudense complex (MLB: 56%, BPP: 0.8), which is sister to S. aff. bermudense (MLB: 80%, BPP: 0.99), along with numerous soil DNA sequences associated with these 2 taxa. Clade C (MLB: 100% and BPP: 1) only contains S. dictyosporum, a species distributed in Africa and Southeast Asia. Clade D (MLB: 81%, BPP: 1) includes the Mexican taxon S. aff. citrinum from soil DNA, which is sister to S. citrinum s.l.
Species of Scleroderma occur throughout Mexico (Fig. 1A, Table 2); however, there are no records for the states of Baja California Sur and Zacatecas (Fig. 1B). The records obtained from soil eDNA are mainly concentrated in the central states of the country such as Estado de México, Hidalgo, and Michoacán; as well as the Yucatán Peninsula (Fig. 1C).
Regarding the clades obtained by eDNA, we found that they are distributed in 3 main vegetation types that correspond to tropical broadleaf forest, subtropical broadleaf forest, and tropical coniferous forest (Table 3). The clades of S. aff. bermudense and S. bermudense complex are mainly distributed in tropical broadleaf forest and subtropical broadleaf forest, while S. capeverdeanum distributes in tropical coniferous forest and subtropical coniferous forest. In the case of S. areolatum, it was only sequenced tropical broadleaf forests soil. The Transmexican Volcanic Belt is the physiographic province with the highest diversity of Scleroderma, being S. areolatum, S. cepa, and S. citrinum the most common species (Fig. 1D, E, F). Meanwhile, the largest number of records corresponds to S. bermudense complex from the Yucatán Peninsula.
There are clades of Neotropical distribution that include basidiomes and eDNA sequences such as those found within the S. bermudense complex; both, its basidiomes and soil DNA come mainly from the Yucatán Peninsula. Some OTUs of S. bermudense complex were found abundantly in the soil, such as S. aff. bermudense sp. 8 with 3,877 sequences. However, S. aff. bermudense sp. 9 and S. aff. bermudense sp. 10, have small distribution areas, apparently restricted to Mexico (Table 2).
Regarding the distribution of Scleroderma in Mexican ecoregions, there are more reports in the temperate sierras and less in the warm-dry forests. The ecoregions corresponding to the great deserts of Mexico remain among the least explored (Fig. 1G). In the southeast there are more records of S. bermudense found in the warm-humid forests of the Yucatán Peninsula, and in smaller numbers in warm-humid forests of Chiapas, Tabasco, and Veracruz.
Discussion
Our phylogenetic analysis matches partially with the results obtained in former molecular studies reporting that Scleroderma is divided into 2 main clades (Phosri et al., 2009; Rusevska et al., 2014). However, it is more consistent with current phylogenies including a larger number of species such as those of Wu et al. (2023) and Yang et al. (2025), who also found 4 main clades. It also does not fully correspond with the sections proposed by Guzmán (1967, 1970).
Our phylogeny and that of Yang et al. (2025) are similar across all major clades; even while our study includes only ITS sequences. Clade A corresponds to section Scleroderma and includes S. albidum s.l., S. areolatum, and S. cepa/laeve. Guzmán (2013) proposed S. laeve as a synonym of S. albidum; however, our results suggest that, instead, it may be a synonym of S. cepa. This section also comprises S. erubescens, S. separatum, S. squamulosum, and S. vinaceum, which cluster with species assigned to section Scleroderma, traditionally characterized by echinulate basidiospores and the absence of clamp connections. Nevertheless, Wu et al. (2023) reported that both S. erubescens and S. separatum possess clamp connections. This suggests that section Scleroderma may not be exclusively composed of species without clamp connections.
Clade B includes species in Section Sclerangium and S. anomalosporum, a species with smooth basidiospores (Baseia et al., 2016). Clade C remains the same, including only S. dictyosporum. This species was formerly placed in section Scleroderma, now recognized as section Reticulatae; however, in our analysis it forms an independent clade. Clade D corresponds to section Reticulatae.
Similarly, Yang et al. (2025) concluded that basidiospore ornamentation is not a reliable character for distinguishing section Scleroderma from section Sclerangium. Many species in section Scleroderma may also produce subreticulate basidiospores (e.g., S. areolatum, S. cepa, and S. nastii), whereas several new species in section Sclerangium can produce echinulate basidiospores (e.g., S. navigatum, S. xanthochroum, and S. zengchengense). The authors further proposed that peridium thickness may serve as an additional distinguishing feature.
Our results suggest that the number of Scleroderma species in Mexico is greater than the 15 previously reported (Guzmán et al., 2013; Ortíz-Rivero et al., 2021; Sánchez-Ledesma et al., 2023). We reported for the first time S. capeverdeanum s.l.and S. venenatum, as well as 10 clades that do not match with species previously sequenced. Conservatively the diversity of Scleroderma for Mexico is around 24 taxa. The discrepancy between species richness in voucher specimens versus DNA sequences may be explained due to scarce formation of basidiomes or because most of the described species have not been sequenced (Tedersoo et al., 2014).
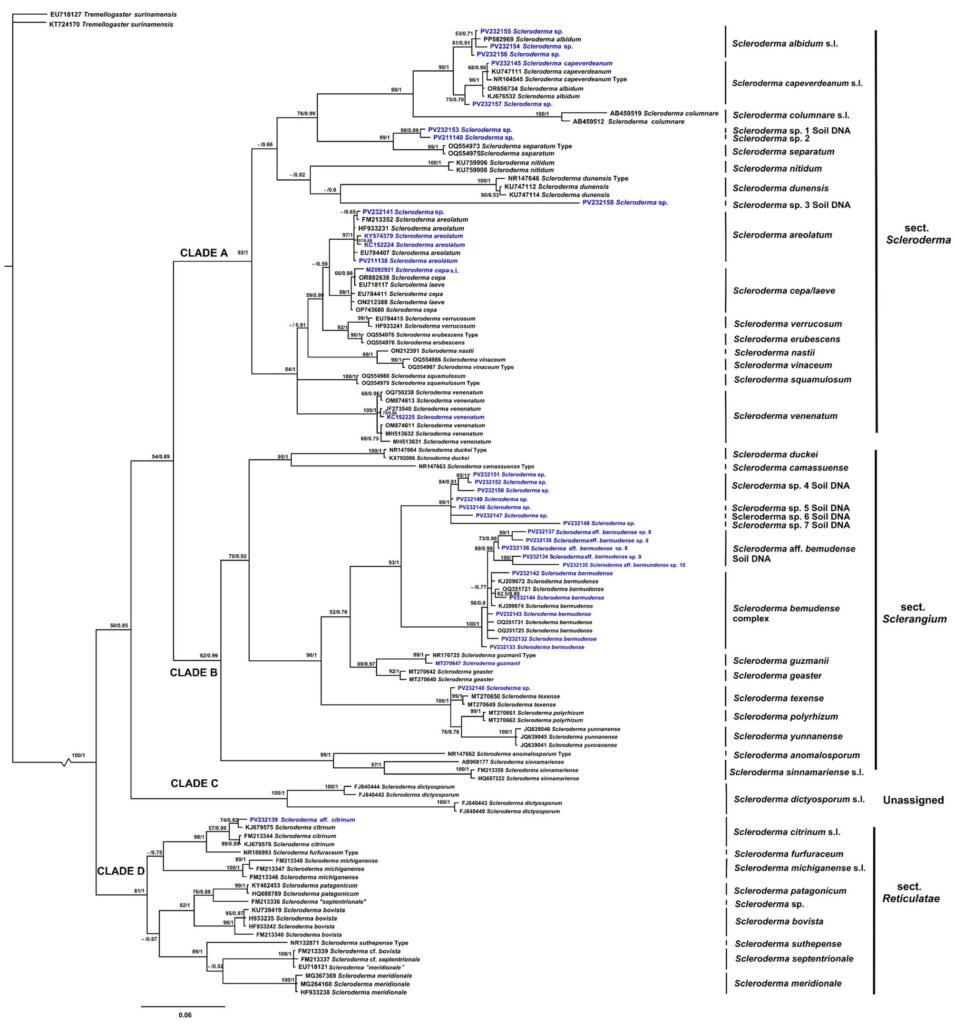
Table 2
Distribution and abundance of Scleroderma species obtained from soil samples.
| Species | Distribution | Abundance (Number of sequences) | Number of sites in Mexico |
| Scleroderma albidum s.l. | Estado de México | 5 | 1 |
| Scleroderma areolatum | Michoacán | 5 | 1 |
| Scleroderma bermudense | Quintana Roo and Yucatán | 164 | 2 |
| Scleroderma aff. bermudense sp. 8 | Morelos, Estado de México, Quintana Roo, and Yucatán | 3,877 | 5 |
| Scleroderma aff. bermudense sp. 9 | Yucatán | 1 | 1 |
| Scleroderma aff. bermudense sp. 10 | Yucatán | 10 | 1 |
| Scleroderma capeverdeanum s.l. | Hidalgo and Estado de México | 74 | 2 |
| Scleroderma aff. citrinum | Hidalgo | 1 | 1 |
| Scleroderma texense | Veracruz | 35 | 2 |
| Scleroderma sp. 1 | Veracruz | 3 | 1 |
| Scleroderma sp. 3 | Campeche and Quintana Roo | 3 | 2 |
| Scleroderma sp. 4 | Campeche, Michoacán and Yucatán | 33 | 8 |
| Scleroderma sp. 5 | Campeche and Yucatán | 33 | 5 |
| Scleroderma sp. 6 | Yucatán | 1 | 1 |
| Scleroderma sp. 7 | Campeche and Yucatán | 5 | 3 |
Table 3
Distribution of Scleroderma species in different types of vegetation.
| Species | Type of vegetation |
| Scleroderma albidum s.l. | Tropical coniferous forest |
| Scleroderma areolatum | Tropical broadleaf forest |
| Scleroderma bermudense | Subtropical broadleaf forest |
| Scleroderma aff. bermudense sp. 8 | Tropical broadleaf forest, subtropical broadleaf forest and tropical coniferous forest |
| Scleroderma aff. bermudense sp. 9 | Subtropical broadleaf forest |
| Scleroderma aff. bermudense sp. 10 | Subtropical broadleaf forest |
| Scleroderma capeverdeanum s.l. | Tropical coniferous forest and subtropical coniferous forest |
| Scleroderma aff. citrinum | Subtropical broadleaf forest |
| Scleroderma texense | Tropical coniferous forest |
| Scleroderma sp. 1 | Tropical coniferous forest |
| Scleroderma sp. 3 | Tropical broadleaf forest and subtropical broadleaf forest |
| Scleroderma sp. 4 | Tropical broadleaf forest, subtropical broadleaf forest, and tropical coniferous forest |
| Scleroderma sp. 5 | Tropical broadleaf forest and subtropical broadleaf forest |
| Scleroderma sp. 6 | Subtropical broadleaf forest |
| Scleroderma sp. 7 | Tropical broadleaf forest and subtropical broadleaf forest |
In the case of some OTUs of the S. bermudense complex, they could be considered endemic to Mexico, as they were only recorded at 1 sampling site. Other studies have reported that S. bermudense represents one of the most abundant endemic taxa in the Caribbean, mainly associated with Coccoloba uvifera (Bâ et al., 2024; Põlme et al., 2017). This association is mediated by the environmental stress of the coastal dunes where they develop, which causes a high specificity of the symbiosis (Bâ et al., 2024; Põlme et al., 2017). We found several clades of S. bermudense s.l.with high genetic differentiation not associated with Coccoloba species. Thus, there is a species complex associated with S. bermudense with an unsolved taxonomy and ecology in the Yucatán Peninsula.
Species such as S. areolatum and S. cepa have been recorded throughout the country through their basidiomes (Guzmán & Herrera, 1973; Guzmán et al., 2013). Scleroderma areolatum was reported from Durango, Guerrero, Hidalgo, Jalisco, México City, Michoacán, Nuevo León, Oaxaca, Puebla, and Veracruz (Guzmán & Herrera, 1973). Through ITS analysis it has been confirmed that S. cepa s.l. is an ectomycorrhizal symbiont of pecan trees in Chihuahua and Coahuila and that it is possibly a species complex (Sánchez-Ledesma et al., 2023). However, the type specimens of both species have not been sequenced, so it is important to carry out their type studies given the importance they can have in agroecosystems. In fact, species complexes indicating cryptic lineages in fungi are common (Sánchez-Ramírez, 2015) and may include taxa that can be morphologically distinguished, as well as cryptic species that are only discernible through molecular data, as this study found for the S. albidum, S. bermudense, and S. cepa complexes.
We recorded for the first time S. venenatum in Mexico, a species considered toxic in China (Zhang et al., 2020). The presence of this species in the country is particularly relevant given that some Scleroderma species are traditionally consumed in Mexico. For example, the consumption of S. laeve has been reported in Morelos and S. texense in Oaxaca (Cortés-Pérez et al., 2021; De Ávila et al., 1980). In contrast, S. venenatum was recorded in Querétaro and can be distinguished from edible species by its sessile, brown to grayish brown basidiomes and well-developed rhizomorphs (Zhang et al., 2020). Regarding the identity of the species that have been reported as edible in Mexico, their identification was only carried out with morphology and given their importance in cultural contexts, it would be advisable to obtain molecular data from the basidiomes to corroborate their identity.
A higher diversity of Scleroderma in the Transmexican Volcanic Belt shows that, while for other organisms the system represents a barrier or a filter, fungi use it as a corridor (Gómez-Reyes et al., 2018). In this region, the complex topography, habitat variability, and the convergence between Nearctic and Neotropical biota contribute to a higher diversity of fungal species in general (Cifuentes-Blanco et al., 1997) and in particular for Scleroderma.
Acknowledgements
The first author thanks the Secretaría de Ciencia, Humanidades, Tecnología e Innovación (Secihti) for the scholarship granted (CVU 415474) and the Instituto de Ciencias Biológicas, Universidad Autónoma de Ciencias y Artes de Chiapas (UNICACH) for all the facilities provided. This article contains part of the results of the PhD thesis project for the Doctorado en Ciencias en Biodiversidad y Conservación de Ecosistemas Tropicales (UNICACH). This research was funded by UNAM-PAPIIT IV200223 grant awarded to RGO.
References
Bâ, A. M., Séne, S., Manokari, M., Galardis, M. B., Sylla, S. N., Selosse, M. A. et al. (2024). Coccoloba uvifera L. associated with Scleroderma bermudense Coker: a pantropical ectomycorrhizal symbiosis used in restoring of degraded coastal sand dunes. Mycorrhiza, 34, 375–389. https://doi.org/10.1007/s00572-024-01170-8
Bandou, E., Lebailly, F., Muller, F., Dulormne, M., Toribio, A., Chabrol, J. et al. (2006). The ectomycorrhizal fungus Scleroderma bermudense alleviates salt stress in seagrape (Coccoloba uvifera L.) seedlings. Mycorrhiza, 16, 559–565. https://doi.org/10.1007/s00572-006-0073-6
Baseia, I. G., Silva, B. D. B., Ishikawa, N. K., Soares, J. V. C., Franca, I. F., Ushijima, S. et al. (2016). Discovery or extinction of new Scleroderma species in Amazonia? Plos One, 11, 1–19. https://doi:10.1371/journal.pone.0167879
Binder, M., & Bresinsky, A. (2002). Derivation of a polymorphic lineage of Gasteromycetes from boletoid ancestors. Mycologia, 94, 85–98. https://doi.org/10.1080/15572536.2003.11833251
Bonito, G. M., Gryganskyi, A. P., Trappe, J. M., & Vilgalys, R. (2010). A global meta-analysis of Tuber ITS rDNA sequences: species diversity, host associations and long-distance dispersal. Molecular Ecology, 19, 4994–5008. https://doi.org/10.1111/j.1365294X.2010.04855.x
Brock, P. M., Doring, H., & Bidartondo, M. I. (2009). How to know unknown fungi: the role of a herbarium. New Phytologist, 181, 719–724. https://doi.org/10.1111/j.1469-8137.2008.02703.x
Bullaín-Galardis, M. M., López-Sánchez, R. C., Pruneau, L., Eichler-Lobermann, B., Fall, F., & Bâ, A. M. (2024). Using the ectomycorrhizal symbiosis between Coccoloba uvifera L. and Scleroderma bermudense Coker to restore a degraded coastal sand dune in Cuba. Trees, 38, 127–138. https://doi.org/10.1007/s00468-023-02470-w
Burnham, K., & Anderson, D. (2002). Model selection and multimodel inference: a practical information-theoretic approach. New York: Springer.
Cho, S., Kwag, Y., Han, S., Lee, D., & Kim, C. (2022). Two new records of Scleroderma species (Sclerodermataceae, Boletales) in South Korea. The Korean Journal of Mycology, 50, 115–123. https://doi.org/10.4489/KJM.20220011
Cifuentes-Blanco, J., Villegas-Ríos, M., Villarreal-Ordaz, J. L., & Sierra Galván, S. (1997). Diversity of macromycetes in pine-oak forests in the Neovolcanic Axis, Mexico. In M. E. Palm, & I. H. Chapela (Eds.), Mycology in sustainable development: expanding concepts, vanishing borders (pp. 111–121). Boone, North Carolina: Parkway Publishers, Inc.
Conabio (Comisión Nacional para el Conocimiento y Uso de la Biodiversidad). (2008). Ecorregiones terrestres de México. Escala 1:1000000. México. Digital cartographic dataset. Mexico. https://www.conabio.gob.mx/informacion/gis/
Cortés-Pérez, A., Pérez-Pacheco, C. K., Yescas-Arreola, E., & Ramírez-Cruz, V. (2021). Primer registro de Scleroderma texense (Boletales, Sclerodermatinae) como una especie comestible en la Sierra Sur de Oaxaca, México. Scientia Fungorum, 52, 1–8. https://doi.org/10.33885/sf.2021.52.1386
Crous, P. W., Wingfield, M. J., Richardson, D. M., Le Roux, J. J., Strasberg, D., Edwards, J. et al. (2016). Fungal planet description sheets. Persoonia, 36, 316–458. https://doi:10.3767/003158516X692185.
Dai, Y. C., & Yang, Z. L. (2008). A revised checklist of medicinal fungi in China. Mycosystema, 27, 801–824.
Darriba, D., Taboada, G. L., Doallo, R., & Posada, D. (2012). jModelTest 2: more models, new heuristics and parallel computing. Nature Methods, 9, 772. http://doi:0.1038/nmeth.2109
De Ávila, A. B., Welden, A. L., & Guzmán, G. (1980). Notes on the ethnomycology of Hueyapan, Morelos, México. Journal of Ethnopharmacology, 2, 311–321. https://doi.org/10.1016/S0378-8741(80)81013-0
Felsenstein, J. (1985). Confidence limits on phylogenies: an approach using the bootstrap. Evolution, 39, 783–791. http//doi:10.1111/j.1558-5646.1985.tb00420.x
Fernández-Miranda, C. E. (2014). Hongos ectomicorrícicos de plantas leñosas en terrenos degradados de Asturias por actividad minera e industrial (Ph.D. Thesis). Universidad de Oviedo. Oviedo, España.
GBIF.org (2024). 27 August 2024 GBIF Occurrence Download https://doi.org/10.15468/dl.bbmwbt
Gómez-Reyes, V. M., Vázquez-Marrufo, G., Ortega-Gómez, A. M., & Guevara-Guerrero, G. (2018). Ascomicetos hipogeos de la región occidental del Sistema Volcánico Transversal, México. Acta Botanica Mexicana, 125, 37–48. https://doi.org/10.21829/abm125.2018.1327
Guzmán, G. (1967). Taxonomía del género Scleroderma Pers. emend. Fr. Ciencia, 2, 195–208.
Guzmán, G. (1970). Monografía del género Scleroderma Pers. emend. Fr. (Fungi-Basidiomycetes). Darwiniana, 16, 233–407.
Guzmán, G., & Herrera, T. (1973). Especies de macromicetos citadas de México IV. Gasteromicetos. Boletín de la Sociedad Mexicana de Micología, 2, 105–119.
Guzmán, G., Cortés-Pérez, A., Guzmán-Dávalos, L., Ramírez-Guillén, F., & Sánchez-Jácome, M. R. (2013). An emendation of Scleroderma, new records, and review of the known species in Mexico. Revista Mexicana de Biodiversidad, 84, 173–191. https://doi.org/10.7550/rmb.31979
Guzmán-Dávalos, L., & Guzmán, G. (1985). Hongos del estado de Jalisco, V. El género Scleroderma. Revista Mexicana de Micología, 1, 109–128.
Hawksworth, D. L. (1991). The fungal dimension of biodiversity: magnitude, significance and conservation, Mycological Research, 95, 641–655
Hawksworth, D. L., & Lücking, R. (2017). Fungal diversity revisited: 2.2 to 3.8 million species. Microbiology Spectrum, 5, 10–1128. https://doi.org/10.1128/microbiolspec.funk-0052-20
Heeger, F., Bourne, E. C., Baschien, C., Yurkov, A., Bunk, B., Spröer, C. et al. (2018). Long-read DNA metabarcoding of ribosomal RNA in the analysis of fungi from aquatic environments. Molecular Ecology Resources, 18, 1500–1514. http://doi.org/10.1111/1755-0998.12937
Karun, N. C., Mahadevakumar, S., & Sridhar, K. R. (2022). On the Scleroderma in southwest India. Species, 23, 297–312.
Kirk, P. M., Cannon, P. F., Minter, D. W., & Stalpers, J. A. (2008). Ainsworth & Bisby’s Dictionary of the Fungi (10ª Ed.). Wallingford, UK: CAB International.
Korpelainen, H., & Pietiläinen, M. (2017). Diversity of indoor fungi as revealed by DNA metabarcoding. Genome, 60, 55–64. http//doi.org/10.1139/gen-2015-0191
Kumla, J., Suwannarach, N., Bussaban, B. & Lumyong, S. (2013). Scleroderma suthepense, a new ectomycorrhizal fungus from Thailand. Mycotaxon, 123, 1–7. http//doi.org/10.5248/123.1
Lu, X., Malajczuk, N., & Dell, B. (1998). Mycorrhiza formation and growth of Eucalyptus globulus seedlings inoculated with spores of various ectomycorrhizal fungi. Mycorrhiza, 8, 81–86. https://doi.org/10.1007/s005720050216
Montagner, D. F., Coelho, G., Silveira, A. O., Baldoni, D. B., & Antoniolli, Z. (2015). Morphological and molecular analyses in Scleroderma (Basidiomycota) associated with exotic forests in Pampa biome, southern Brazil. Mycosphere, 6, 337–344. https://doi.org/10.5943/mycosphere/6/3/9
Nouhra, E. R., Hernández, C. M. L., Pastor, N., & Crespo, E. M. (2012). The species of Scleroderma from Argentina, including a new species from the Nothofagus forest. Mycologia, 104, 488–495. https://doi.org/1 0.3852/11-082
Ortiz-Rivero, J., Watling, R., Guzmán-Dávalos, L., & Martín, M. P. (2021). The many-rooted earthball-Scleroderma geaster and S. polyrhizum revisited, with the description of a new species. Phytotaxa, 510, 1–17. https://doi.org/10.11646/phytotaxa.510.1.1
Pera, J., & Parladé, J. (2005). Inoculación controlada con hongos ectomicorrícicos en la producción de planta destinada a repoblaciones forestales: estado actual en España. Sistemas y Recursos Forestales, 14, 419–433.
Persoon, C. H. (1801). Synopsis methodica Fungorum. Göttingen: Henricus Dieterich.
Phosri, C., Martín, M. P., Watling, R., Jeppson, M., & Sihanonth, P. (2009). Molecular phylogeny and re-assessment of some Scleroderma spp. (Gasteromycetes). Anales del Jardín Botánico de Madrid, 66, 83–91.
Põlme, S., Bahram, M., Kõljalg, U., & Tedersoo, L. (2017). Biogeography and specificity of ectomycorrhizal Fungi of Coccoloba uvifera. In L. Tedersoo (Ed.), Biogeography of mycorrhizal symbiosis. Ecological studies (pp 345–360). Cham, Switzerland: Springer.345https://doi.org/10.1007/978-3-319-56363-3_16
Rambaut, A. (2018). FigTree v1.4.4. Institute of Evolutionary Biology, University of Edinburgh. http://tree.bio.ed.ac.uk/software/figtree/
Rambaut, A., Drummond, A. J., Xie, D., Baele, G., & Suchard, M. A. (2018). Posterior summarization in Bayesian phylogenetics using tracer 1.7. Systematic Biology, 67, 901–904. https://doi.org/10.1093/sysbio/syy032
Rebriev, Y. A., & Zvyagina, E. A. (2022). Scleroderma furfuraceum (Boletales, Agaricomycetes) a new species from the Russian Far East. Phytotaxa, 555, 169–177. https://doi.org/10.11646/phytotaxa.555.2.5
Rincón, A., Álvarez, I. F., & Pera, J. (2001). Inoculation of containerized Pinus pinea L. seedlings with seven ectomycorrhizal fungi. Mycorrhiza, 11, 265–271.
Ruankaew-Disyatat, N., Yomyart, S., Sihanonth, P., & Piapukiew, J. (2016). Community structure and dynamics of ectomycorrhizal fungi in a dipterocarp forest fragment and plantation in Thailand. Plant Ecology & Diversity, 9, 577–588. https://doi.org/10.1080/17550874.2016.1264018
Ruiz-González, L. E., Vázquez-Zea, J. A., Vega-Villasante, F., Guzmán-Davalos, L., & Guerrero-Galván, S. R. (2017). Evaluation of the toxicity of Basidiomycota fungi on the hatching of Artemia franciscana cysts. Revista Iberoamericana de Micologia, 34, 220–224. https://doi.org/10.1016/j.riam.2017.03.007
Rusevska, K., Karadelev, M., Phosri, C., Dueñas, M., Watling, R., & Martín, M. P. (2014). Rechecking of the genus Scleroderma (Gasteromycetes) from Macedonia using barcoding approach. Turkia Journal of Botany, 38, 375–385. http//doi.org/10.3906/bot-1301-36
Sánchez-Ledesma, J. A., Garibay-Orijel, R., Guevara-Guerrero, G., Ávila-Rodríguez, V., & Arreola-Ávila, J. G. (2023). Macromicetos asociados con Carya illinoinensis en la Comarca Lagunera, México. Revista Mexicana de Biodiversidad, 94, e944074. https://doi.org/10.22201/ib.20078706e.2023.94.4074
Sánchez-Ramírez, S. (2015). Scaling macro and micro evolutionary dynamics in the Caesar’s mushrooms (Amanitasect. Caesareae) (Ph.D. Thesis). University of Toronto, Canada.
Sanon, K. B., Bâ, A. M., Delaruelle, C., Duponnois, R., & Martin, F. (2009). Morphological and molecular analyses in Scleroderma species associated with some Caesalpinioid legumes, Dipterocarpaceae and Phyllanthaceae trees in southern Burkina Faso. Mycorrhiza, 19, 571–584. https://doi.org/10.1007/s00572-009-0272-z
Santolamazza-Carbone, S., Iglesias-Bernabé, L., Sinde-Stompel, E., & Gallego, P. P. (2023). Soil microbiota impact on Boletus edulis mycelium in chestnut orchards of different ages. Applied Soil Ecology, 185, 104790. https://doi.org/10.1016/j.apsoil.2022.104790
Sato, Y., Tomonari, H., Kaneko, Y., & Yo, K. (2020). Mushroom poisoning with Scleroderma albidum: a case report with review of the literature. Acute Medicine & Surgery, 7, e460. https://doi:10.1002/ams2.460
Sayers, E. W., Cavanaugh, M., Clark, K., Pruitt, K. D., Sherry, S. T., Yankie, L., & Karsch-Mizrachi, I. (2024). GenBank 2024 update. Nucleic Acids Research, 52, D134–D137. https://doi.org/10.1093/nar/gkad903
Sims, K. P., Watling, R., & Jeffries, P. (1995). A revised key to the genus Scleroderma. Mycotaxon, 56, 403–420. https://doi.org/10.5962/p.415939
Tao, J., Wang, X., Long, Y., Gao, Z., Zhang, G., Guo, Z. et al. (2024). Determining gene order patterns in the Suillus and Boletales through comparative analysis of their mitogenomes. International Journal of Molecular Sciences, 25, 9597. https://doi.org/10.3390/ijms25179597
Tedersoo, L., & Anslan, S. (2019). Towards PacBio-based pan-eukaryote metabarcoding using full-length ITS sequences. Environmental Microbiology Reports, 11, 659–668. https://doi.org/10.1111/1758-2229.12776
Tedersoo, L., Bahram, M., Põlme, S., Kõljalg, U., Yorou, N. S., Wijesundera, R. et al. (2014). Global diversity and geography of soil fungi. Science, 346, 1078–1089. https://doi.org/10.1126/science.1256688
Tedersoo, L., Bahram, M., Zinger, L., Nilsson, R. H., Kennedy, P. G., Yang, T. et al. (2022). Best practices in metabarcoding of fungi: from experimental design to results. Molecular Ecology, 31, 2769–2795. https://doi.org/10.1111/mec.16460
Tedersoo, L., Mikryukov, V., Anslan, S., Bahram, M., Khalid A. N., Corrales, A. et al. (2021). The Global Soil Mycobiome consortium dataset for boosting fungal diversity research. Fungal Diversity, 111, 573–588. https://doi.org/10.1007/s13225-021-00493-7
Truong, C., Mujic, A. B., Healy, R., Kuhar, F., Furci, G., Torres, D.et al.(2017). How to know the fungi: combining field inventories and DNA-barcoding to document fungal diversity. New Phytologist, 214, 913–919. https://doi.org/10.1111/nph.14509
Valdés-Ramírez, M., Ambriz-Parra, E., Camacho-Vera, A., & Fierros-González, A. M. (2010). Inoculación de plántulas de pinos con diferentes hongos e identificación visual de la ectomicorriza. Revista Mexicana de Ciencias Forestales, 1, 53–63.
Wang, X., Liu, P., & Yu, F. (2004). Color atlas of wild commercial mushrooms in Yunnan. Kunming: Yunnan Science and Technology Press.
Wilson, A. W., Binder, M., & Hibbett, D. S. (2012). Diversity and evolution of ectomycorrhizal host associations in the Sclerodermatineae (Boletales, Basidiomycota). New Phytologist, 194, 1079–1095. https://doi:10.1111/j.1469-8137.2012.04109.x
Wilson, A.W., Binder, M., & Hibbett, D. S. (2011). Effects of gasteroid fruiting body morphology on diversification rates in three independent clades of fungi estimated using binary state speciation and extinction analysis.
Evolution, 65, 1305–322. https://doi.org/10.1111/j.1558-5646.2010.01214.x.
Wu, R., Zhou, L., Qu, H., & Ge, Z. W. (2023). Updates on Scleroderma: four new species of section Scleroderma from Southwestern China. Diversity, 15, 775. https://doi.org/10.3390/d15060775
Yang, K. L., Lin, J. Y., Li, G. M., & Yang, Z. L. (2025). Updates of Scleroderma (Basidiomycota, Boletales): new data from 18 selected species in China. Phytotaxa, 706, 209–254. https://doi.org/10.11646/phytotaxa.706.3.3
Yao, Q., Wu, Z., Zhong, J., Yu, C., Li, H., Hu, Q. et al. (2023). A network system for the prevention and treatment of mushroom poisoning in Chuxiong Autonomous Prefecture, Yunnan Province, China: implementation and assessment. BMC Public Health, 23, 1979. https://doi.org/10.1186/s12889-023-16042-7
Zhang, C., Xu, X., & Liu, J. (2013). Scleroderma yunnanense, a new species from South China, in Mycotaxon, 125, 193–200. https://doi.10.5248/125.193
Zhang, Y. Z., Sun, C.Y., Sun, J., Zhang, K. P., Zhang, H. S., Guo, X. et al. (2020). Scleroderma venenatum sp. nov., S. venenatum var. macrosporum var. nov. and S. suthepense new to China. Phytotaxa, 438, 107–118. https://doi.org/10.11646/phytotaxa.438.2.4
Zhong, J., Li H., Zhang, Y., Yu C., Pu, Y., Peng, J. et al. (2021). An investigation of food poisoning by mistaken eating of Scleroderma cepa. Chinese Journal of Food Hygiene, 33, 616–619.