AdminRevista
Posts by :

Lo pasado, pasado: actualización taxonómica y distribución geográfica de la flora vascular de la Estación de Biología Tropical Los Tuxtlas, Veracruz, México
What’s done is done: taxonomic update and geographical distribution of the vascular flora of the Los Tuxtlas Tropical Biology Station, Veracruz, Mexico
Guillermo Ibarra-Manríquez *, Guadalupe Cornejo-Tenorio y Santiago Sinaca-Colín
Universidad Nacional Autónoma de México, Instituto de Investigaciones en Ecosistemas y Sustentabilidad, Antigua carretera a Pátzcuaro Núm. 8701, 58190 Morelia, Michoacán, México
*Autor para correspondencia: gibarra@iies.unam.mx (G. Ibarra-Manríquez)
Recibido: 21 julio 2025; aceptado: 10 octubre 2025
Resumen
El objetivo principal del presente trabajo es actualizar la nomenclatura y el inventario de las especies registradas en la Estación de Biología Tropical Los Tuxtlas (ELT), localizada en el estado de Veracruz, con base en la consulta de literatura florístico-taxonómica, ejemplares de herbario, bases de datos electrónicas y taxónomos. Se registran 934 especies, 571 géneros y 139 familias. Las 20 familias con mayor número de especies abarcan 58.8% de la flora, destacando Orchidaceae (108 especies), Asteraceae (61) y Fabaceae (58); los géneros más relevantes fueron Epidendrum (16), Piper (14) y Ficus (10). Un total de 48 familias (34.5%) y 410 géneros (71.8%) están representados por una especie. Las formas de crecimiento más frecuentes son las hierbas (453 especies, 48.5%) y árboles (259, 27.7%). Noventa y tres especies (10%) son endémicas de México. Las especies de la ELT que se comparten exclusivamente con Centroamérica son las más frecuentes (29.8%), seguidas por las que se registran conjuntamente en Centroamérica, Antillas y Sudamérica (23.2%). Se enfatiza la importancia de continuar con el inventario de esta reserva ubicada en una de las regiones con mayor tasa de deforestación en México.
Palabras clave: Endemismo; Forma de crecimiento; Hábito; Selva alta perennifolia
Abstract
The main objective of this work is to update the nomenclature and inventory of the flora of the Los Tuxtlas Tropical Biology Station, located in Veracruz State, based on floristic-taxonomic literature, herbarium specimens, electronic databases, and support of taxonomists. We registered 934 species, 571 genera, and 139 families. The 20 families with the greatest number of species comprise 58.8% of the flora, highlighting Orchidaceae (108 species), Asteraceae (61), and Fabaceae (58); the most specious genera were Epidendrum (16), Piper (12), and Ficus (10). Forty-eight families (34.5%) and 410 genera (71.8%) are represented by one species. The most common growth forms were herbs (453, 48.5%) and trees (259, 27.7%). Ninety-three species (10%) are endemic to Mexico, and 29.8% of the species are shared exclusively with Central America, followed by the area encompassing Central America, Antilles, and South America (23.2%). The importance of maintaining the inventory of this conservation area in one of Mexico’s regions with the highest rates of deforestation, is emphasized.
Key words: Endemism; Growth form; Life form; Tropical rain forest
Introducción
La región de Los Tuxtlas, localizada en el estado de Veracruz, forma parte de una de las áreas con mayor número de especies del bioma bosque tropical húmedo en México (Villaseñor, 2016) y fue incluida en la provincia biogeográfica de la Costa del Golfo de México por Rzedowski (1978), perteneciente al Reino Neotropical, lo que concuerda con la delimitación de este reino propuesta por Liu et al. (2023). En esta región se decretó, en 1998, la Reserva de la Biosfera Los Tuxtlas, con una extensión de 155,122-46-90 ha (Conanp, 2006). El establecimiento de esta reserva concuerda con su designación como una de las áreas prioritarias más importantes para la conservación de los bosques húmedos de México (Villaseñor et al., 2003). En la zona núcleo Volcán San Martín Tuxtla de esta reserva de la biosfera, se asienta la Estación de Biología Tropical Los Tuxtlas (ELT), perteneciente a la Universidad Nacional Autónoma de México.
De acuerdo con Reynoso et al. (2017), la ELT tiene como áreas de investigación fundamentales el inventario de las especies que protege y estudios sobre la ecología y restauración de la selva tropical húmeda de la región de Los Tuxtlas. Ambas áreas de investigación requieren conocimiento confiable sobre la identidad taxonómica de las especies que conforman su flora. En particular, la primera de ellas no ha perdido vigencia, puesto que los inventarios florísticos son un componente fundamental de agendas de investigación nacionales e internacionales (e.g., Daly et al., 2012; Villaseñor y Meave, 2022), ya que sus resultados permiten realizar distintos tipos de análisis para la obtención de patrones biogeográficos, ecológicos y evolutivos de la biodiversidad (Antonelli et al., 2023; Raven et al., 2020; Sarukhán et al., 2015; Ulloa et al., 2017; Villaseñor y Meave, 2022; Villaseñor y Ortiz, 2014).
Con base en el material de colectores botánicos pioneros como Juan Ismael Calzada, Refugio Cedillo Trigos, Guadalupe Martínez Calderón, Marino Rosas R. y Ángel Villegas Herrera, así como en el trabajo florístico, iniciado en febrero de 1982 para establecer un herbario de referencia para la ELT, Ibarra-Manríquez y Sinaca (1987) publicaron el primer listado de plantas de esta área de conservación. Posteriormente, con el objetivo de aportar datos más detallados de cada especie —e.g., nombre común, forma de crecimiento, fenología reproductiva, usos—, estos autores publicaron una lista comentada (Ibarra-Manríquez y Sinaca, 1995, 1996a, b), en la que se reportaron 940 especies y 543 géneros, agrupados en 137 familias. Un análisis de los usos que para el ser humano tiene este contingente florístico se encuentra en Ibarra-Manríquez, Ricker et al. (1997), así como de su potencial para actividades de restauración regional (Ibarra-Manríquez 2017).
Otras publicaciones que pueden citarse relacionadas con el conocimiento florístico de la ELT son las diagnosis de especies de palmas y trepadoras (Campos et al., 2004; Ibarra-Manríquez, 1988), así como de los frutos y semillas de cerca de 350 especies (Ibarra-Manríquez et al., 2015; Sánchez-Garfías et al., 1991). Más recientemente, Cornejo-Tenorio et al. (2019) publicaron una guía ilustrada que incluye 464 especies con diversas formas de crecimiento. Para toda la región de Los Tuxtlas, existe un manual de determinación de especies de árboles (Vázquez et al., 2010), así como el trabajo de Villaseñor et al. (2018), en el que se analizan distintos aspectos de la flora y que incluye una lista de 2,548 especies.
En el presente siglo, han ocurrido avances sustanciales en el conocimiento florístico y taxonómico de las plantas neotropicales. Por ejemplo, hace todavía un par de décadas habría sido difícil pensar que se contaría con un listado de la flora mexicana (Villaseñor, 2016) y, menos probable aún, con un inventario florístico para todo el continente americano (Ulloa et al., 2017). Estos progresos han sido impulsados, entre otros aspectos, por el desarrollo de bases electrónicas que manejan un gran cúmulo de datos (e.g., POWO, 2025; Tropicos, 2025), en combinación con la conformación del Angiosperm Phylogeny Group, el cual se ha abocado a la elaboración de un sistema de clasificación que refleje, cada vez con mayor precisión, las relaciones de parentesco entre los diferentes taxones de las plantas vasculares de todo el mundo (APG IV, 2016).
Como se describe en el párrafo previo, la lista aportada por Ibarra-Manríquez y Sinaca (1995, 1996a, b), la más detallada para toda la flora de la ELT, requiere una revisión nomenclatural, lo cual fue sugerido previamente por Ibarra-Manríquez (2017). En consecuencia, el presente estudio tiene como objetivos principales: 1) actualizar la nomenclatura taxonómica de las especies registradas en esta área de conservación, 2) adicionar a su inventario las especies descritas desde 1997 a la fecha y 3) documentar la forma de crecimiento, el hábito y la distribución geográfica de cada especie.
Materiales y métodos
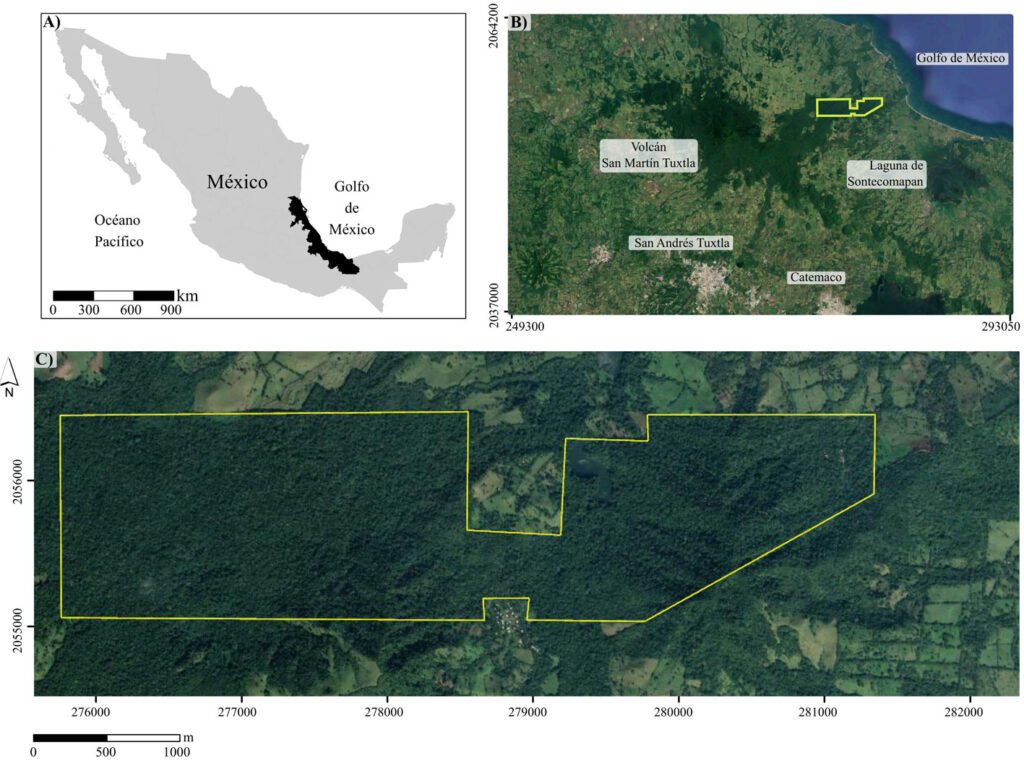
La Estación de Biología Tropical Los Tuxtlas es un área de conservación de 640 ha, con elevaciones entre 130 y 560 m (fig. 1), que pertenece al municipio de San Andrés Tuxtla, Veracruz. El relieve de la sierra de Los Tuxtlas es principalmente volcánico, cuya actividad data del Terciario; los sedimentos más antiguos (arcillas tobáceas y areniscas marinas de la formación La Laja) han sido fechados del Oligoceno (Martin-Del Pozzo, 1997). La información detallada sobre el clima de la región de Los Tuxtlas, que incluye mapas de los promedios anuales de temperatura y precipitación, puede consultarse en Gutiérrez-García y Ricker (2011). Con base en Soto y Gama (1977), se infiere que el clima en la ELT es cálido-húmedo [Af (m)], con lluvias todo el año, precipitación promedio anual entre 3,000 y 4,000 mm, precipitación del mes más seco mayor de 60 mm, lluvia invernal con respecto a la anual menor de 18%, temperatura media anual mayor de 22 oC y temperatura del mes más frío superior a 18 oC. Recientemente, Ek-Rodríguez et al. (2022) reportan para la ELT promedios anuales de temperatura (24.2 °C) y precipitación (3,433 mm), con una época seca corta (abril y mayo).
El tipo de vegetación en la ELT es la selva alta perennifolia (Miranda y Hernández, 1963), bosque tropical perennifolio (Rzedowski, 1978) o con base en la clasificación de Holdridge, bosque tropical húmedo (Ibarra-Manríquez et al., 1997; Gutiérrez-García y Ricker, 2011). Ibarra-Manríquez, Martínez-Ramos et al. (1997) enlistan las especies más frecuentes en 3 variantes del bosque tropical húmedo que difieren en fisonomía, elevación y tipos de suelo sobre los que se establecen (figs. 2-5; ver material suplementario para las autoridades taxonómicas de las especies). Los autores destacaron la presencia en el estrato bajo del bosque (1-10 m) de las palmas, particularmente de Astrocaryum mexicanum y Chamaedorea spp. Otras especies importantes de este estrato son Acalypha skutchii (Euphorbiaceae), Piper spp. (Piperaceae), Trophis mexicana (Moraceae) y otras pertenecientes a Urticaceae (Myriocarpa longipes, Urera glabriuscula y Urera simplex) y Rubiaceae (Faramea occidentalis, Palicourea faxlucens y Palicourea tetragona). En el estrato medio y alto se encuentran Bursera simaruba (Burseraceae), Calatola uxpanapensis (Metteniusaceae), Cecropia obtusifolia (Urticaceae), Croton schiedeanus (Euphorbiaceae), Ceiba pentandra (Malvaceae), Cymbopetalum baillonii (Annonaceae), Damburneya ambigens (Lauraceae), Dendropanax arboreus (Araliaceae), Garcinia intermedia (Clusiaceae), Guarea glabra (Meliaceae), Licaria velutina (Lauraceae), Omphalea oleifera (Euphorbiaceae), Pterocarpus rohrii (Fabaceae) y especies de Moraceae (Brosimum alicastrum, Ficus isophlebia, Ficus yoponensis, Poulsenia armata y Pseudolmedia glabrata. Entre las lianas destacan varias especies de Bignoniaceae (e.g., Callichlamys latifolia, Fridericia schumanniana, Tanaecium pyramidatum), así como Abuta panamensis (Menispermaceae), Forsteronia acouci (Apocynaceae) y Salacia cordata (Celastraceae). En lo que se refiere a las hierbas, ya sean terrestres, epífitas o hemiepífitas, es evidente la abundancia de especies de Acanthaceae (Aphelandra aurantiaca, Schaueria parviflora), Araceae (Anthurium spp., Dieffenbachia oerstedii, Philodendron spp., Spathiphyllum ortgiesii, Syngonium spp.), Orchidaceae (Epidendrum spp., Nidema boothii, Oncidium sphacelatum, Prosthechea cochleata) y Piperaceae (Peperomia spp.).

La vegetación cambia sus hojas de manera paulatina a lo largo del año, con algunas especies caducifolias (Ibarra-Manríquez, Martínez-Ramos et al., 1997), particular-
mente durante la época de secas, por ejemplo, Bernoullia flammea, Ceiba pentandra (ambas Malvaceae), Bursera simaruba (Burceraceae), Dussia mexicana, Erythrina folkersii, Vatairea lundelli (todas Fabaceae), Ficus isophlebia, Handroanthus guayacan (Bignoniaceae), Jacaratia dolichaula (Caricaceae) y Omphalea oleifera. La altura de los árboles del dosel puede alcanzar entre 10-20 m en las cimas de los cerros El Vigía o Cerro Azul (Lázaro Cárdenas), mientras que en sitios con menor pendiente pueden alcanzar de 30 a 40 m (Ibarra-Manríquez, Martínez-Ramos et al., 1997). Para información más detallada sobre la vegetación consultar Bongers et al. (1988), Popma et al. (1988), Manríquez, Ricker et al. (1997), Ibarra-Manríquez, Martínez-Ramos (1997), Navarrete-Segueda et al. (2021), Miranda-Gallegos et al. (2023) y Ek-Rodríguez et al. (2022, 2024, 2025).


La lista de especies para helechos y grupos afines está ordenada con base en PPG I (2016) y para las angiospermas la clasificación de APG IV (2016). Las familias, géneros y especies siguen un orden alfabético. La familia y especie con el que los integrantes de la flora fueron citados en Ibarra-Manríquez y Sinaca (1995, 1996a, b) fueron revisados y en caso de ser necesario, se actualizaron nomenclaturalmente con base en lo indicado por estudios taxonómicos (tabla 1), la revisión de las bases de datos POWO (2025) y Tropicos (2025), así como la consulta directa con taxónomos; estas fuentes de consulta fueron también usadas para indicar la distribución geográfica de cada especie.

La forma de crecimiento, hábito, altura de las especies, nombre común, así como la asociación con pastizales antropogénicos o vegetación ruderal de las especies, se derivaron de observaciones en campo realizadas de febrero de 1982 a junio de 2025, así como de la consulta de material depositado en el Herbario Nacional (MEXU) y el herbario de referencia que se encuentra en la ELT. Las formas de crecimiento y hábito siguen las definiciones de Moreno (1984). En particular, los árboles y arbustos fueron clasificados con base en su altura, ya que los primeros son plantas leñosas ≥ 3 m, mientras que los arbustos no superan este valor. En el caso de las especies con hábito trepador, las leñosas se denominan lianas, mientras que las que no presentan esta condición son trepadoras. Para las especies cuyo ejemplar tipo fue recolectado en la ELT, se indican los nombres de los colectores, el número de colecta y las siglas de los herbarios en los que se encuentran depositados el holotipo e isotipos. El listado excluye las especies cultivadas como Mangifera indica L. (Anacardiaceae) y Pachira aquatica Aubl. (Malvaceae).
Resultados
La lista compilada incluye 934 especies, 571 géneros y 139 familias (material suplementario). Entre los grupos de clasificación con jerarquía más alta (tabla 2; material suplementario), las Eudicotiledóneas son las que tienen una representación mayor respecto al número de familias (68.3%), géneros (67.4%) y especies (60.9%). Las 20 familias con mayor número de especies abarcan 59.1% de la flora, destacando Orchidaceae (108 especies), Asteraceae (61), Fabaceae (58), Rubiaceae (35) y Poaceae (28) (tabla 3). A nivel de género, los más relevantes al respecto son Epidendrum L. (16 especies), Piper L. (13), Ficus L. (10), así como Peperomia Ruiz et Pav. y Solanum L. (9 especies cada uno). Cuarenta y ocho familias (34.5%) y 410 géneros (71.8%) están representados por 1 especie. Un total de 27 especies fueron descritas como nuevas para la ciencia con base en colectas realizadas en la ELT; de 1997 a la fecha, 45 especies son integrantes adicionales de su flora (material suplementario).
Las formas de crecimiento más frecuentes fueron las hierbas (48.5%) y los árboles (27.7%); respecto al hábito de crecimiento, 77.6% de las especies son terrestres, seguidas en importancia por las trepadoras y epífitas que representan 17.3% y 16%, respectivamente (tabla 4). Las distribuciones geográficas con mayores registros de especies fueron las que se comparten exclusivamente entre México y Centroamérica (278 especies, 29.8%), ocupando el segundo sitio la agrupación formada por estas 2 áreas con Antillas y Sudamérica (217, 23.2%) (tabla 5). Catorce especies son cosmopolitas, 3 pantropicales y 14 introducidas (material suplementario). Un total de 93 especies (10%) son endémicas de México y 17 restringen su presencia a la región de Los Tuxtlas o Veracruz (tabla 6, fig. 6). Cerca de 24% de las especies se asocian con pastizales antropogénicos y vegetación ruderal (material suplementario), sin que se haya registrado su presencia en el bosque tropical perennifolio de la reserva, tanto en su fase madura como en etapas sucesionales tempranas o intermedias.
Discusión
Florística
La dominancia actual de las angiospermas entre las plantas vasculares del mundo es un hecho ampliamente documentado y se constata en el presente estudio (tabla 2), ya que engloban 91.5% de las especies, porcentaje prácticamente igual al que reportan Villaseñor et al. (2018) para toda la región de Los Tuxtlas (91.2%). El comparativo de la riqueza taxonómica reportado por Ibarra-Manríquez y Sinaca (1995, 1996a, b) y el del presente estudio no presentan diferencias cuantitativas notables a nivel de familias (139 y 138, respectivamente) ni de especies (940 y 934). Sin embargo, cualitativamente los cambios son numerosos (material suplementario). A nivel de familia, existen casos en que algunas de ellas han sido reconocidas como distintas (e.g., Phyllanthaceae, antes incluida en Euphorbiaceae; Dipentodontaceae, anteriormente en Celastraceae) o, por el contrario, como sinónimos (e.g., Asclepiadaceae de Apocynaceae; Bombacaceae y Tiliaceae de Malvaceae; Hippocrateaceae de Celastraceae; Myrsinaceae y Theophrastaceae de Primulaceae).
Tabla 1
Referencias florístico-taxonómicas consultadas para la elaboración del material suplementario. La etiqueta de varias incluye familias de helechos y licófitos.
| Familias | Referencias |
| Acanthaceae | Ramamoorthy y Uribe (1988), Daniel (2002, 2004, 2005) |
| Annonaceae | Ortiz-Rodriguez (2022), Ortiz-Rodríguez et al. (2024) |
| Apocynaceae | Morales (1997), Alvarado-Cárdenas et al. (2020) |
| Araceae | Krömer et al. (2019) |
| Arecaceae | Bacon y Bailey (2006), Henderson (2011) |
| Asteraceae | Villaseñor y Ortiz (2025) |
| Bromeliaceae | Espejo-Serna et al. (2005), Espejo-Serna y López-Ferrari (2018) |
| Celastraceae | Lombardi (2014), Biral (2021) |
| Costaceae | García-Mendoza e Ibarra-Manríquez (1991) |
| Cyperaceae | González-Elizondo et al. (2018) |
| Elaeocarpaceae | Palacios-Wassenaar y Castillo-Campos (2020) |
| Erythroxylaceae | Palacios-Wassenaar y Castillo-Campos (2019) |
| Fabaceae | Sousa y Rudd (1993), Pennington (1997), Zamora (2006), Sousa (2009, 2010), Aviles et al. (2022) |
| Gesneriaceae | Ramírez-Roa y Ibarra-Manríquez (1998), Clavijo et al. (2021), Ramírez-Roa et al. (2023) |
| Icacinaceae | Vera-Caletti y Wendt (2001) |
| Lamiaceae | Martínez-Gordillo et al. (2017) |
| Lauraceae | Rowher (1993), Lorea-Hernández (2002) |
| Loganiaceae | Islas-Hernández y Alvarado-Cárdenas (2023) |
| Malvaceae | Fryxell (1992) |
| Moraceae | Ibarra-Manríquez et al. (2012), Berg (2015), Hernández-Esquivel et al. (2020) |
| Orchidaceae | Salazar (1988) |
| Passifloraceae | Fragoso-Martínez y Castillo-Campos (2023) |
| Piperaceae | Vergara-Rodríguez et al. (2017), Callejas-Posada (2020), Carmona-Hernández et al. (2022) |
| Poaceae | Dávila et al. (2018) |
| Polygonaceae | Ancona et al. (2025) |
| Proteaceae | Edwards y Prance (2003) |
| Rubiaceae | Lorence y Nee (1987), Torres-Montúfar y Ochoterena (2013), Torres-Montúfar y Torres-Díaz (2022) |
| Sambucaceae | Villareal (2003) |
| Sapotaceae | Swenson et al. (2023) |
| Selaginellaceae | Zhou y Zhang (2023) |
| Smilacaceae | Ferrufino-Acosta (2010) |
| Thelypteridaceae | Riba (2009), Salino et al. (2015) |
| Urticaceae | Monro (2009) |
| Varias | Acebey et al. (2015), Smith y Tejero-Díez (2014), Krömer et al. (2020), Ruiz et al. (2025) |
Tabla 2
Síntesis taxonómica de la flora vascular de la Estación de Biología Tropical Los Tuxtlas.
| Taxones | Familias | Géneros | Especies |
| Lycopodiopsida | 2 | 4 | 11 |
| Polypodiopsida | 16 | 37 | 68 |
| Magnólidas | 9 | 21 | 55 |
| Monocotiledóneas | 17 | 124 | 231 |
| Eudicotiledóneas | 95 | 385 | 569 |
| Total | 139 | 571 | 934 |
Tabla 3
Familias y géneros con mayor número de especies en la zona de estudio.
| Familias | Especies | Géneros | Especies |
| Orchidaceae | 108 | Epidendrum | 16 |
| Asteraceae | 61 | Piper | 14 |
| Fabaceae | 58 | Ficus | 10 |
| Rubiaceae | 35 | Peperomia | 9 |
| Poaceae | 28 | Solanum | 9 |
| Euphorbiaceae | 26 | Eugenia | 8 |
| Araceae | 23 | Inga | 8 |
| Piperaceae | 23 | Passiflora | 8 |
| Solanaceae | 23 | Asplenium | 7 |
| Apocynaceae | 20 | Chamaedorea | 7 |
| Lauraceae | 19 | Cyperus | 7 |
| Bromeliaceae | 17 | Ocotea | 7 |
| Malvaceae | 17 | Begonia | 6 |
| Bignoniaceae | 16 | Desmodium | 6 |
| Moraceae | 16 | Lasiacis | 6 |
| Verbenaceae | 13 | Miconia | 6 |
| Arecaceae | 12 | Mikania | 6 |
| Melastomataceae | 12 | Philodendron | 6 |
| Polypodiaceae | 12 | Pleopeltis | 6 |
| Total | 539 (57.7%) | 152 (16.3%) |
Tabla 4
Número de especies de las formas de crecimiento y hábito de las plantas vasculares en la Estación de Biología Tropical Los Tuxtlas.
| Formas de crecimiento | Especies (%) | Hábito | Especies (%) |
| Árboles | 259 (27.7) | Epífitas | 149 (16) |
| Arbustos | 85 (9.1) | Hemiepífitas | 24 (2.6) |
| Helechos arborescentes | 3 (0.3) | Hemiparásitas | 3 (0.3) |
| Hierbas | 453 (48.5) | Rupícolas | 14 (1.5) |
| Palmas | 12 (1.3) | Parásitas | 1 (0.1) |
| Trepadoras leñosas (lianas) | 94 (10.1) | Terrestres | 725 (77.6) |
| Trepadoras herbáceas | 67 (7.2) | Trepadoras | 161 (17.3) |
Tabla 5
Número de especies de la flora vascular de la Estación de Biología Tropical Los Tuxtlas registradas en distintas áreas geográficas de América.
| País/región | Especies (%) |
| México | 93 (10) |
| México-Centroamérica | 278 (29.8) |
| México-Centroamérica-Antillas | 18 (1.9) |
| México-Centroamérica-Antillas-Sudamérica | 217 (23.2) |
| México-Centroamérica-Sudamérica | 198 (21.2) |
| México-Norteamérica | 1 (0.1) |
| México-Norteamérica-Centroamérica | 3 (0.3) |
| México-Norteamérica-Centroamérica | 2 (0.2) |
| México-Norteamérica-Centroamérica-Antillas-Sudamérica | 74 (7.9) |
| México-Sudamérica | 1 (0.1) |
En el material suplementario se puede revisar que 202 especies son actualmente consideradas como sinónimos y 48 han sido redeterminadas; en conjunto, representan 26.7% del total de la flora, con un promedio anual de 8.6 especies (1997-2025). En ambos casos, su clasificación actual puede incluso asociarse con distintos géneros, como lo ejemplifican Bolbitis bernoullii, sinónimo de Mickelia bernoullii (Dryopteridaceae), Oerstedianthus brevipes de Ardisia tuerckheimii (Primulaceae), Psychotria faxlucens de Palicourea faxlucens (Rubiaceae) o Rheedia edulis de Garcinia intermedia (Clusiaceae); en este rubro, destacan particularmente Bignoniaceae y Lauraceae, ya que alrededor de la mitad de sus especies fueron transferidas a otros géneros. Finalmente, se han descrito varias especies nuevas para la ciencia (material suplementario, tabla 6), que abarcan diversas formas de crecimiento y familias (e.g., Acanthaceae, Annonaceae, Gesneriaceae, Magnoliaceae, Polygonaceae, Proteaceae, Rubiaceae, Sapotaceae).
Las 15 familias con mayor número de especies concentran 52.5% del total registrado para la ELT (tabla 3) y al comparar este rubro con Villaseñor et al. (2018), nuevamente existe una coincidencia alta (50.8%). Ambos estudios concuerdan en 13 de ellas y de las 5 primeras, solo difieren en la posición particular que ocupan Orchidaceae, Asteraceae y Fabaceae. La jerarquía de estas familias se explica por su ubicación entre las más diversas de México y del continente americano (Ulloa et al., 2017; Villaseñor, 2016). Sin embargo, es conveniente considerar que 67.2 y 89.3% de las especies de Asteraceae y Poaceae, respectivamente, se registran únicamente en ambientes ruderales o pastizales de origen antropogénico (material suplementario). En el caso de los géneros con mayor diversidad, 9 de 15 concuerdan entre la región de Los Tuxtlas y el presente estudio, en particular Epidendrum (Orchidaceae), Piper y Peperomia (Piperaceae), Ficus (Moraceae), Solanum (Solanaceae) y Asplenium L. (Aspleniaceae). Los 3 primeros géneros son también encontrados entre los de mayor riqueza en México y América (Ulloa et al., 2017; Villaseñor, 2016).
En situación opuesta, 48 familias (34.5%) y 410 géneros (71.8%) tienen solo una especie en la ELT. Este resultado guarda relación con lo que se registra en México (Villaseñor, 2016), como acontece con Haemodoraceae y Lacistemataceae. Una situación análoga ocurre con numerosos géneros, entre los que pueden citarse Ampelocera Klotzsch (Ulmaceae), Aphananthe Planch. (Cannabaceae), Astrocaryum G. Mey (Arecaceae), Calophyllum L. (Calophyllaceae), Dialium L. (Fabaceae), Lunania Hook. (Salicaceae), Mosquitoxylum Krug et Urb. (Anacardiaceae), Nidema Britton et Millsp. (Orchidaceae), Ochroma Sw. (Malvaceae), Odontocarya Miers (Menispermaceae), Petrea L. (Verbenaceae), Pimenta Lindl. (Myrtaceae), Pleuranthodendron L.O. Williams (Salicaceae), Poulsenia Eggers (Moraceae), Sapindus L. (Sapindaceae), Tuxtla Villaseñor et Strother (Asteraceae) o Vatairea Aubl. (Fabaceae).
Tabla 6
Especies con distribución restringida a la región de Los Tuxtlas o al estado de Veracruz; se indica con un asterisco las que fueron descritas después de las publicaciones de Ibarra-Manríquez y Sinaca (1995, 1996a, b).
| Especies/área | Familia | Forma de crecimiento |
| Región de Los Tuxtlas | ||
| Capparidastrum tuxtlense* | Capparaceae | Árbol |
| Clethra tuxtlensis* | Clethraceae | Árbol |
| Costus dirzoi | Costaceae | Hierba |
| Drymonia mexicana* | Gesneriaceae | Arbusto |
| Hoffmannia altipetens* | Rubiaceae | Arbusto |
| Magnolia sinacacolinii* | Magnoliaceae | Árbol |
| Miconia ibarrae | Melastomataceae | Árbol |
| Mosannona depressa subsp. abscondita* | Annonaceae | Árbol |
| Piper marginecontinuum* | Piperaceae | Arbusto |
| Tridimeris tuxtlensis* | Annonaceae | Árbol |
| Veracruz | ||
| Arachnothryx pumae* | Rubiaceae | Árbol o arbusto |
| Croton sousae* | Euphorbiaceae | Árbol |
| Daphnopsis megacarpa | Thymelaeaceae | Arbusto |
| Eugenia colipensis | Myrtaceae | Árbol |
| Eugenia inirebensis | Myrtaceae | Árbol |
| Macroclinium pachybulbon | Orchidaceae | Hierba |
| Ruellia tuxtlensis | Acanthaceae | Hierba |
Formas de crecimiento y hábito
La fisonomía del bosque tropical perennifolio se debe a la abundancia y al tamaño de los árboles, primordialmente a los del estrato de mayor altura y la ELT no es la excepción al respecto (Bongers et al., 1988; Ibarra-Manríquez, Martínez-Ramos et al., 1997; Navarrete-Segueda et al., 2021). La importancia del componente arbóreo no se relaciona solo con la estructura de los bosques (e.g., contribución a la biomasa), sino también por su desempeño en aspectos funcionales, como la captura de carbono y las interacciones que mantiene con polinizadores y frugívoros (Beckman y Sullivan, 2023; Fuster, et al., 2018; Lutz et al., 2018; Ollerton, 2017; Spicer et al., 2020). Si bien en la ELT esta forma de crecimiento es relevante por su número de especies (259; 27.7%), es superada ampliamente por las hierbas (453, 48.5%).
Este patrón de dominancia del número de especies de hierbas respecto a los árboles debe destacarse en los estudios florísticos de los bosques tropicales, puesto que ha sido registrado previamente (Ibarra-Manríquez et al., 2021; Linares-Palomino et al., 2009; Meave et al., 2017; Rojas-Martínez y Flores-Olvera, 2019). A nivel de bioma, este patrón de riqueza se mantiene, ya que la proporción de hierbas y árboles es de 45% y 30%, respectivamente (Spicer et al., 2020). Para la flora de México, Villaseñor y Ortiz (2014) destacaron la predominancia de las hierbas perennes o anuales (13,408) sobre los árboles (4,044), un aspecto que había sido sugerido primeramente por Rzedowski (1991a). Como una consecuencia de lo anterior, el hábito terrestre es el preponderante en la ELT (77.6%), debido al efecto aditivo de la riqueza de las hierbas y árboles, aunado al aporte de las trepadoras, tanto leñosas como herbáceas. Para los bosques tropicales, Spicer et al. (2020) concuerdan en resaltar la dominancia de este hábito, al englobar 80% de las especies, con el remanente adscrito a las epífitas.

Distribución geográfica y endemismo
El 29.8% de la flora vascular de la ELT se comparte exclusivamente con Centroamérica, mientras que 21.2% se registra en esta área en conjunto con Sudamérica (tabla 5), posiciones que coinciden con lo reportado por Villaseñor et al. (2003) en su estudio biogeográfico de los bosques tropicales húmedos de México (31.4 y 12.7%, respectivamente). Estas afinidades biogeográficas también coinciden con Wendt (1993), quien determinó que el componente arbóreo del dosel de los bosques tropicales de México tenía sus mayores nexos con Centroamérica y con lo destacado por Rzedowski (1991a, b), en relación con las afinidades marcadamente meridionales del bosque tropical perennifolio de México.
Al respecto, es interesante destacar las distribuciones vicariantes de 4 especies, que sólo se han recolectado en la región de Los Tuxtlas y en localidades de Costa Rica (Haydenoxylon calzadae [Celastraceae], Styphnolobium parviflorum [Fabaceae] y Tuxtla pittieri [Asteraceae]) y Honduras (Justicia tuxtlensis [Acanthaceae]), lo que demanda estudios más detallados para explicarlas debidamente, ya que no parecen asociarse a una exploración deficiente de la flora, tomando en cuenta los distintos proyectos florísticos que se están implementando desde hace décadas en la región mesoamericana (e.g., Flora de Costa Rica, Flora de Nicaragua, Flora Mesoamericana).
Por otro lado, se debe discutir lo expuesto por Villaseñor et al. (2018), quienes resaltaron que 6 especies de la región de Los Tuxtlas solo se encuentran en la ELT, lo cual no es correcto para Justicia tuxtlensis, debido a su distribución vicariante previamente expuesta. Esta aseveración tampoco es válida para Capparidastrum tuxtlense (Capparaceae) y Hoffmannia altipetens (Rubiaceae), ya que han sido colectadas tanto en la ELT como en otras localidades de la región. Las 3 especies restantes carecen de registros que certifiquen su presencia en la reserva. Styrax tuxtlensis P.W. Fritsch (Styracaceae) se conoce únicamente de un registro proveniente del volcán Santa Marta, lo que hace muy improbable su presencia en la ELT. En el caso de Guadua tuxtlensis Londoño et Ruiz-Sánchez (Poaceae), es factible que pueda ser colectada en los perímetros de la ELT que colindan con pastizales para la crianza de ganado (fig. 1), ya que es uno de los tipos de vegetación en los que se le ha reportado. De igual manera, la presencia de Diospyros tuxtlensis Provance et A.C. Sanders (Ebenaceae) es viable considerando su colecta en localidades no tan lejanas al oeste de la reserva. Hasta ahora, las especies que se conocen exclusivamente de la ELT son Piper marginecontinuum (Piperaceae) y Tridimeris tuxtlensis (Callejas Posada, 2020; Ortiz-Rodríguez et al., 2024).
La flora endémica de México registrada en la ELT representa 10% del total, una cifra mayor que la sugerida por Rzedowski (1991a) para el bosque tropical perennifolio (5%), similar (9.6%) a la que reportó Wendt (1993) y menor (20.5%) que la citada en Villaseñor et al. (2003). A pesar de la discrepancia de estos valores, existe concordancia en que la región de Los Tuxtlas ha desempeñado un papel relevante como área de concentración de especies arbóreas endémicas (Wendt, 1993), junto con el norte de Oaxaca (Tuxtepec) y el SE de Veracruz (Uxpanapa). Las marcadas similitudes florísticas del bosque tropical húmedo de Veracruz son mayores con Chiapas, Oaxaca, Tabasco y Puebla (Villaseñor y Ortiz, 2025). A nivel de familia, es interesante señalar que 7 de las 8 especies registradas de Gesneriaceae son endémicas de México.
Como conclusiones generales del estudio se debe resaltar que la actualización taxonómica de la flora vascular de la ELT modifica el reconocimiento de una proporción importante de especies, géneros y familias, y por su relevancia, es una actividad que requiere de actualizaciones continuas. Lo dinámico de este proceso se refleja en la tasa de cambios nomenclaturales registrada para la reserva de 1997 a la fecha (8.6 especies anualmente). Se espera que la presente contribución facilite recabar información de diferentes áreas del conocimiento sobre las especies citadas en el material suplementario, por ejemplo, coordinar iniciativas para la restauración regional (Ibarra-Manríquez, 2017). El número de especies registrado en esta área protegida (934 especies) representa 27.8% de las 3,362 que citan Villaseñor y Ortiz (2025) para este bioma en Veracruz, un porcentaje muy importante si se considera la pequeña extensión de esta reserva (640 ha).
Se recomienda continuar con el inventario de la ELT, con el objetivo de cuantificar la composición florística actual con más detalle, que permita, entre otras cosas, detectar la abundancia de las especies y realizar actividades que promuevan su conservación, particularmente para aquellas poco abundantes o con una distribución restringida a ciertas áreas de la ELT (Ibarra-Manríquez, Martínez-Ramos et al., 1997); lo anterior podría ser más aplicable para las especies que registran un bajo número de exsiccatas (material suplementario). Sin duda alguna, esta actividad prioritaria también podría identificar especies nuevas para la ciencia, como lo demuestran los hallazgos registrados desde la década de los 80 hasta la fecha. Una evidencia que respalda lo anterior es que el material tipo de 27 especies fue recolectado dentro de los límites territoriales de la ELT (material suplementario), de las cuales 15 fueron descritas de 1997 a la fecha. Se espera que el presente estudio fomente acciones para conservar el valioso patrimonio biológico del bosque tropical húmedo que resguarda la ELT, límite más norteño de este tipo de vegetación en América. Se debe valorar aún más este papel, especialmente si se considera su localización en elevaciones bajas, que, debido a diversos factores, cada vez tiene menor extensión en la región de Los Tuxtlas (Bonilla-Moheno y Aide, 2020; von Thaden et al., 2020). También sería extremadamente valioso atemperar el deterioro biológico señalado por Ibarra-Manríquez (2017), especialmente en las partes con mayor acceso a actividades humanas.
Agradecimientos
Dos árbitros anónimos y M. Socorro González-Elizondo, en su calidad de editora asociada, aportaron valiosas sugerencias para mejorar una primera versión del presente estudio. Agradecemos a los taxónomos que amablemente revisaron las especies de las familias de su especialidad: Leonardo O. Alvarado-Cárdenas (Apocynaceae), Thomas F. Daniel (Acanthaceae), Mario Adolfo Espejo Serna (Bromeliaceae), Francisco Gerardo Lorea Hernández (Lauraceae). Se reconoce el apoyo de Armando Navarrete Segueda en la elaboración de la figura 1 y finalmente, el de María Antonieta Arizmendi Espinosa en las distintas fases editoriales para finalizar esta publicación.
Referencias
Acebey, A. R., Krömer, T., Vázquez-Torres, M. y Tejero-Díez, J. D. (2015). Helechos y licofitos de la Reserva de la Biosfera Los Tuxtlas, Veracruz, México. Botanical Sciences, 93, 1–32. https://doi.org/10.17129/botsci.124
Alvarado-Cárdenas, L. O., Lozada-Pérez, L., Islas-Hernández, C. S., Cortez, E. B., Maya-Mandujano, K. G. y Chávez-Hernández, M. G. (2020). Apocináceas de ayer y hoy. Conocimiento histórico y reevaluación de la diversidad y distribución de Apocynaceae en México. Botanical Sciences, 98, 393–416. https://doi.org/10.17129/botsci.2525
Ancona, J. J., Ortiz-Díaz, J. J. y Hernández-Ledesma, P. (2025). Eight new species of Coccoloba sect. Campderia (Polygonaceae, Eriogonoideae) from Mexico and Central America. Acta Botanica Mexicana, 132, e2435. https://doi.org/10.21829/abm132.2025.2435
Antonelli, A., Fry, C., Smith, R. J., Eden, J., Govaerts, R. H. A., Kersey, P. et al. (2023). State of the World’s plants and fungi 2023. Royal Botanic Gardens, Kew. England. https://doi.org/10.34885/wnwn-6s63
APG IV (The Angiosperm Phylogeny Group). (2016). An update of the Angiosperm Phylogeny Group classification for the orders and families of flowering plants: APG IV. Botanical Journal of the Linnean Society, 181, 1–20. https://doi.org/10.1111/boj.12385
Aviles, P. G., Koenen, E. J. M., Riina, R., Hughes, C. E., Ringelberg, J. J., Carnevali Fernández-Concha, G. et al. (2022). Re-establishment of the genus Pseudalbizzia (Leguminosae, Caesalpinioideae, mimosoid clade): the New World species formerly placed in Albizia. Phytokeys, 205, 371–400. https://doi.org/10.3897/phytokeys.205.76821
Bacon, C. D. y Bailey, C. D. (2006). Taxonomy and conservation: a case study from Chamaedorea alternans. Annals of Botany, 98, 755–763. https://doi.org/10.1093/aob/mcl158
Beckman, N. G. y Sullivan, L. L. (2023). The causes and consequences of seed dispersal. Annual Review of Ecology, Evolution, and Systematics, 54, 403–427. https://doi.org/10.1146/annurev-ecolsys-102320-104739
Berg, C. C. (2015). Moraceae. En G. Davidse, M. Sousa, S. Knapp y F. Chiang (Eds.), Flora Mesoamericana, Saururaceae a Zygoplyllaceae (pp. 90–116). St. Louis Missouri: Sheridan Books.
Biral, L. (2021). A new combination and a new synonym in Haydenoxylon (Celastraceae). Novon, 29, 74–76. https://doi.org/10.3417/2021646
Bongers, F., Popma, J., Meave-del Castillo, J. y Carabias, J. (1988). Structure and floristic composition of the lowland rain forest of Los Tuxtlas, Mexico. Vegetatio, 74, 55–80. https://doi.org/10.1007/BF00045614
Bonilla-Moheno, M. y Aide, T. M. (2020). Beyond deforestation: land cover transitions in Mexico. Agricultural Systems,
178, 102734. https://doi.org/10.1016/j.agsy.2019.102734
Callejas-Posada, R. (2020). Piperaceae. En G. Davidse, C. Ulloa-Ulloa, H. Hernández-Macías y S. Knapp (Eds.) Flora Mesoamericana (pp. 1–590). St. Louis Missouri: Sheridan Books.
Campos, V. A., Kelly, L. M. y Delgado, S. A. (2004). Bejucos y otras trepadoras de la Estación de Biología Tropical Los Tuxtlas, Veracruz, México. México D.F.: Instituto de Biología, Universidad Nacional Autónoma de México.
Carmona-Hernández, O., Laccetti, L., Martínez-Hernández, M. D. J., Luna-Rodríguez, M., Fernández, M. D. S., Guerrero-Analco, J. A. et al. (2023). Plant conservation in the Mesoamerican biodiversity hotspot: a case study on the Piper genus in Veracruz (Mexico). Tropical Ecology, 64, 324–336. https://doi.org/10.1007/s42965-022-00271-9
Clavijo, L., Ramírez-Roa A. y Clark, J. L. (2021). Drymonia mexicana (Gesneriaceae), a new endemic species from Veracruz (Mexico). Journal of the Botanical Research Institute of Texas, 15, 60–63. https://doi.org/10.17348/jbrit.v15.i1.1050
Conanp (Comisión Nacional de Áreas Naturales Protegidas). (2006). Programa de conservación y manejo Reserva de la Biosfera Los Tuxtlas. México D.F.: Comisión Nacional de Áreas Naturales Protegidas.
Cornejo-Tenorio, G., Ibarra-Manríquez, G. y Sinaca-Colín, S. (2019). Flora de Los Tuxtlas. Guía Ilustrada. México D.F.: Universidad Nacional Autónoma de México.
Daly, M., Herendeen, P. S., Guralnick, R. P., Westneat, M. W. y McDade, L. (2012). Systematics agenda 2020: the mission evolves. Systematic Biology, 61, 549–552. https://doi.org/10.1093/sysbio/sys044
Daniel, T. F. (2002). New and reconsidered Mexican Acantha-
ceae IX. Justicia. Proceedings of the California Academy of Sciences, 53, 37–49.
Daniel, T. F. (2004). Further range extensions of Mexican Acanthaceae. Polibotánica, 18, 1–12.
Daniel, T. F. (2005). Catalog of Honduran Acanthaceae with taxonomic and phytogeographic notes. Contributions from the University of Michigan Herbarium, 24, 51–108.
Dávila, P., Mejía-Saulés, M. T., Soriano-Martínez, A. M. y Herrera-Arrieta, Y. (2018). Conocimiento taxonómico de la familia Poaceae en México. Botanical Sciences, 96, 462–514. https://doi.org/10.17129/botsci.1894
Edwards, K. S. y Prance, G. Y. (2003). Four new species of Roupala (Proteaceae). Brittonia, 55, 61–68. https://www.jstor.org/stable/3218415?seq=1
Ek-Rodríguez, I. L., Coates, R., Sinaca-Colín, S. e Ibarra-Manríquez, G. (2022). Liana community attributes in one of the northernmost neotropical rainforests. Botanical Sciences, 100, 353–369. https://doi.org/10.17129/botsci.2955
Ek-Rodríguez, I. L., Meave, J. A., Navarrete-Segueda, A., González-Arqueros, M. L. e Ibarra- Manríquez, G. (2024). Environmental heterogeneity influences liana community differentiation across a neotropical rainforest landscape. Ecology et Evolution, 14, e11170. https://doi.org/10.1002/ece3.11170
Ek-Rodríguez, I. L., Navarrete-Segueda, A., Siebe, C., Meave, J. A., Vela-Correa, G. e Ibarra- Manríquez, G. (2025). Relief, soil and tree community attributes jointly shape liana community structure and diversity in a Neotropical rainforest landscape. Catena, 255, 109017. https://doi.org/10.1016/j.catena.2025.109017
Espejo-Serna, A. y López-Ferrari, A. R. (2018). La familia Bromeliaceae en México. Botanical Sciences, 96, 533–554. https://doi.org/10.17129/botsci.1918
Espejo-Serna, A., López-Ferrari, A. R. y Ramírez-Morillo, I. (2005). Bromeliaceae. Flora de Veracruz. Xalapa: Instituto de Ecología, A.C. Flora de Veracruz. https://doi.org/10.21829/fv.343.2005.136
Fragoso-Martínez, I. y Castillo-Campos, G. (2023). Passifloraceae. Flora de Veracruz. Xalapa: Instituto de Ecología, A.C. Flora de Veracruz. https://doi.org/10.21829/fv.585.20
23.200
Ferrufino-Acosta, L. (2010). Taxonomic revision of the genus Smilax (Smilacaceae) in Central America and the Caribbean Islands. Willdenowia, 40, 227–280. https://doi.org/10.3372/wi.40.40208
Fuster, F., Kaiser-Bunbury, C., Olesen, J. M. y Traveset, A. (2018). Global patterns of the double mutualism pheno-
menon. Ecography, 42, 826–835. https://doi.org/10.1111/ecog.04008
Fryxell, P. A. (1992). Malvaceae. Flora de Veracruz. Xalapa: Instituto de Ecología, A.C. Flora de Veracruz. https://doi.org/10.21829/fv.420.1992.68
García-Mendoza, A. e Ibarra-Manríquez, G. (1991). A new species of Costus (Costoideae, Zingiberaceae) from Veracruz, Mexico. Annals of the Missouri Botanical Garden, 78, 1081–1084. https://doi.org/10.2307/2399745
González-Elizondo, M. S., Reznicek, A. A. y Tena-Flores, J. A. (2018). Cyperaceae in Mexico: diversity and distribution. Botanical Sciences, 96, 305–331. https://doi.org/10.17129/botsci.1870
Gutiérrez-García, G. y Martin, R. (2011). Climate and climate change in the region of Los Tuxtlas (Veracruz, Mexico): a statistical analysis. Atmósfera, 24, 347–373.
Henderson, A. (2011). A revision of Geonoma (Arecaceae). Phytotaxa, 17, 1–271. https://doi.org/10.11646/phytotaxa.
17.1.1
Hernández-Esquivel, K., Piedra-Malagón, E. M., Cornejo-Tenorio, G., Mendoza-Cuenca, L., González-Rodríguez, A., Ruiz-Sánchez, E. et al. (2020). Unraveling the extreme morphological variation in the neotropical Ficus aurea complex (subg. Spherosuke, sect. Americanae, Moraceae). Journal of Systematics and Evolution, 58, 263–281. https://doi.org/10.1111/jse.12564
Ibarra-Manríquez, G. (1988). The palms of a tropical rain forest in Veracruz, Mexico. Principes, 32, 147–155.
Ibarra-Manríquez, G. (2017). Avances en la investigación botánica en la Estación de Biología Tropical Los Tuxtlas ¿Qué hacer ahora? En V. H. Reynoso, R. I. Coates y C. M. L. Vázquez (Eds.), Avances y perspectivas en la investigación de los bosques tropicales y sus alrededores: la región de Los Tuxtlas (pp. 101–116). Ciudad de México: Instituto de Biología, Universidad Nacional Autónoma de México.
Ibarra-Manríquez, G., Cornejo-Tenorio, G., González-Castañeda, N., Piedra-Malagón, E. M. y Luna, A. (2012). El género Ficus L. (Moraceae) en México. Botanical
Sciences, 90, 389–452. https://doi.org/10.17129/botsci.472
Ibarra-Manríquez, G., Cornejo-Tenorio, G., Hernández-Esquivel, K. B., Rojas-López, M., Sánchez-Sánchez, L. (2021). Vegetación y flora vascular del ejido Llano de Ojo de Agua, Depresión del Balsas, municipio de Churumuco, Michoacán, México. Revista Mexicana de Biodiversidad, 92, e923482. https://doi.org/10.22201/ib.20078706e.2021.92.3482
Ibarra-Manríquez, G., Martínez-Morales, M. y Cornejo-Tenorio, G. (2015). Frutos y semillas del bosque tropical perennifolio. región de Los Tuxtlas. Veracruz. Ciudad de México: Comisión Nacional para el Conocimiento y Uso de la Biodiversidad.
Ibarra-Manríquez, G., Martínez-Ramos, M., Dirzo, M. y Núñez-Farfán, J. (1997). La Vegetación. En S. E. Gónzález, R. Dirzo y R. C. Vogt (Eds.), Historia natural de Los Tuxtlas (pp. 61–85). Ciudad de México: Universidad Nacional Autónoma de México.
Ibarra-Manríquez, G., Ricker, M., Ángeles, G., Sinaca-Colín, S. y Sinaca-Colín, M. A. (1997). Useful plants of the Los Tuxtlas rain forest (Veracruz, Mexico): considerations of their market potential. Economic Botany, 51, 362–376. https://doi.org/10.1007/BF02861046
Ibarra-Manríquez, G. y Sinaca-Colín, S. (1987). Lista florística de la Estación de Biología Tropical Los Tuxtlas. Listados Florísticos de México VII. México D.F.: Instituto de Biología, Universidad Nacional Autónoma de México.
Ibarra-Manríquez, G. y Sinaca-Colín, S. (1995). Lista florís-
tica comentada de la Estación de Biología Tropical “Los Tuxtlas”, Veracruz, México. Revista de Biología Tropical, 43, 75–115.
Ibarra-Manríquez, G. y Sinaca-Colín, S. (1996a). Lista florística comentada de la Estación de Biología Tropical Los Tuxtlas, Veracruz. México: Mimosaceae a Verbenaceae. Revista de Biología Tropical, 44, 41–60.
Ibarra-Manríquez, G. y Sinaca-Colín, S. (1996b). Lista comentada de plantas de la Estación de Biología Tropical Los Tuxtlas, Veracruz, México (Violaceae-Zingiberaceae). Revista de Biología Tropical, 44, 427–447.
Islas-Hernández, C. S. y Alvarado-Cárdenas, L. O. (2023). Sinopsis del género Spigelia (Loganiaceae) en Norteamérica, Centroamérica y el Caribe. Acta Botanica Mexicana, 130, e2202. https://doi.org/10.21829/abm130.2023.2202
Krömer, T., Acebey, A. R., Armenta-Montero, S. y Croat, T. B. (2019). Diversity, distribution, and conservation status of Araceae in the state of Veracruz, Mexico. Annals of the Missouri Botanical Garden, 104, 10–32. https://doi.org/10.3417/2018214
Krömer, T., Espejo-Serna, A., López-Ferrari A. R., Acebey, A. R., García-Cruz, J. y Mathieu, G. (2020). Las angiospermas epífitas del estado de Veracruz, México: diversidad y distribución. Revista Mexicana de Biodiversidad, 91, e913
415. https://doi.org/10.22201/ib.20078706e.2020.91.3415
Linares-Palomino, R., Cardona, V., Hennig, E. I., Hensen, I., Hoffmann, D., Lendzion, J. et al. (2009). Non-woody life-form contribution to vascular plant species richness in a tropical American forest. Plant Ecology, 201, 87–99. https://doi.org/10.1007/s11258-008-9505-z
Liu, Y., Borregaard, M. K., Xu, X., Dimitrov, D., Pellissier, L., Shrestha, N. et al. (2023). An updated floristic map of the world. Nature Communications, 14, 2990. https://doi.org/10.1038/s41467-023-38375-y
Lombardi, J. A. (2014). Celastraceae (Hippocrateoideae e Salacioideae). Flora Neotropica, 114, 1–227.
Lorea-Hernández, F. G. (2002). La familia Lauraceae en el sur de México: diversidad, distribución y estado de conservación. Botanical Sciences, 71, 59–70. https://doi.org/10.17129/botsci.1663
Lorence, D. H. y Nee, M. (1987). Randia retroflexa (Rubiaceae), a new species from southern Mexico. Brittonia, 39, 371–375. https://doi.org/10.2307/2807136
Lutz, J. A., Furniss, T. J., Johnson, D. J., Davies, S. J., Allen, D., Alonso, A. et al. (2018). Global importance of large-diameter trees. Global Ecology and Biogeography, 27, 849–864. https://doi.org/10.1111/geb.12747
Martin-Del Pozzo, A. L. (1997). Geología. En E. Gónzález, R. Dirzo y R. C. Vogt (Eds.). Historia natural de Los Tuxtlas (pp. 25–31). Ciudad de México: Universidad Nacional Autónoma de México.
Martínez-Gordillo, M., Bedolla-García, B., Cornejo-Tenorio, G., Fragoso-Martínez, I., García-Peña, M. R., González-Gallegos, J. G. et al. (2017). Lamiaceae de México. Botanical Sciences, 95, 780–806. https://doi.org/10.17129/botsci.1871
Meave, J. A., Rincón-Gutiérrez, A., Ibarra-Manríquez, G., Gallardo-Hernández, C., Romero-Romero, M. A. (2017). Checklist of the vascular flora of a portion of the hyper-humid region of La Chinantla, Northern Oaxaca Range, Mexico. Botanical Sciences, 95, 722–759. https://doi.org/10.17129/botsci.1812
Miranda, F. y Hernández, E. (1963). Los tipos de vegetación de México y su descripción. Botanical Sciences, 28, 29–179. https://doi.org/10.17129/botsci.1084
Miranda-Gallegos, K. V., Navarrete-Segueda, A., Cortés-Flores, J., González-Arqueros, M. L., Acosta-Pérez, E. E. e Ibarra-Manríquez, G. (2023). Landscape heterogeneity drives spatial distribution of palm community in a Neotropical rainforest reserve affected by defaunation. Botanical Sciences, 101, 654–669. https://doi.org/10.17129/botsci.3204
Monro, A. K. (2009). Two new species and a nomenclatural synopsis of Myriocarpa (Urticaceae) from Mesoamerica. Novon, 19, 85–95. https://doi.org/10.3417/2006211
Morales, J. F. (1997). A synopsis of the genus Prestonia (Apocynaceae) section Tomentosae in Mesoamérica. Novon, 7, 59–66. https://doi.org/10.2307/3392074
Moreno, N. P. (1984). Glosario botánico ilustrado. México D.F.: Compañía Editorial Continental, S.A. de C.V.
Navarrete-Segueda, A., Cortés-Flores, J., Cornejo-Tenorio, G., González-Arqueros, L., Torres-García, M. e Ibarra-Manríquez, G. (2021). Timber and non-timber forest products in the northernmost Neotropical rainforest: ecological factors unravel their landscape distribution. Journal of Environmental Management, 279, 111819. https://doi.org/10.1016/j.jenvman.2020.111819
Ollerton, J. (2017). Pollinator diversity: distribution, ecological function, and conservation. Annual Review of Ecology, Evolution and Systematics, 48, 353–376. https://doi.org/10.1146/annurev-ecolsys-110316-022919
Ortiz-Rodríguez, A. E. (2022). Naming the long-known: a new species of Desmopsis (Annonaceae) endemic to Mexico. Acta Botanica Mexicana, 129, e2110. https://doi.org/10.21829/abm129.2022.2110
Ortiz-Rodríguez, A. E., Nge, F. J., Rodrígues‑Vaz, C., Soulé, V., Schatz, G. E., Martínez‑Velarde, M. F. et al. (2024). Taxonomy, systematics and conservation of the highly threatened and endemic Mexican genus Tridimeris (Annonaceae). Plant Systematics and Evolution, 310, 47. https://doi.org/10.1007/s00606-024-01929-8
Palacios-Wassenaar O. M. y Castillo-Campos, G. (2019). Erythroxylaceae. Flora de Veracruz. Xalapa. Instituto de Ecología, A.C. https://doi.org/10.21829/fv.43.2019.182
Palacios-Wassenaar O. M. y Castillo-Campos, G. (2020). Elaeocarpaceae. Flora de Veracruz. Xalapa. Instituto de Ecología, A.C. https://doi.org/10.21829/fv.489.2020.185
Pennington, T. D. (1997). The genus Inga: botany. Belgium: The Royal Botanic Gardens, Kew. Continental Printing.
Popma, J., Bongers, F. y Meave-del Castillo, J. (1988). Patterns in the vertical structure of the tropical lowland rain forest of Los Tuxtlas, Mexico. Vegetatio, 74, 81–91. https://doi.org/10.1007/BF00045615
POWO (2025). Plants of the World Online. Facilitated by the Royal Botanic Gardens, Kew. Recuperado 13 abril, 2025, de: https://powo.science.kew.org/
PPG (The Pteridophyte Phylogeny Group) 1. (2016). A community-derived classification for extant lycophytes and ferns. Journal of Systematics and Evolution, 54,
563–603. https://doi.org/10.1111/jse.12229
Ramamoorthy, T. P. y Uribe, Y. H. (1988). A new name and a new species in Mexican Ruellia (Acanthaceae). Plant Systematic and Evolution, 159, 161–163. https://doi.org/10.1007/BF00935968
Ramírez-Roa, A. e Ibarra-Manríquez, G. (1998). A new species of Solenophora (Gesneriaceae) from southeast Veracruz, Mexico. Novon, 7, 281–284. https://doi.org/10.2307/3391945
Ramírez-Roa, A., Paniagua-Ibáñez, M. y Mora-Jarvio, M. A. (2023). Gesneriaceae. Flora de Veracruz. Xalapa: Instituto de Ecología, A.C. https://doi.org/10.21829/fv.579.2023.199
Raven, P. H., Gereau, R. E., Phillipson, P. B., Chatelain, C., Jenkins, C. N. y Ulloa, U. C. (2020). The distribution of biodiversity richness in the tropics. Science Advances, 6, eabc6228. https://doi.org/10.1126/sciadv.abc6228
Reynoso, V. H., Coates, R. I. y Vázquez, C. M. L. (Eds.). (2017). Avances y perspectivas en la investigación de los bosques tropicales y sus alrededores: la región de Los Tuxtlas. Ciudad de México: Instituto de Biología, Universidad Nacional Autónoma de México.
Riba, R. (1989). A new species of Thelypteris subg. Goniopteris from the state of Veracruz, Mexico. American Fern Journal, 79, 122–124. https://doi.org/10.2307/1547294
Rojas-Martínez, C. y Flores-Olvera, H. (2019). Florística de la sierra El Pelado, Acatlán, Puebla, México. Revista Mexicana de Biodiversidad, 90, e902694. https://doi.org/10.22201/ib.20078706e.2019.90.2694
Rowher, J. G. (1993). Lauraceae: Nectandra. Flora Neotropica, 60, 1https://doi.org/10.17129/botsci.3673332.
Ruiz, G. M. G., Gómez D. J. A. y Krömer, T. (2025). Las plantas vasculares endémicas de Veracruz: análisis de 30 años de conocimiento. Botanical Sciences, 103, 876–898. https://doi.org/10.17129/botsci.3673
Rzedowski, J. (1978). Vegetación de México. México D.F.: Limusa.
Rzedowski, J. (1991a). El endemismo en la flora fanerogámica mexicana: una apreciación analítica preliminar. Acta Botanica Mexicana,15, 47–64. https://doi.org/10.21829/abm15.1991.620
Rzedowski, J. (1991b). Diversidad y orígenes de la flora fanerogámica de México. Acta Botanica Mexicana, 14, 3–21. https://doi.org/10.21829/abm14.1991.611
Salazar, G. A. (1988). Mormodes tuxtlensis, nueva especie de Veracruz, México. Orquídea (Méx.), 11, 51–62.
Salino, A., Almeida, T. E. y Smith, A. R. (2015). New combinations in Neotropical Thelypteridaceae. Phytokeys, 57, 11–50. https://doi.org/10.3897/phytokeys.57.5641
Sánchez-Garfias, B., Ibarra-Manríquez, G. y González-García, L. (1991). Manual de identificación de frutos y semillas anemócoros de árboles y lianas de la Estación “Los Tuxtlas”, Veracruz, México. Cuadernos 12. México D.F.: Instituto de Biología, Universidad Nacional Autónoma de México.
Sarukhán, J., Urquiza-Haas, T., Koleff, P., Carabias, J., Dirzo, R., Ezcurra, E. et al. (2015). Strategic actions to value, conserve, and restore the natural capital of megadiversity countries: the case of Mexico. BioScience, 65, 164–173. https://doi.org/10.1093/biosci/biu195
Smith, A. R. y Tejero-Díez, J. D. (2014). Pleopeltis (Polypodiaceae), a redefinition of the genus and nomenclatural novelties. Botanical Sciences, 92, 43–58. https://doi.org/10.17129/botsci.29
Soto, M. y Gama, L. (1997). Climas. En E. González, R. Dirzo y R. C. Vogt (Eds.), Historia natural de Los Tuxtlas (pp. 7–23). México D.F.: Universidad Nacional Autónoma de México.
Sousa, S. M. (2009). Adiciones al género Inga (Ingeae, Mimosoideae, Leguminosae) para la Flora Mesoamericana. Acta Botanica Mexicana, 89,25–41. https://doi.org/10.21829/abm89.2009.303
Sousa, S. M. (2010). Revisión del complejo de Lonchocarpus cruentus (Leguminosae: Papilionoideae: Millettieae), con descripciónes de cinco especies nuevas. Brittonia, 62, 321–336. https://doi.org/10.1007/s12228-009-9116-6
Sousa, S. M. y Rudd, V. E. (1993). Revision del género Styphnolobium (Leguminosae: Papilionoideae: Sophoreae). Annals of the Missouri Botanical Garden, 80, 270–283. https://doi.org/10.2307/2399827
Spicer, M. E., Mellor, H. y Carson, A. P. (2020). Seeing beyond the trees: a comparison of tropical and temperate plant growth forms and their vertical distribution. Ecology, 101, e02974. https://doi.org/10.1002/ecy.2974
Swenson, U., Lepschi, B., Lowry, P. P. II, Terra-Araujo, M. H., Santos, K., Nylinder, S. et al. (2023). Reassessment of generic boundaries in Neotropical Chrysophylloideae (Sapotaceae): eleven reinstated genera and narrowed circumscriptions of Chrysophyllum and Pouteria. Taxon, 72, 307–359. https://doi.org/10.1002/tax.12894
Torres-Montúfar, A. y Ochoterena, H. (2013). Dos especies nuevas de la familia Rubiaceae de la sierra de los Tuxtlas, Veracruz, México. Revista Mexicana de Biodiversidad, 84, 1082–1089. https://doi.org/10.7550/rmb.32503
Torres-Montúfar, A. y Torres-Díaz, A. N. (2022). Las Rubiáceas de México: ¿Ya está hecho el trabajo? Botanical Sciences, 100, 446–468. https://doi.org/10.17129/botsci.2847
Tropicos. (2025). Tropicos.org. Missouri Botanical Garden. Recuperado 03 mayo, 2025, de: https://tropicos.org
Ulloa, U. C., Acevedo-Rodríguez, P., Beck, S., Belgrano, M. J., Bernal, R., Berry, P. E. et al. (2017). An integrated assessment of the vascular plant species of the Americas. Science, 358, 1614–1617. https://doi.org/10.1126/science.aao0398
Vázquez, T. M., Armenta, M. S., Campos, J. J. y Carvajal, H. C. I. (2010). Árboles de la región de Los Tuxtlas. Veracruz: Comisión Organizadora del Estado de Veracruz de Ignacio de la Llave para la Conmemoración del Bicentenario de la Independencia Nacional y del Centenario de la Revolución Mexicana/ Secretaría de Educación-Gobierno de Veracruz.
Vera-Caletti, P. y Wendt, T. (2001). Una nueva especie de Calatola (Icacinaceae) de México y Centroamérica. Acta Botanica Mexicana, 54, 39–49. https://doi.org/10.21829/abm54.2001.867
Vergara-Rodríguez, D., Mathieu, G., Samain, M. S., Armenta-Montero, S. y Krömer, T. (2017). Diversity, distribution, and conservation status of Peperomia (Piperaceae) in the state of Veracruz, Mexico. Tropical Conservation Science, 10, 1–28. https://doi.org/10.1177/1940082917702383
Villareal, Q. J. A. (2003). Sambucaceae. Flora de Veracruz. Xalapa: Instituto de Ecología, A.C./ Universidad de California, Riverside. https://doi.org/10.21829/fv.350.2003.
129
Villaseñor, J. L. (2016). Checklist of the native vascular plants of Mexico. Revista Mexicana de Biodiversidad, 87, 559–902. https://doi.org/10.1016/j.rmb.2016.06.017
Villaseñor, J. L. y Meave, J. A. (2022). Floristics in Mexico today: insights into a better understanding of biodiversity in a megadiverse country. Botanical Sciences, 100, S14–
S33. https://doi.org/10.17129/botsci.3050
Villaseñor, J. L., Meave, J. A., Ortiz, E. e Ibarra-Manríquez, G. (2003). Biogeografía y conservación de los bosques tropicales húmedos de México. En J. J. Morrone y J. Llorente (Eds.), Una perspectiva latinoamericana de la biogeografía (pp. 209–216). México D.F.: Comisión Nacional para el Conocimiento y Uso de la Biodiversidad/ Universidad Nacional Autónoma de México.
Villaseñor, J. L. y Ortiz, E. (2014). Biodiversidad de las plantas con flores (División Magnoliophyta) en México. Revista Mexicana de Biodiversidad, 85 (Suplem.), S134–S142. https://doi.org/10.7550/rmb.31987
Villaseñor, J. L. y Ortiz, E. (2025). Floristic richness comparison among the Mexican states. Revista Mexicana de Biodiversidad, 96, e965505. https://doi.org/10.22201/ib.20078706e.2025.96.5505
Villaseñor, J. L., Ortiz, E. y Campos-Villanueva, A. (2018). High richness of vascular plants in the tropical Los Tuxtlas Region, Mexico. Tropical Conservation Science, 11, 1–12. https://doi.org/10.1177/1940082918764259
von Thaden, J. J., Laborde, J., Guevara, S. y Mokondoko-Delgadillo, P. (2020). Dinámica de los cambios en el uso del suelo y cobertura vegetal en la Reserva de la Biosfera Los Tuxtlas (2006-2016). Revista Mexicana de Biodiversidad, 91, e913190. https://doi.org/10.22201/ib.20078706e.2020.91.3190
Wendt, T. (1993). Composition floristic affinities and origins of the canopy tree flora of the Mexican Atlantic slope rain forest. En T. P. Ramamoorthy, R. Bye, A. Lot y J. Fa (Eds.), Biological diversity of Mexico: origins and distribution (pp. 595–680). Nueva York: Oxford University Press.
Zamora, N. A. (2006). Two new species of Ormosia (Leguminosae – Papilionoideae, Sophoreae) from Mesoamérica. Edinburgh Journal of Botany, 63, 183–190. https://doi.org/10.1017/S0960428606000552
Zhou, X. M. y Zhang, L. B. (2023). Phylogeny, character evolution, and classification of Selaginellaceae (lyco-phytes). Plant Diversity, 45, 630e684. https://doi.org/
10.1016/j.pld.2023.07.003

Citizen science suggests decreased diversity of insects in Mexico, a megadiverse country
Datos de ciencia ciudadana sugieren un decremento en la diversidad de insectos de México, país megadiverso
Jorge Soberon *
University of Kansas, Biodiversity Institute, Dyche Hall, 1345 Jayhawk Blvd, Lawrence, KS 66045, USA
*Corresponding author: jsoberon@ku.edu (J. Soberon)
Received: 09 July 2025; accepted: 26 August 2025
Abstract
An analysis of iNaturalist data on several taxonomic groups of insects in Mexico is presented. A decreasing trend was observed in species diversity per year for 4 families of butterflies, bumblebees, and dragonflies and damselflies. Analyses were performed on several potential vegetation types (sensu Rzedowsky), and the roles of deforestation and pesticide use on the identified trends were explored. Challenges in using unsystematic data to estimate trends are discussed, and several hypotheses are provided to explain the results.
Keywords: Butterflies; Bumblebees; Damselflies; Dragonflies, iNaturalist
Resumen
Se presenta un análisis de datos de iNaturalist sobre varios grupos taxonómicos de insectos en México. Se observó una tendencia decreciente en la diversidad de especies por año para 4 familias de mariposas, y para abejorros y libélulas. Se realizaron análisis sobre varios tipos potenciales de vegetación (sensu Rzedowsky) y se exploró el papel de la deforestación y el uso de pesticidas en las tendencias identificadas. Se discuten los desafíos del uso de datos no sistemáticos para estimar tendencias y se presentan varias hipótesis para explicar los resultados.
Palabras clave: Mariposas, Abejorros; Caballitos del diablo; Libélulas; NaturaLista
Introduction
Evidence indicates a decrease in insect populations in many countries (Edwards et al., 2025; Hallmann et al., 2017). This finding is worrisome for many reasons, including that insects are key components of ecosystems and provide societies with important ecosystem services, such as pollination (Potts et al., 2010). Moreover, insects have substantial but largely unappreciated cultural importance (Duffus et al., 2021), not only worldwide but particularly in countries such as Mexico, where insects have culinary uses (Ramos-Elorduy & Viejo-Montesinos, 2007), have been important for ancestral cultures (Beutelspacher, 1989), and have economic and societal value (Ayala et al., 2012; Rogel-Fajardo et al., 2011).
Most detailed evidence of the decline in insects has come from countries in temperate zones that have developed formal monitoring schemes (Streitberger et al., 2024; Thomas, 2005). In contrast, tropical regions are less well studied (Sánchez-Herrera et al., 2024), and the existing evidence is contradictory (Bonadies et al., 2024; Boyle et al., 2025; Wagner et al., 2021). For instance, studies on Hemiptera (Lucas et al., 2016) and on saturniid moths (Basset et al., 2017) have indicated no trends in Barro Colorado Island, Panama. Similarly, in Veracruz, Mexico, well monitored fruit flies have shown no trends (Aluja et al., 2012; Ordano et al., 2013). In contrast, decreases in saturniid larvae have been reported in Costa Rica (Salcido et al., 2020), and declines in arthropod biomass have been reported in Puerto Rico and, on the basis of a few data points, in Chamela, Mexico (Lister & García, 2018). The monarch butterfly, perhaps the best monitored insect species in Mexico, has shown consistent decreases in its wintering aggregations (Thogmartin et al., 2017; Vidal & Rendón-Salinas, 2014; Zylstra et al., 2021).
Because of its history, climate, topography, and cultural milieu (Ramamoorthy et al., 1993), Mexico is among the world’s megadiverse countries (Mittermeier et al., 1997). Therefore, assessing the trends in insect populations in Mexico should be prioritized. Unfortunately, long-term insect monitoring in Mexico is rare. Although Mexico has a long history of entomological research, including many collections and hundreds of publications (Michán & Llorente, 2002), monitoring has been limited to only a few species. Although the reasons for the lack of national monitoring schemes like those existing in other countries should be determined, this study does not attempt to do so. It takes as a premise that, in Mexico, just a few systematically obtained insect time series of more than 2-3 years long are available. This study is aimed at estimating insect biodiversity trends in Mexico, despite the absence of systematic monitoring efforts.
Systematic monitoring results are compiled in several worldwide time series databases, such as the Living Planet Index (Almond et al., 2020) and the Global Population Dynamics database (NERC Centre for Population Biology, 1999). Unfortunately, these databases have sparse insect information and contain no data for Mexico. Another possibility is using so-called citizen science (CS) data (Cohn, 2008), which, although opportunistic and unsystematic, is often abundant. Data on insects collected by non-professionals have been used to estimate phenology and distributions (Soroye et al., 2018). However, using such data to estimate population trends is challenging, as discussed below.
In Mexico, perhaps the most comprehensive CS initiative is iNaturalist (known as Naturalista in Mexico). iNaturalist began its operations in Mexico in 2008, although in 2013 the initiative came under the leadership of the national biodiversity agency, Conabio, under the name of Naturalista (Macías & Freire, 2017) and obtained funding from the Slim Foundation. Therefore, in Mexico, iNaturalist began in earnest in 2013. Despite this relatively late start, Mexico is the third country in amount of data (Mason et al., 2025) and it contains more than 80,000 records tagged as “research level” for 3 families of butterflies, and the damselflies, dragonflies, and bumblebees. This substantial information may be used to assess trends. However, CS data must be corrected for biases, of which are many (Crall et al., 2011). Specifically, in Mexico, the number of observers (and thus of observations) in iNaturalist increases each year (Table S1), and this bias should be considered when using such data.
Indeed, a major problem in using opportunistic CS data to estimate trends is correcting for biases in recording efforts (Di Cecco et al., 2021). Several methods can be used to address this problem (Isaac et al., 2014; Outhwaite, 2019; Tang et al., 2021). One of the simplest methods is correcting bias by obtaining the quotient of the metric used to report biodiversity to some measure of the effort invested in a locality, for a given period. What is “effort,” and how can it be measured in iNaturalist data? Collection effort is difficult to define but can be described in terms of: 1) the time spent collecting, 2) the method of collection and number of collectors, or 3) the number of specimens or species observed (Gulland, 1969; Willott, 2001). iNaturalist data allows for extraction of a measure of time (number of monthly observations in a year), but data quality (beyond the “research” tag, which refers to the reliability of the name assigned to the species), remains unreported, and worse, in the case of iNaturalist, this quality is known to change (Di Cecco et al., 2021). Di Cecco (2021) has suggested that, in iNaturalist, observers with at least 2 observations are more reliable than those with just 1 observation. Therefore, as a measure of effort, this study used the number of observers with 2 or more observations. As biodiversity measures it is used the number of species, and the number of observations, pooled for spatial units and year. Two indices are then calculated: number of species/effort, and number of records/effort.
Ordinary regressions of metrics against time often experience problems of autocorrelated errors and non-equal variances (heteroskedasticity). These are characteristic of time series (Shumway & Stoffer, 2005) and must be accounted for. One method of addressing the complexities of analysis of count time series data is using a package such as “trim” (in the R platform), which assumes a Poisson model for the underlying data (Pannekoek, 1998). This approach corrects for the autocorrelation of errors and for heteroskedasticity. Trim has frequently been used for European (van Strien et al., 2019) and tropical American (Novoyny & Basset, 2000) data, but the key assumption of count data (a discrete scale) complicates analysis of continuous-scale indices, or data including many non-occurrences, because the software is sensitive to the presence of zeroes, or NAs, in the data.
Another possibility is estimating whether a significant trend exists in the data, by using a non-parametric Mann-Kendall test (Lyubchich et al., 2013). An ordinary least squares linear regression (OLS) of metric against time is first performed, and the existence of trends (linear or monotonic) is subsequently determined. This method uses the sign of the slope in the OLS, and the significance is tested with the Mann-Kendall test.
Additional methods can be used, such as, for a single species, the logit of the probability of occupancy of a cell, on a time unit (van Strien et al., 2019) and fit a generalized linear model of predictors, by using the length of the list of species as a measure of effort (Szabo et al., 2010). Then several single species regressions can be combined in an index (van Strien et al., 2019). One problem with this approach is that generalized linear modeling is based on an assumption of independence of errors, which might be violated in a time series.
A statistically more sophisticated modification of the above idea is reporting the proportion of occupied sites under a hierarchical model that separates the actual presence from the act of observation (Outhwaite, 2019). Although apparently very rigorous, this approach has its own problems, including the need to define an appropriate model for the “present” and “observer” components, and the need to have replicated visits to the same site within the same season (van Strien et al., 2019).
Another possibility is using generalized least squares (GLS) regressions, which allow for autocorrelated errors and heteroskedasticity. The R package “nlme” implements this technique. This method can fit an ordinary regression of the index against covariates such as time, and another regression including autocorrelation with power variance decay in its model. Subsequently, the 2 models can be compared with the Akaike criterion, and the best model can be retained. This option was used here, with the simplest ARIMA model with lag = 1 as a model of correlated errors.
This study further assessed whether any existing trends might have differed for different ecological regions of Mexico. A variety of subdivisions of Mexico have been suggested, according to different ecological perspectives, at different spatial resolutions (Anonymous, 1997; Challenger & Soberón, 2012; Miranda & Hernández, 1963; Olson et al., 2001). Here, Rzedowsky’s potential vegetation types were used (Rzedowsky, 1986). Although coarse-grained, these types are based primarily on straightforward floristic criteria, are well known in Mexico, and have a small number of categories.
An important caveat in using CS data is that species that are difficult to identify by sight should be avoided. This work focused on 3 families of butterflies (Papilionidae, Pieridae, and Nymphalidae), with 316 names (skippers and the smallest families in the Papilionoidea were excluded); 23 names for bumblebees; and 293 names for the Odonata (both Zygoptera and Anysoptera). In addition, as a comparison, data on 307 names for Solanaceae were included. The numbers of names (without proper taxonomic validation by experts), as reported by iNaturalist, are listed in Table 1.
For the butterflies, although using species identified as indicators of “conservation” status (Orta et al., 2022) would have been interesting, most species identified by these authors as indicators had only a few records in the iNaturalist database. Therefore, the analysis was performed not by individual species, but by pooling all the data in the 3 families of butterflies, all the dragon and damselflies, and all the bumblebees.
For obtaining uncertainty bands, grids of hexagons covering the territory of Mexico were defined at several resolutions (Fig. 1). For a given year and taxonomic group, the means and variances over hexagons were determined. Each unique combination of year and hexagon defined an “event,” and thus the abundance metrics were: 1) the number of observations per event (cumulative monthly observations); and 2) the number of different species per event. As a measure of effort, the total number of different observers with at least 2 observations in each “event” was used. The final index was the average over all hexagons with at least 1 record, for a given year, of the number of observations or the number of species, per observer.
Table 1
Numbers of scientific names for the different taxonomic groups in the 4 most visited potential vegetation classes. The butterflies are the Papilionidae (swallowtails), Pieridae (sulfurs), and Nymphalidae (brushfoots).
| All Mexico | Xerophytic Shrub | Pine Oak Forest | Grasslands | Tropical Deciduous Forest | |
| Butterflies | 413 | 153 | 220 | 220 | 198 |
| Odonata | 292 | 185 | 187 | 105 | 184 |
| Bombus | 23 | 16 | 20 | 9 | 14 |
| Solanaceae | 307 | 176 | 210 | 111 | 153 |
Changing the hexagon area might potentially change the results. This problem, described as the “modifiable areal unit problem,” has been long known to geographers (Openshaw, 1984). Fortunately, in this case, the qualitative results were not affected by the resolution of the hexagons (data correlations among resolutions always exceeded 0.7). Consequently, only the analysis using the largest (2 degrees) hexagons (n = 81) is reported.
The literature has suggested that the decrease in insect abundance has been due to: 1) increased use of pesticides, 2) decreased habitat area (or increased transformed land area), and 3) climate change. At the scale of the whole country, regressions of data versus time series of pesticide use and deforestation rates are reported.

Materials and methods
CS data are not ideally suited to the estimation of trends, primarily because of the biased and uneven methods of sampling sites, times, and species. This work used 1 of the 3 methods proposed by Isaac et al. (2014): correcting the reported number of sightings according to a measure of effort. iNaturalist data were downloaded from the Global Biodiversity Information Network (GBIF), as detailed in Table 2.
Data were divided into subsets (keeping records with coordinates) for the 4 largest families in the Papilionoidea: Papilionidae (5,583 records), Pieridae (29,994 records), Nymphalidae (20,145 records), and Lycaenidae (2,326 records). The Lycaenidae was removed from the analysis because many species are relatively difficult to determine visually. Data for the genus Bombus (bumblebees, 6,543 records) and the 2 suborders of the Odonata (the Zygoptera, 12,772 records, and the Anisoptera, 24,003 records) were also downloaded. For comparison purposes, observations of the nightshade family, the Solanaceae (39,014 records), were downloaded. The number and positions of every observation in Mexico are presented in figure 2.
Table 2
Digital Object Identifiers (DOIs) from GBIF for the datasets used in the work.
| Taxon | GBIF DOI | iNaturalist Records | Unique names |
| Nymphalidae | doi.org/10.15468/dl.qta4zp | 20,145 | 223 |
| Papilionidae | doi.org/10.15468/dl.uu4unc | 5,583 | 13 |
| Pieridae | doi.org/10.15468/dl.zug4ee | 29,994 | 80 |
| Lycaenidae | doi.org/10.15468/dl.xgad6g | 2,326 | 97 |
| Odonata | doi.org/10.15468/dl.atdkfz | 36,775 | 292 |
| Anisoptera | 24,003 | 163 | |
| Zygoptera | 12,772 | 129 | |
| Bombus | doi.org/10.15468/dl.c6h4jz | 6,543 | 23 |
| Solanaceae | doi.org/10.15468/dl.597nj5 | 39,014 | 307 |
Data tagged as “research quality” in the downloaded GBIF data were retained, and basic data cleaning was performed to keep the coordinates inside Mexico. No attempt was made to correct for outdated taxonomy or other known issues present in aggregator data (Chapman, 2005).
Data can be organized as time series, by pooling the observations in a year. This method has a drawback of potentially missing seasonality; however, pooling by month produces tables that are too sparse and therefore are difficult to analyze. To include some measure of uncertainty in the trends, the averages of the calculated indices over all non-empty (i.e., with at least 1 observation) hexagons of 2 degrees of surface were determined, and its standard error calculated.
Two indices were used: different_species/observer and records/observer. The first is a measure of diversity, whereas the second is a measure of abundance. Findings for both are reported. “Observers” refers to the number of observers with at least 2 registered observations.
To summarize trends, a useful statistic may be the slope of a linear model of index as a function of time, which requires regressions of index vs. year. However, as previously discussed, the errors in many time series are not independent, and the equal variance assumption of ordinary least squares is also often violated. If uncorrec-
ted, these problems interfere with rigorous calculations of probability under a null hypothesis (McShane et al., 2019). Among the many methods for addressing this problem, generalized least squares regressions (Baillie & Kim, 2018), which enable inclusion of an autoregressive structure of correlations and violations of homoscedasticity, were chosen herein. Two models were fitted to the data: an ordinary linear least squares, and a first order auto-regressive, moving average model (ARIMA) (Shumway & Stoffer, 2005) allowing for heteroskedasticity. The 2 models were compared with an ANOVA (Fox & Weisberg, 2019), and the most likely model (based on the Akaike criterion; see Fox & Weisberg, 2019) was used. This process permitted to obtain, in a rigorous way, the probability for the observed slope values, under a null hypothesis of a slope equal to zero. Reporting the “significance” of slopes has been substantially criticized (McShane et al., 2019). Therefore, the probability (rather than the “significance”) of the slope, based on the assumption of a null model of no trend, is reported. Very small probabilities are highlighted.
The regressions included 2 possible causal factors: forest loss and use of pesticides. The deforestation rate was obtained from the Global Forest Watch website (Sims et al., 2024) with a threshold of 30% of forest cover, as recommended by Sims et al. (2024). This dataset has maintained methodological consistency (Hansen et al., 2013) and therefore is preferable to the INEGI Series (Gebhardt et al., 2015). Agrochemical use was determined as the amount of pesticides used per hectare of cropland, as reported on the FAO Web site. The data came from government reports https://www.fao.org/faostat/en/#data/RP. A discussion of the FAO dataset’s strengths and problems has been provided by Shattuck et al. (2023).
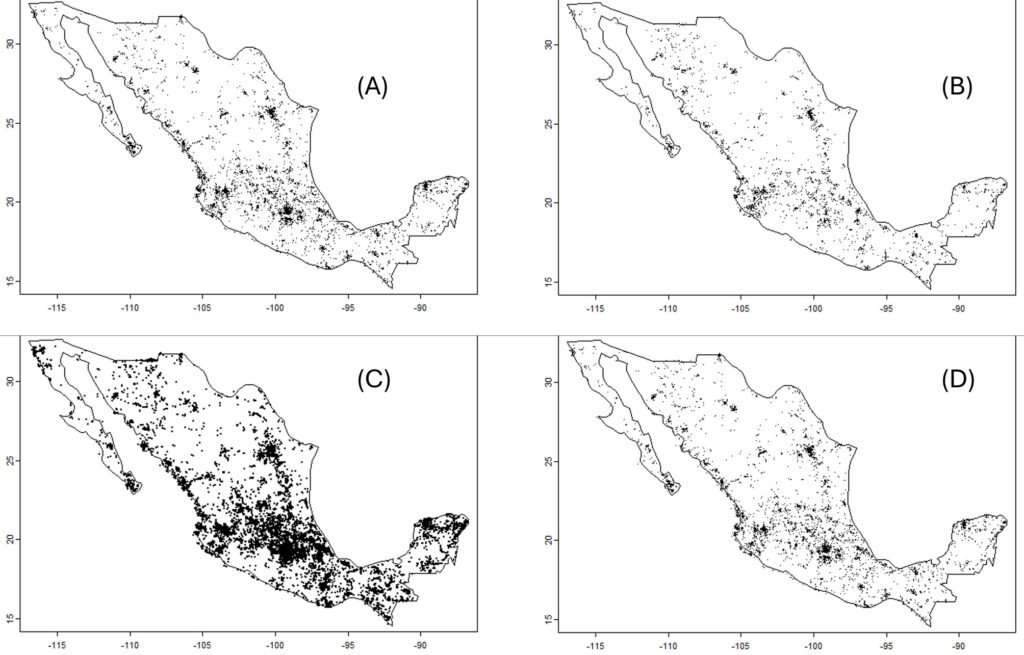
Because the probability of the observed values of the slope of the index of diversity per unit of effort vs. time, under a null hypothesis of 0 slope, was small in most cases, the regression was assumed to remove the time trend, and factors affecting just the residuals were searched for. That is, the residual of the index vs. time regressions was regressed against 2 predictors: deforestation rate and use of pesticides. The results are shown in the Supplementary materials.
To aggregate by “biome,” the subdivision of the Potential Vegetation of Mexico (Rzedowsky, 1986) was selected. A shapefile of Rzedowsky’s map at 1:4,000,000 scale, available at Conabio Geoportal, is produced by Instituto de Geografía, UNAM México. This map was used to pool the iNaturalist records according to potential vegetation, by using the 4 categories with the highest number of iNaturalist reports.
An informal survey was circulated among scientists working in 3 major ecology research centers in Mexico (INECOL, Veracruz, Instituto de Ecología, UNAM, and Ecosur, Chiapas). A total of 37 questionnaires were sent with Qualtrics. The questions are provided in the Supplementary materials. The main data tables and R code are openly available (Creative Commons CC0: 1) at https://github.com/jsoberon/iNaturalistInsectsMexico
Results
The informal questionnaire received 27 responses out of 37 requests. Among the respondents, 84% stated that they have observed a decrease in the number of insects either in streetlights in villages, or in the windshields or radiators of field vehicles. Although these answers lacked statistical rigor, they suggested a widespread perception among field biologists in Mexico that insect populations are becoming smaller.
The iNaturalist data provided a more nuanced picture. Before examining the trends in biodiversity indices, basic data were analyzed. Indeed, both the number of species and the number of observers (with more than 2 observations) increased (Fig. 3).
The numbers of observed species and observers both increase over time. The increased number of observers introduced an important bias in the data, given that more species (or more individuals) would reasonably be expected to be reported if more observers were present. However, although the diversity of insects appeared to be decreasing, the evidence of a decrease in abundance was unclear (Fig. 4; Tables 3, 4).
Diversity per unit effort appeared to decrease (Table 3). However, the trends in the abundance (observations/number observers) were either positive or indistinguishable from 0 (Table 4). Box plots of the slopes of the regressions for the 2 indices (species, and observations) are shown in figure 5.
The above results suggest that diversity is decreasing, but abundance is stable. This finding is inconsistent with the informal perceptions of field biologists (as indicated by the questionnaire), most of whom perceived diminished insect abundance. Among the few insect species whose abundance in Mexico has been monitored systematically, Danaus plexippus (monarch butterfly) populations are decreasing (Vidal & Rendón-Salinas, 2014; Zylstra et al., 2021), whereas Anastrepha fruit fly populations appear to be stable (Aluja et al., 2012; Ordano et al., 2013). Comparing these 2 cases is challenging, because monarch butterflies are affected by a variety of factors occurring on a continental scale, whereas fruit flies might be affected primarily by local factors.
Might the negative trend in diversity correlate with predictors often associated with insect loss? Forest cover, as measured via remote sensing over 15 years (Hansen et al., 2013), is decreasing in Mexico (Supplementary materials). Pesticide use per hectare of crop, as reported by the FAO, increased until 2018, when the FAO database indicated an abrupt decrease (Supplementary materials). The causes of this decrease, if real, are unknown; however, after the COVID-19 pandemic, Mexico’s primary sector experienced a marked decrease in activity (Sánchez et al., 2022), which may explain a drop in the use of agrochemicals. Regressions of the residuals of the diversity/effort vs. time models against 2 predictors, deforestation rate and pesticide use per hectare, were not associated with small probabilities of an H0 of 0 slope (Supplementary materials). Consequently, the data did not provide evidence that negative slopes in insect diversity were due to pesticide use or deforestation.
Finally, for the major taxonomic groups, the slopes of the generalized least squares, in the first 4 potential vegetation types according to Rzedowsky (1986) were most negative for bumblebees in tropical deciduous forest, followed by pine-oak forest and xerophytic shrub. For the butterflies, the most negative slope was in pine-oak forest, followed by tropical deciduous forest and xerophytic shrub (Supplementary materials: Table S3). In the case of the Solanaceae, a group included for comparison purposes, the slope is only negative in the grasslands vegetation type.
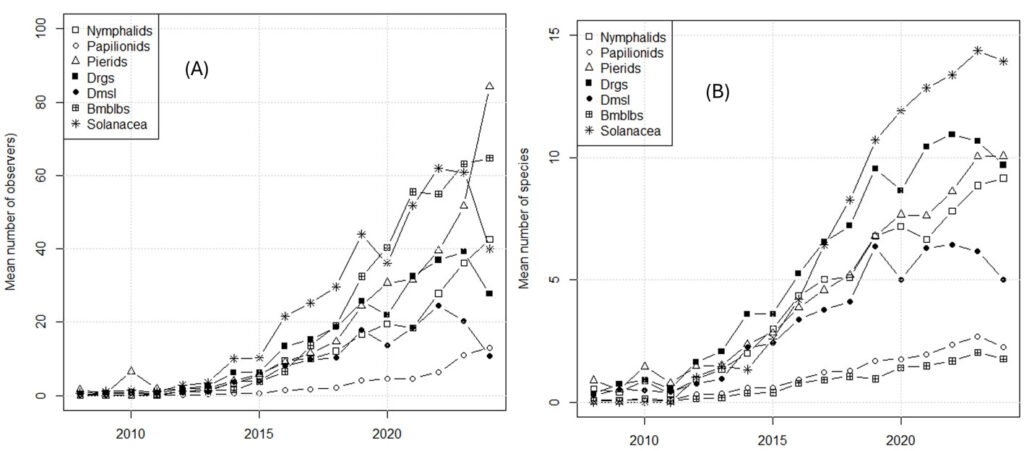
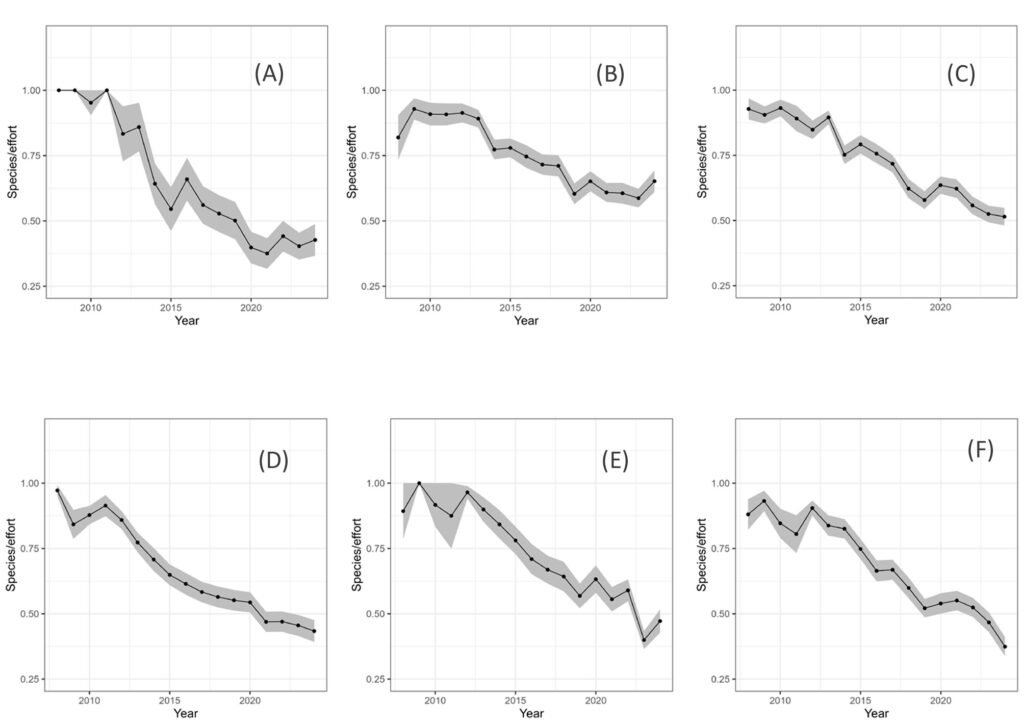
Table 3
Regression analysis (generalized least squares) of diversity/observer vs. time in the iNaturalist data, for the main taxonomic groups. The analysis was performed over the mean values in hexagons of 2 degrees of resolution. With the exception of the Zygoptera, for which the first order autoregressive model did not converge, the ordinary least squares regression did not significantly differ with respect to models with autocovariance and heteroskedasticity. Consequently, the table shows the slope of ordinary linear models of different_species/effort with respect to time. The probabilities of the obtained values under a null hypothesis of slope of zero were very small, with the exception of the dragonflies and swallowtails (Fig. 2).
| Taxon | Species, 2 degrees | |||
| Slope | p | Model | n | |
| Bombus | -0.0428 | 0.0000237 | OLS | 6,543 |
| Anisoptera | -0.0206 | 0.00155 | OLS | 24,003 |
| Zygoptera | 0.0003 | 0.942 | OLS_NO_CNV | 12,772 |
| Nymphalidae | -0.0372 | 0.000000242 | OLS | 20,145 |
| Papilionidae | -0.0108 | 0.104 | OLS | 5,583 |
| Pieridae | -0.0247 | 0.000202 | OLS | 29,994 |
Table 4
Regression analysis (generalized least squares) of records/observer vs. time in the iNaturalist data, for the main taxonomic groups. The data were averaged over hexagons of 2 degrees of resolution. Except for the Zygoptera, for which a first order autoregressive model was used, the ordinary least squares regression did not significantly differ with respect to models with autocovariance and heteroskedasticity. Consequently, the table shows the slope of ordinary linear regressions of number_of_records/effort with respect to time. Notably, every regression had a positive, low probability slope.
| Taxon | Slope | p | Model | n |
| Bombus | 0.0446 | 0.00000766 | OLS | 6,543 |
| Anisoptera | 0.0201 | 0.00649 | OLS | 24,003 |
| Zygoptera | 0.0366 | 6.86E-08 | ARIMA | 12,772 |
| Nymphalidae | 0.0089 | 0.034 | OLS | 20,145 |
| Papilionidae | 0.0669 | 0.00000176 | OLS | 5,583 |
| Pieridae | 0.0154 | 0.0000984 | OLS | 29,994 |

Discussion
The results show a tendency to decrease the number of species with time, for the insects, and a much less marked negative trend for the Solanaceae. This suggests that CS data does capture some sort of biological signal in the data. However, a diminishing trend of diversity, together with a stable pattern of abundance, are compatible with several hypotheses. One entirely biological hypothesis is that insect diversity, but not abundance, is decreasing. If the rarest species are disappearing, then the country is homogenizing (McKinney & Lockwood, 1999). Thus, Mexico’s highly diverse and unique insect biodiversity is slowly being replaced by a more homogeneous, more cosmopolitan set of species. This rather alarming possibility, supported by the CS data, must be more directly assessed in the field.
Another explanation for the observed negative trend in insect species numbers might be that, over time, observers have reached the asymptote of the total number of species available to be observed. Since the total number of species in any given area is probably roughly constant, with sufficient effort, no more than that constant number can be reported; however, if the number of observers is increasing, a negative trend in the index of species/observers would result. The total number of species in the database, for each taxonomic group, is shown in Table 1. The average number of species reported per hexagon was well below that total (Supplementary materials: Table S4), thus suggesting that a saturation effect was not present, and the results presented here indeed indicate a decreasing trend in insect diversity. This complex point is discussed at more length in the Supplementary materials.
Finally, the negative trend is also compatible with a hypothesis regarding the quality of iNaturalist observers in which the number of observers has increased, as indicated by the data, whereas the observers’ discrimination ability or interests might have changed over time, perhaps because they focused on common species. Unfortunately, the very nature of the information in CS data makes assessing this effect very difficult. This aspect essentially describes the main problem with using unstructured CS data: because the methods are not standardized, any trend in the data might be explained by a trend in the behavior of the observers.
What explanations can be deduced for the absence of trends in the number of observations/effort? One possibility is that the presence of more observers simply resulted in more observations, and the number of observations and observers with more than 2 observations are roughly proportional. This means that the lack of trend could be an artifact of the data.
These results should be considered as hypotheses to be examined through more direct methods. Nonetheless, the results strongly suggest decreasing numbers of species in butterflies (important from a cultural perspective and perhaps a pollination perspective), bumblebees (important as pollinators), and Odonata (important as insect predators and as indicators). Therefore, the biodiversity of some of the most important and underappreciated groups of species in Mexico appears to be decreasing. If confirmed, this result would be highly alarming. Indeed, insects are key components of ecosystems (Noriega et al., 2018). Although for most insect species in Mexico we do not have direct documentation of their role, or of the economic and cultural value of their services, we have substantial indirect evidence of their importance as pollinators (Ashworth et al., 2009), as natural enemies of agricultural pests (López et al., 1999; Aluja et al., 2014), and as potential for non-conventional agri-business (López-Gutierrez et al., 2023). If substantiated, the decrease we report should be a major cause of alarm for Mexicans.
What might be causing a decrease? In a study in Europe (Schuch et al., 2012), in which a similar decrease in diversity was reported in a family of bugs of agricultural importance, a concomitant loss of non-agricultural habitat for the insects was reported. The study was conducted at the species level, and the authors argue that the more specialized, less tolerant species are those disappearing because of agricultural expansion. Again, monitoring using standardized procedures is required to test this idea.
Climate change is often cited as a cause of population decline in insects. However, climate change is a long-term phenomenon that occurs at the scale of many decades. To demonstrate climate change as a factor affecting population size, modeling or documentation of the effect of mean and variance in climatic variables on long-term population time series is necessary (Batalden et al., 2014; Boggs, 2016). The data used in this work is not appropriate for this purpose.
The results suggested that negative trends might not be identical among ecological regions. However, interestingly, the iNaturalist data indicated that pine-oak forest, xeric shrub, and tropical deciduous forest might be hotspots of diversity loss. This finding is somewhat surprising, given the widespread concern regarding tropical rainforests. Of course, the results may be due to the scarcity of data for tropical wet vegetation types.
CS data exists in substantial and growing amounts. It is a very valuable source of data. However, as with any data, it contains biases that are sometimes difficult to remove. The unavoidable conclusion is that Mexico must crucially invest in countrywide insect monitoring schemes based on systematic methods. Several approaches could be used. The first is improving CS schemes, providing training, and applying standard protocols, as performed in Canada, the USA, and many European countries (Streiter et al., 2024). This approach might be useful only for conspicuous, easily identifiable species, yet it markedly influences public environmental awareness and therefore should be maintained (Dickinson et al., 2012). Several methods using advanced technologies include computer vision, bioacoustics, and metagenomics can also be used (Van Klink et al., 2022). For bats, monitoring is already underway in Mexico (Zamora-Gutiérrez et al., 2020). Adoption of high technology methods would require funding, training, and substantial analytical capacity.
Regardless of the method chosen, in Mexico, the fourth most biologically diverse country on the planet, monitoring as many biodiversity components as possible is critical, and insects, “the little things that run the world” in the words of E. O. Wilson, appear to be disappearing very rapidly. Societies need to pay attention.
Acknowledgements
I am very grateful to Luis Eguiarte and Rodrigo Medellín, of the Mexican Instituto de Ecología, for their detailed and positive criticism of the methods I used. Their feedback led to several changes in the data analysis methods. Exequiel Ezcurra, of the University of California at Irvine, and Carlos Martinez, formerly of the University of Wyoming, also made very helpful statistical suggestions. My students Jennifer Ramos and Anahí Quezada helped me download and organize the data and patiently discussed the project with me. I gratefully thank the field ecologists in Mexico who took the time to respond to the questionnaire regarding their experiences with insects in the field.
References
Almond, R. E., Grooten, M., & Peterson, T. (2020). Living planet report 2020-Bending the curve of biodiversity loss. Gland, Switzerland: World Wildlife Fund.
Aluja, M., Ordano, M., Guillén, L., & Rull, J. (2012). Understanding long-term fruit fly (Diptera: Tephritidae) population dynamics: implications for areawide manage-
ment. Journal of Economic Entomology, 105, 823–836. https://doi.org/dx.doi.org/10.1603/EC11353
Aluja, M., Sivinski, J., Van Driesche, R., Anzures-Dadda, A., & Guillén, L. (2014). Pest management through tropical tree conservation. Biodiversity and Conservation, 23, 831–853. https://doi.org/10.1007/s10531-014-0636-3
Anonymous. (1997). Ecological regions of North America. Towards a common perspective. Montreal: Comisión de Cooperación Ambiental de América del Norte.
Ashworth, L., Quesada, M., Casas, A., Aguilar, R., & Oyama, K. (2009). Pollinator-dependent food production in Mexico. Biological Conservation, 142, 1050–1057. https://doi.org/10.1016/j.biocon.2009.01.016
Ayala, R., González, V. H., & Engel, M. S. (2012). Mexican stingless bees (Hymenoptera: Apidae): diversity, distribution, and indigenous knowledge. Pot-honey: a legacy of stingless bees. New York: Springer. https://doi.org/10.1007/978-1-4614-4960-7_9
Baillie, R. T., & Kim, K. H. (2018). Choices between OLS with robust inference and feasible GLS in time series regressions. Economics Letters, 171, 218–221. https://doi.org/10.1016/j.econlet.2018.07.036
Basset, Y., Lamarre, G. P., Ratz, T., Segar, S. T., Decaëns, T., Rougerie, R. et al. (2017). The Saturniidae of Barro Colorado Island, Panama: a model taxon for studying the long-term effects of climate change? Ecology and Evolution, 7, 9991–10004. https:// doi.org/10.1002/ece3.3515
Batalden, R. V., Oberhauser, K., & Peterson, A. T. (2014). Ecological niches in sequential generations of eastern North American Monarch butterflies (Lepidoptera: Danaidae): the ecology of migration and likely climate change implications. Environmental Entomology, 36, 1365–1373. https://doi.org/10.1603/0046-225x(2007)36[1365:enisgo]2.0.co;2
Beutelspacher, C. R. (1989). Las mariposas entre los antiguos mexicanos. Mexico D.F.: Fondo de Cultura Económica.
Boggs, C. L. (2016). The fingerprints of global climate change on insect populations. Current Opinion in Insect Science, 17, 69–73. https://doi.org/10.1016/j.cois.2016.07.004
Bonadies, E., Lamarre, G. P., Souto-Vilarós, D., Pardikes, N. A., Silva, J. A. R., Perez, F. et al. (2024). Population trends of insect pollinators in a species-rich tropical rainforest: stable trends but contrasting patterns across taxa. Biology Letters, 20, 20240170. https://doi.org/10.1098/rsbl.2024.0170
Boyle, M. J., Bonebrake, T. C., Dias da Silva, K., Dongmo, M. A., Machado-França, F., Gregory, N. et al. (2025). Causes and consequences of insect decline in tropical forests. Nature Reviews Biodiversity, 1, 315–331. https://doi.org/10.1038/s44358-025-00038-9
Challenger, A., & Soberón, J. (2012). Los ecosistemas terrestres, en Capital natural de México. Conocimiento Actual de la Biodiversidad. México D.F.: Conabio.
Chapman, A. (2005). Principles and methods of data cleaning-primary species and species-occurrence data. Copenhagen: Global Biodiversity Information Facility.
Cohn, J. P. (2008). Citizen science: Can volunteers do real research? Bioscience, 58, 192–197. https://doi.org/10.1641/B580303
Crall, A. W., Newman, G. J., Stohlgren, T. J., Holfelder, K. A., Graham, J., & Waller, D. M. (2011). Assessing citizen science data quality: an invasive species case study. Conservation Letters, 4, 433–442. https:// doi.org/10.1111/j.1755-263X.2011.00196.x
Di Cecco, G. J., Barve, V., Belitz, M. W., Stucky, B. J., Guralnick, R. P., & Hurlbert, A. H. (2021). Observing the observers: How participants contribute data to iNaturalist and implications for biodiversity science. Bioscience, 71, 1179–1188. https://doi.org/10.1093/biosci/biab093
Dickinson, J. L., Shirk, J., Bonter, D., Bonney, R., Crain, R. L., Martin, J. et al. (2012). The current state of citizen science as a tool for ecological research and public engagement. Frontiers in Ecology and the Environment, 10, 291–297. https://doi.org/10.1093/biosci/biab093
Duffus, N. E., Christie, C. R., & Morimoto, J. (2021). Insect cultural services: how insects have changed our lives and how can we do better for them. Insects, 12, 377. https://doi.org/10.3390/insects12050377
Edwards, C. B., Zipkin, E. F., Henry, E. H., Haddad, N. M., Forister, M. L., Burls, K. J. et al. (2025). Rapid butterfly declines across the United States during the 21st century. Science, 387, 1090–1094. https://doi.org/10.1126/science.adp4671
Fox, J., & Weisberg, S. (2019). Nonlinear regression, nonlinear least squares, and nonlinear mixed models in R. In J. Fox, & Weisberg, S. (Eds.), An R companion to applied regression, 3rd Ed. (pp. 1–31). Los Angeles: Sage Publications.
Gebhardt, S., Maeda, P., Wehrmann, T., Argumedo-Espinoza, J., & Schmidt, M. (2015). A proper land cover and forest type classification scheme for Mexico. The International Archives of the Photogrammetry, Remote Sensing and Spatial Information Sciences, 40, 383–390. https://doi.org/10.5194/isprsarchives-XL-7-W3-383-2015
Gulland, J. A. (1969). Manual of methods for fish stock assessment. Part 1. Fish Population Analysis. Rome: Fishery Resources and Exploitation Division, FAO.
Hallmann, C. A., Sorg, M., Jongejans, E., Siepel, H., Hofland, N., Schwan, H. et al. (2017). More than 75 percent decline over 27 years in total flying insect biomass in protected areas. Plos One, 12, e0185809. https://doi.org/10.1371/journal.pone.0185809
Hansen, M. C., Potapov, P. V., Moore, R., Hancher, M., Turubanova, S. A., Tyukavina, A., D. et al. (2013). High-resolution global maps of 21st-century forest cover change. Science, 342, 850–853. https://doi.org/ 10.1126/science.1244693
Isaac, N. J., van Strien, A. J., August, T. A., de Zeeuw, M. P., & Roy, D. B. (2014). Statistics for citizen science: extracting signals of change from noisy ecological data. Methods in Ecology and Evolution, 5, 1052–1060. https://doi.org/10.1111/2041-210X.12254
Lister, B. C., & García, A. (2018). Climate-driven declines in arthropod abundance restructure a rainforest food web. Proceedings of the National Academy of Sciences, 115, E10397–E10406. https://doi.org/10.1073/pnas.1722477115
López-Gutiérrez, S., Pozo, C., Muñoz, D. E. R., & Baltazar, E. B. (2023). The live butterfly trade as bio-business in southern Mexico. Journal of Rural and Community Development, 18, 131–152.
López, M., Aluja, M., & Sivinski, J. (1999). Hymenopterous larval–pupal and pupal parasitoids of Anastrepha flies (Diptera: Tephritidae) in Mexico. Biological Control, 15, 119–129. https://doi.org/10.1006/bcon.1999.0711
Lucas, M., Forero, D., & Basset, Y. (2016). Diversity and recent population trends of assassin bugs (Hemiptera: Reduviidae) on Barro Colorado Island, Panama. Insect Conservation and Diversity, 9, 546–558. https://doi.org/10.1111/icad.12191
Lyubchich, V., Gel, Y. R., & El-Shaarawi, A. (2013). On detecting non-monotonic trends in environmental time series: A fusion of local regression and bootstrap. Environmetrics, 24, 209–226. https://doi.org/ 10.1002/env.2212
Macías, C. G. V., & Freire, L. R. (2017). Plataforma Naturalista en México: herramienta de ciencia ciudadana. Índice, 12, 762.
Mason, B. M., Mesaglio, T., Barratt-Heitmann, J., Chandler, M., Chowdhury, S., Gorta, S. B. Z. et al. (2025). iNaturalist accelerates biodiversity research. Bioscience, 2025, biaf104. https://doi.org/10.1093/biosci/biaf104
McKinney, M. L., & Lockwood, J. L. (1999). Biotic homogenization: a few winners replacing many losers in the next mass extinction. Trends in Ecology & Evolution, 14, 450–453. https://doi.org/10.1016/S0169-5347(99)01679-1
McShane, B. B., Gal, D., Gelman, A., Robert, C., & Tackett, J. L. (2019). Abandon statistical significance. The American Statistician, 73, 235–245. https://doi.org/10.1080/00031305.2018.1527253
Michán, L., & Llorente, J. (2002). Hacia una historia de la Entomología en México. Publicaciones Especiales del Museo de Zoologia, UNAM, Mexico, 3, 3–52.
Miranda, F., & Hernández, E. (1963). Los tipos de vegetación de México y su clasificación. Botanical Sciences, 215, 29–179. https://doi.org/10.17129/botsci.1084
Mittermeier, R., Robles-Gil, P., & Mittermeier, C. (1997). Megadiversidad: los países biológicamente mas ricos del mundo. México D.F.: CEMEX.
NERC Centre for Population Biology, I. C. (1999). The global population dynamics database. https://knb.ecoinformatics.org/view/doi%3A10.5063%2FAA%2Fnceas.167.1
Noriega, J. A., Hortal, J., Azcárate, F. M., Berg, M. P., Bonada, N., Briones, M. J. et al. (2018). Research trends in ecosystem services provided by insects. Basic and Applied Ecology, 26, 8–23. https://doi.org/10.1016/j.baae.2017.09.006
Novoyny, V., & Basset, Y. (2000). Rare species in communities of tropical insect herbivores: pondering the mystery of singletons. Oikos, 89, 564. https://doi.org/10.1034/j.1600-0706.2000.890316.x
Olson, D. M., Wikramanayake, E. D., Burges, N. D., Powell, G. V. N., Underwood, E. C., D’Amico, J. A. et al. (2001). Terrestrial ecorregions of the world: a new map of life on earth. Bioscience, 51, 933. https://doi.org/10.1641/0006-3568(2001)051[0933:TEOTWA]2.0.CO;2
Openshaw, S. (1984). The modifiable areal unit problem. Norwich: Geo Books.
Ordano, M., Guillén, L., Rull, J., Lasa, R., & Aluja, M. (2013). Temporal dynamics of diversity in a tropical fruit fly (Tephritidae) ensemble and their implications on pest management and biodiversity conservation. Biodiversity and Conservation, 22, 1557–1575. https://doi.org/10.1007/s10531-013-0468-6
Orta, C., Reyes-Agüero, J. A., Luis-Martínez, M. A., Muñoz-Robles, C. A., & Méndez, H. (2022). Mariposas bioindicadoras ecológicas en México. Acta Zoológica Mexicana, 38, 1–33. https://doi.org/10.21829/azm.2022.3812488
Outhwaite, C. L. (2019). Modelling biodiversity trends from occurrence records. London: UCL (University College London).
Pannekoek, J., & van Strien, A. (1998). TRIM 2.0 for Windows (TRends and Indices for Monitoring Data).
Potts, S. G., Biesmeijer, J. C., Kremen, C., Neumann, P., Schweiger, O., & Kunin, W. E. (2010). Global pollinator declines: trends, impacts and drivers. Trends in Ecology & Evolution, 25, 345–353. https://doi.org/10.1016/j.tree.2010.01.007
Ramamoorthy, T. R., Bye, R., Lot, A., & Fa, J. (1993). Biological diversity of Mexico: origins and distribution. Oxford: Oxford University Press. https://doi.org/ 10.1007/bf02908211
Ramos-Elorduy, J., & Viejo-Montesinos, J. (2007). Los insectos como alimento humano: breve ensayo sobre la entomofagia, con especial referencia a México. Boletín de la Real Sociedad Española de Historia Natural Sección Biológica, 10, 61–84.
Rogel-Fajardo, I., Rojas-Lopez, A., & Ortega-Vega, S. Y. (2011). El turismo alternativo como estrategia de conservación de la reserva de la biosfera de la mariposa monarca (2008-2010). Quivera Revista de Estudios Territoriales, 13, 115–133.
Rzedowsky, J. (1986). Vegetación de México. México D.F.: Limusa.
Salcido, D. M., Forister, M. L., García-Lopez, H., & Dyer, L. A. (2020). Loss of dominant caterpillar genera in a protected tropical forest. Scientific Reports, 10, 422. https://doi.org/10.1038/s41598-019-57226-9
Sánchez-Herrera, M., Forero, D., Calor, A. R., Romero, G. Q., Riyaz, M., Callisto, M. F. et al. (2024). Systematic challenges and opportunities in insect monitoring: a Global South perspective. Philosophical Transactions of the Royal Society B, 379, 20230102. https://doi.org/10.1098/rstb.2023.0102
Sánchez, M. V., Cicowiez, M., & Ortega, A. (2022). Prioritizing public investment in agriculture for post-COVID-19 recovery: a sectoral ranking for Mexico. Food Policy, 109, 102251. https://doi.org/10.1016/j.foodpol.2022.102251
Schuch, S., Bock, J., Krause, B., Wesche, K., & Schaefer, M. (2012). Long-term population trends in three grassland insect groups: a comparative analysis of 1951 and 2009. Journal of applied Entomology, 136, 321–331. https://doi.org/10.1111/j.1439-0418.2011.01645.x
Shumway, R. H., & Stoffer, D. S. (2005). Time series analysis and its applications (Springer texts in Statistics). Cham, Switzerland: Springer-Verlag.
Sims, M. J., Stanimirova, R., Raichuk, A., Neumann, M., Richter, J., Follett, F. et al. (2024). Global drivers of forest loss at 1 km resolution. Environmental Research Letters, 20, 074027. https://doi.org/10.1088/1748-9326/add606
Soroye, P., Ahmed, N., & Kerr, J. T. (2018). Opportunistic citizen science data transform understanding of species distributions, phenology, and diversity gradients for global change research. Global Change Biology, 24, 5281–5291. https://doi.org/10.1111/gcb.14358
Streitberger, M., Stuhldreher, G., Fartmann, T., Ackermann, W., Ludwig, H., Pütz, S. et al. (2024). The German insect monitoring scheme: establishment of a nationwide long-term recording of arthropods. Basic and Applied Ecology, 80, 81–91. https://doi.org/10.1016/j.baae.2024.08.004
Szabo, J. K., Vesk, P. A., Baxter, P. W., & Possingham, H. P. (2010). Regional avian species declines estimated from volunteer-collected long-term data using List Length Analysis. Ecological Applications, 20, 2157–2169. https://doi.org/10.1890/09-0877.1
Tang, B., Clark, J. S., & Gelfand, A. E. (2021). Modeling spatially biased citizen science effort through the eBird database. Environmental and Ecological Statistics, 28, 609–630. https://doi.org/10.1007/s10651-021-00508-1
Thogmartin, W. E., Diffendorfer, J. E., López-Hoffman, L., Oberhauser, K., Pleasants, J., Semmens, B. X. et al. (2017). Density estimates of monarch butterflies overwintering in central Mexico. PeerJ, 5, e3221. https://doi.org/10.7717/peerj.3221
Thomas, J. (2005). Monitoring change in the abundance and distribution of insects using butterflies and other indicator groups. Philosophical Transactions of the Royal Society B: Biological Sciences, 360, 339–357. https://doi.org/10.2307/2265653
Van Klink, R., August, T., Bas, Y., Bodesheim, P., Bonn, A., Fossøy, F. et al. (2022). Emerging technologies revolutionise insect ecology and monitoring. Trends in Ecology & Evolution, 37, 872–885. https://doi.org/10.1016/j.tree.2022.06.001
van Strien, A. J., van Swaay, C. A., van Strien-van Liempt, W. T., Poot, M. J., & Wallis De Vries, M. F. (2019). Over a century of data reveal more than 80% decline in butterflies in the Netherlands. Biological Conservation, 234, 116–122. https://doi.org/10.1016/j.biocon.2019.03.023
Vidal, O., & Rendón-Salinas, E. (2014). Dynamics and trends of overwintering colonies ofthe monarch butterfly in Mexico. Biological Conservation, 180, 165–175. http://dx.doi.org/10.1016/j.biocon.2014.09.041
Wagner, D. L., Fox, R., Salcido, D. M., & Dyer, L. A. (2021). A window to the world of global insect declines: Moth biodiversity trends are complex and heterogeneous. Proceedings of the National Academy of Sciences, 118, e2002549117. https://doi.org/10.1073/pnas.2002549117
Willott, S. (2001). Species accumulation curves and the measure of sampling effort. Journal of Applied Ecology, 38, 484–486. https://doi.org/10.1046/j.1365-2664.2001.00589.x
Zamora-Gutiérrez, V., Ortega, J., Ávila-Flores, R., Aguilar-Rodríguez, P. A., Alarcón-Montano, M., Ávila-Torresagatón, L. G. et al. (2020). The Sonozotz project: assembling an echolocation call library for bats in a megadiverse country. Ecology & Evolution, 10, 4928–4943. https://doi.org/10.1002/ece3.6245
Zylstra, E. R., Ries, L., Neupane, N., Saunders, S. P., Ramírez, M. I., Rendón-Salinas, E. et al. (2021). Changes in climate drive recent monarch butterfly dynamics. Nature Ecology & Evolution, 5, 1441–1452. https://doi.org/10.1038/s41559-021-01504-1
Panmictic population of the pollinating moth Tegeticula baja(Lepidoptera: Prodoxidae), across the distribution of its plant hosts
Población panmíctica de la polilla polinizadora Tegeticula baja (Lepidoptera: Prodoxidae) a lo largo de la distribución de sus plantas hospederas
Maria Clara Arteaga a, *, C. Rocio Alamo-Herrera a, Anna Darlene van der Heiden a, b, Nicole Sicaeros-Samaniego a
a Centro de Investigación Científica y de Educación Superior de Ensenada, Departamento de Biología de la Conservación,
Carr. Tijuana-Ensenada 3918, Zona Playitas, 22860 Ensenada, Baja California, Mexico
b Uppsala University, Department of Medical Biochemistry and Microbiology, Hursagatan 3, 752 37, Uppsala, Sweden
*Corresponding author: arteaga@cicese.mx (M.C. Arteaga)
Received: 14 March 2025; accepted: 24 July 2025
Abstract
Ecological interactions and demographic history shape the genetic diversity of populations. Tegeticula baja is the specialist pollinator of different Yucca hosts in the Baja California Peninsula, a region that experienced changes in habitat distribution during the Pleistocene. To assess the effects of host specificity and historical changes in habitat configuration, we i) analyzed the genetic structure of moth populations associated with 3 different Yucca plant species, ii) identified signatures of historical demographic changes, and iii) reconstructed the past potential distribution of T. baja at different periods. We genotyped the COI of 128 moths from 39 locations and estimated genetic diversity, population structure, and demographic history. We found an overall haplotype diversity of 0.708 and a nucleotide diversity of 0.0015. Moth populations associated with the 3 hosts exhibited similar diversity levels, with no evidence of genetic structure. These findings suggest that ecological associations with different host plants do not drive T. baja diversification. Instead, its demographic history has played a more significant role in shaping the levels and the distribution of the genetic diversity.
Keywords: Ecological interaction; Genetic diversity; Historical demography; Insect-plant association; Population structure; Yucca
Resumen
Las interacciones ecológicas y la historia demográfica moldean la diversidad genética de las poblaciones. Tegeticula baja es una polinizadora especialista de diferentes yuccas hospederas en la península de Baja California, una región que experimentó cambios en la distribución de los hábitats durante el Pleistoceno. Para evaluar los efectos de la especificidad del huésped y los cambios históricos en la configuración de los hábitats, i) analizamos la estructura genética de las poblaciones de polillas asociadas a distintos hospederos, ii) identificamos señales de cambios demográficos históricos y iii) reconstruimos su distribución potencial en el pasado. Genotipificamos el COI de 128 polillas de 39 localidades y estimamos la diversidad genética, la estructura poblacional y la historia demográfica. Encontramos una diversidad haplotípica global de 0.708 y una diversidad nucleotídica de 0.0015. Las poblaciones de polillas asociadas a las 3 especies de plantas mostraron niveles de diversidad similares, sin evidencia de estructura genética. Estos hallazgos sugieren que la asociación ecológica con diferentes plantas huésped no impulsa la diversificación de T. baja. En cambio, su historia demográfica ha desempeñado un papel más importante en la configuración de su diversidad genética.
Palabras clave: Interacciones ecológicas; Diversidad genética; Historia demográfica; Asociación planta-insecto; Estructura poblacional; Yucca
Introduction
The ecological interaction between pollinator insects and their host plants plays a crucial role in shaping the distribution and genetic diversity of the species involved, influencing processes such as natural selection, genetic drift, and gene flow among populations (Futuyma, 2000; Gloss et al., 2013, 2016). Additionally, other factors, such as geographic distance (e.g., Driscoe et al., 2019) and demographic history shaped by past climatic changes (e.g., Liu et al., 2016; Smith et al., 2011), can influence the amount of genetic variation within populations and its spatial distribution.
Moths of the genus Tegeticula Zeller are specialist pollinators of the genus Yucca Linnaeus, maintaining an obligate mutualism (Engelmann, 1872; Pellmyr, 2003). Generally, each Tegeticula species pollinates a single Yucca species. However, few moth species pollinate multiple Yucca species (Althoff et al., 2006, 2012). During the flowering period, adult moths emerge and mate within Yucca flowers. The female uses specialized mouthparts to collect pollen and transfer it to other flowers. Upon arrival, she inserts her ovipositor into the ovary to lay her eggs. After, she deposits the pollen onto the flower’s stigma, ensuring fertilization and fruit development, from which the larvae will feed on a small portion of the seeds (Engelmann, 1872; Pellmyr, 2003). This obligate interaction between yucca and yucca moths influences the gene flow, facilitating differentiation and diversification processes in both pollinators and host plants (Arteaga et al., 2020; Leebens-Mack & Pellmyr, 2004; Leebens-Mack et al., 1998)
Yucca plants and yucca moths are distributed across North America (Pellmyr et al., 2008). This region has experienced changes in habitat distribution due to Pleistocene climatic fluctuations, which have impacted the genetic diversity and structure of both Yucca populations and their pollinators (Alemán et al., 2024; Arteaga et al., 2020; De la Rosa et al., 2020; Smith et al., 2011). In the Baja California Peninsula (BCP), Mexico, Tegeticula baja Pellmyr is an endemic moth species distributed from the central to the southern part of the peninsula. This moth pollinates 2 endemic Yucca species and their hybrid populations, which exhibit an allopatric distribution: Yucca valida Brandegee occurs in the arid ecosystems of the Central Desert and the northern Magdalena Plains, hybrid populations are found in the southern Magdalena Plains (Arteaga et al., 2020), and Y. capensis Lenz is located in the tropical dry forest of southern BCP (Lenz, 1998; Pellmyr et al., 2008; Turner et al., 1995).
The flowering phenology of these endemic Yucca species and their hybrid populations is asynchronous and influenced by water availability. Yucca valida blooms from April to July (Turner et al., 1995), hybrid populations flower in August and September, and Y. capensis blooms from September to October (Arteaga et al., 2015; Lenz, 1998). The asynchronous flowering limits the temporal coexistence of moths from different populations, as each group responds to the floral signals of its host plant. Additionally, these moths exhibit short-distance dispersal (Álamo-Herrera et al., 2022). Consequently, the temporal availability of floral resources and the limited dispersal of these pollinators may contribute to genetic structuring among populations across their distribution.
Given the obligate interaction between pollinators and plants, we hypothesize that distinct genetic lineages of T. baja are associated with each host Yucca species, influenced by geographic distances and asynchronous flowering. Additionally, considering historical habitat changes in the BCP (Dolby et al., 2015), we expect an impact on the species’ demographic history. Specifically, we i) examined the genetic structure among moth populations associated with each Yucca species and their hybrids, ii) identified signals of historical population changes, and iii) determined how the distribution of suitable conditions for the species has changed over time. This study will contribute to a better understanding of how ecological interactions and historical environmental changes shape the genetic diversity of pollinating moths in the Yucca-Tegeticula mutualism.
Materials and methods
We visited 75 localities where the endemic yuccas and their hybrids were found (Fig. 1A). We collected 3 to 5 fruits from at least 5 plants per locality. We dissected the fruits and examined them for the presence of T. baja larvae, which are typically found among the seeds. We stored the larvae in 96% ethanol and preserved them at -80 °C. We collected a total of 128 moth larvae from 39 of the 75 localities (Fig. 1A), including 16 sites of Yucca valida (N = 49), 8 of Y. capensis (N = 39), and 15 of hybrid plants (N = 40).
We used 20 mg of larval tissue for DNA extraction following the commercial kit “Qiagen DNeasy Blood & Tissue Kit” protocol. We amplified a fragment of the Cytochrome Oxidase subunit I (COI) marker using primers S1461 (5’-ACAATTTATCGCCTAAACTTCAGCC-3’) and A2302 (5’-CTACAAATCCTAATAATCCATTG-3’; Smith et al., 2009). The PCR mixture consisted of 5 μl of buffer (1X), 2 μl of MgCl (2 mM), 0.4 μl of dNTPs (0.6 mM), 0.2 μl of Taq polymerase (1U), 1 μl of each primer (0.4 mM each), 3 μl of DNA, and 12.4 μl of molecular-grade water, for a total reaction volume of 25 μl. Thermocycler conditions were: initial denaturation at 95 °C for 3 min, 35 cycles of denaturation at 94 °C for 30 s, annealing at 48 °C for 45 s, extension at 72 °C for 1 min, and a final elongation at 72 °C for 1 min. We verified the amplification quality using 1% agarose gels. PCR products were sequenced by SeqXcel (www.seqxcel.com). We also tested protocols for amplifying the nuclear EF1α gene and 9 nuclear microsatellites previously used in other species of the same genus (Drummond et al., 2010; Smith et al., 2008); however, amplifications were unsuccessful.
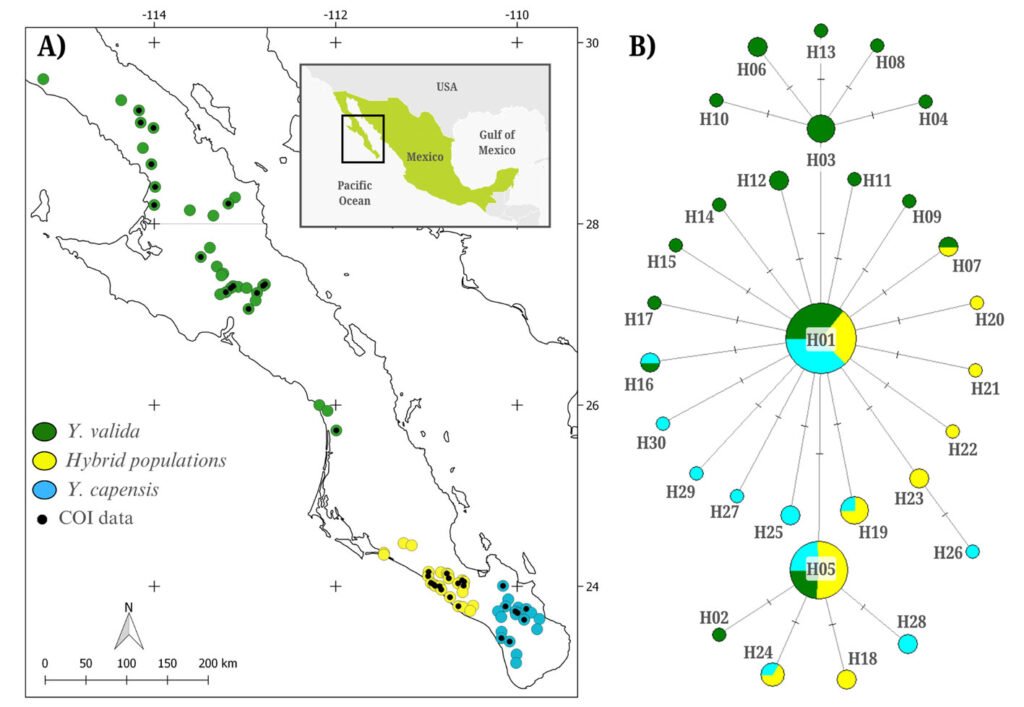
We visualized and edited COI sequences using MEGA X 4.0 and aligned them with MUSCLE (Tamura et al., 2007). We estimated the genetic diversity for the complete dataset and separately for individuals collected in locations from Y. valida, Y. capensis, and the hybrid Y. valida × Y. capensis using DNAsp (Rozas et al., 2003). We calculated the number of polymorphic sites (PS), number of haplotypes (H), haplotype diversity (h), and nucleotide diversity (π). To explore genealogical relationships, we constructed a haplotype network using the Median-Joining method in NETWORK 5.0 (Bandelt et al., 1999). Finally, to assess genetic structure among moths associated with different host plants, we implemented an analysis of molecular variance (AMOVA) using Arlequin (Excoffier et al., 2005).
We assessed the demographic history of T. baja using 3 methods. First, we conducted Tajima’s D test using DNAsp (Rozas et al., 2003). Negative values indicate population expansion, while positive values suggest population reduction (Nakamura et al., 2018). Second, we performed a mismatch distribution analysis in Arlequin, where an unimodal distribution suggests population expansion, while a multimodal distribution indicates a stable population size (Rogers & Harpending, 1992). Finally, we conducted a Bayesian Skyline Plot (BSP) analysis (Drummond et al., 2005) using BEAST 2.6.0 (Bouckaert et al., 2014). As input data, we used the commonly reported nucleotide substitution rate for COI in arthropods (1.77% divergence per lineage per million years; Papadopoulou et al., 2010), a strict molecular clock, and the HKY substitution model defined in JmodelTest2 (Darriba et al., 2012). We ran 100 million steps, sampling every 10,000 generations in the MCMC method. We calculated the effective sample size (ESS) value and constructed the BSP using TRACER 1.7 (Rambaut et al., 2018).
Table 1
Genetic diversity of Tegeticula baja based on the mitochondrial COI marker. The table includes the host plant, sample size (N), number of polymorphic sites (PS), number of haplotypes (H), haplotype diversity (h), and nucleotide diversity (π).
| Host yucca plant | N | PS | H | h | π |
| Yucca valida | 49 | 14 | 17 | 0.733 | 0.0016 |
| Hybrid populations | 40 | 9 | 10 | 0.750 | 0.0015 |
| Yucca capensis | 39 | 11 | 11 | 0.617 | 0.0012 |
We built species distribution models to assess changes in the geographic distribution of suitable conditions for Tegeticula baja and to support the interpretation of its genetic diversity and demographic history estimates. We considered 4 time periods: present (years 1970-2000), mid-Holocene (6,000 years ago), Last Glacial Maximum (22,000 years ago), and Last Interglacial (120,000 – 140,000 years ago). We constructed the models using the 75 sampling points obtained in this study, the 19 bioclimatic variables from the WorldClim 2.1 database (Fick & Hijmans, 2017), and the MAXENT software (Phillips et al., 2006), with 80% of the data used for training and 20% for validation. We configured 2,000 iterations and 10 replicates. The model evaluation included the area under the curve (AUC) and binomial probabilities, where an AUC > 0.9 reflects excellent predictive capacity.
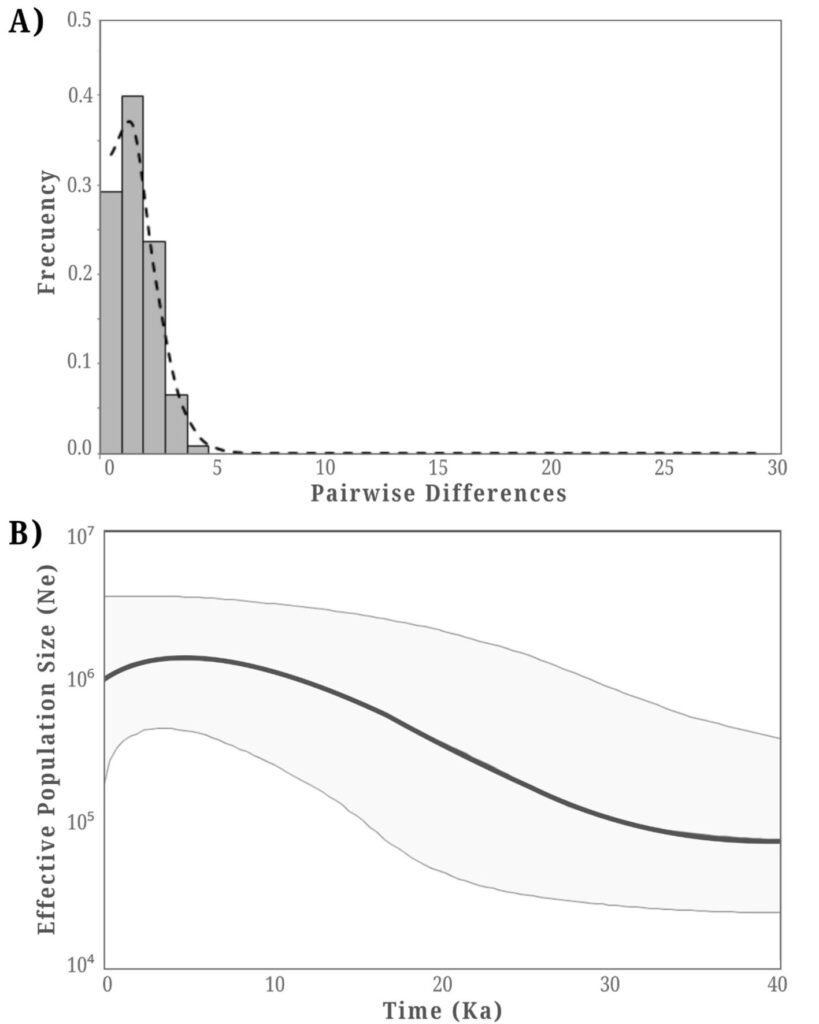
Results
The alignment of 128 sequences resulted in 767 bp with 27 polymorphic sites, defining 30 haplotypes (NCBI ID: PX127684-PX127713; Fig. 1B). The overall haplotype diversity was moderate (h = 0.708), and nucleotide diversity was low (π = 0.0015). Moth populations associated with the 3 host plants exhibited similar diversity levels (Table 1). The haplotype network indicated that haplotypes were closely related, and that the most abundant haplotype was widely distributed among the 3 moth populations pollinating different host plants (Fig. 1B). The AMOVA revealed that the variance among moths associated with different host plants was low and not significant (Fst = 0.011, p = 0.13). In contrast, most variation was found within populations.
Since no signs of genetic structure were detected, demographic analyses and niche modeling were conducted considering all individuals as a panmictic population. Demographic analyses provided evidence of a historical population expansion. Tajima’s D test showed significant negative values (D = -2.27970, p < 0.01), and the mismatch analysis distribution was unimodal (Fig. 2A). Consistently, the BSP analysis suggested a population expansion beginning approximately 25,000 years ago (Fig. 2B).
The species distribution models, with high predictive power (AUC > 0.9), revealed historical changes in the extent and distribution of environmentally suitable areas for T. baja (Fig. 3). During the Last Interglacial (120 ka), the distribution was limited to 2 areas, one in the central and other in the southern regions of the peninsula. During the Last Glacial Maximum (22 ka) and the mid-Holocene (6 ka), an expansion occurred in both regions. Finally, in the present period (1970-2000), the potential distribution of suitable conditions for these moths is observed to be continuously present along the western portion of the peninsula, connecting the central and southern regions.
Discussion
In the obligate mutualism between Yucca plants and Tegeticula moths, the distribution of feeding and oviposition resources provided by host plants determines the presence of moths in the landscape. In this study, we evaluated the genetic structure of Tegeticula baja populations associated with different Yucca species with allopatric distributions. Contrary to our expectations, we found a single panmictic population of pollinator moths throughout its geographic range. Additionally, we detected signals of historical demographic expansion. This suggests that the ecological association with different hosts is not driving the diversification of this species and that its historical demography has played a more relevant role in the distribution of its genetic diversity.
The genetic structure of a species is influenced by multiple factors, such as dispersal capacity, the intensity of ecological interactions, and the climatic history of the areas it inhabits (Driscoe et al., 2019; Futuyma, 2000; Smith et al., 2011). Specifically, T. baja has a limited dispersal distance per generation, and only 1 generation per year (~ 42 m; Álamo-Herrera et al., 2022); its host plants have a discontinuous distribution in the current landscape, and they also exhibit asynchronous flowering (Arteaga et al., 2020; Lenz, 1998; Turner et al., 1995). Together, these factors suggested that we could find genetic structure among moth populations associated with different Yucca species. However, we did not observe significant genetic differentiation. It is possible that the age of origin of its host plants and changes in the distribution of suitable habitat conditions could partially explain this pattern.
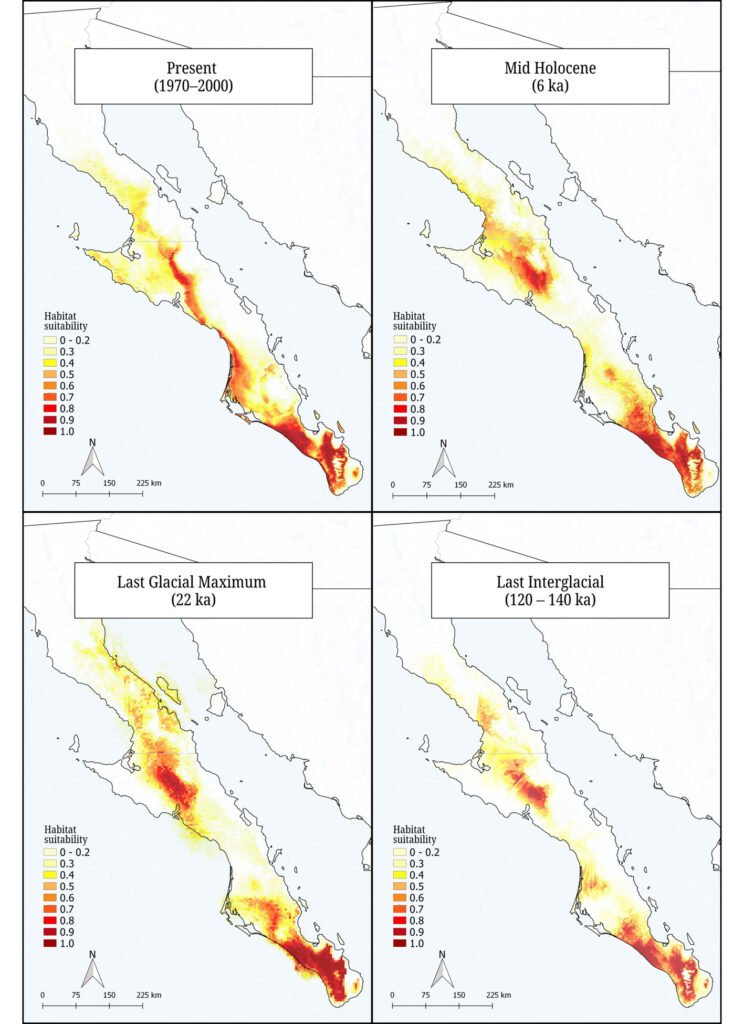
The divergence between the 2 endemic Yucca species of the BCP is estimated to have occurred approximately 500,000 years ago (Alemán et al., 2024). The formation of hybrid populations is even a more recent event, proposed to have occurred during the Pleistocene, around 21,000 years ago, when favorable climatic conditions allowed the co-occurrence of Y. valida and Y. capensis in the same region (Arteaga et al., 2020). This period of change in host plant distribution likely also influenced the distribution of the moth Tegeticula baja. Species distribution models and demographic analyses support this, indicating a population expansion beginning around 25,000 years ago, followed by a stabilization phase approximately 3,000 years ago. These historical changes in habitat configuration likely shaped the demographic history of the moth, reducing the potential for genetic divergence across its range due to alternating periods of population isolation and secondary contact. Although the current distribution of host plants is fragmented, the slow generational turnover of T. baja, with 1 generation per year, suggests that insufficient time has passed for genetic drift to produce detectable genetic structure.
The phylogeographic pattern of Tegeticula baja does not exhibit genetic structuring associated with host plant identity, which contrasts with that of other Tegeticula species, where genetic differentiation is correlated with either geographic distance or Yucca host species. For example, T. yucasella exhibits high genetic differentiation associated with geographic distance and interactions with different Yucca species (Leebens-Mack & Pellmyr, 2004). Similarly, T. maculata, the pollinator of Hesperoyucca Engelmann, exhibits genetic clades associated with the biogeographic history of its region (Althoff et al., 2007; Segraves & Pellmyr, 2001). Although all these studies, including ours, used the same mitochondrial marker (COI subunit), we did not detect genetic differentiation in our samples. These differences between our findings and previous reports may be related to the spatial scale, which is much larger in the study of T. yucasella, and to the time of origin and biogeographic history of the host plants in the case of T. maculata (Segraves & Pellmyr, 2001).
The levels of genetic diversity detected in the panmictic population of T. baja were moderate. In particular, moths associated with Y. valida exhibited a higher number of haplotypes, possibly due to their larger geographic range (Fig. 1A). The overall nucleotide diversity was low (π = 0.0015), falling below values reported for other species in the genus, such as T. antithetica and T. synthetica (π = 0.004 and 0.005, respectively; Smith et al., 2008). This pattern of low genetic diversity observed in T. baja may be related to the historical demographic growth detected in this species. A similar pattern was reported in the panmictic population of the parasitoid wasp Digonogastra sp., which interacts with 2 Tegeticula species in the BCP (π = 0.002; Álamo-Herrera et al., 2024). This supports the idea that the levels and distribution of genetic variation in these moths are more related to their historical demography than to their ecological interactions.
In conclusion, integrating genetic data with species distribution models allows us to understand how the climatic history of the BCP has influenced the distribution of genetic diversity and demographic changes in this species. Since the moths’ life cycle depends on Yucca flowering, which in turn responds to precipitation, climate change is likely to affect the population dynamics of these insects. Periods of extreme drought, such as those occurring in recent decades in the peninsula, may impact moth demography and exacerbate a population decline. Future studies focusing on the ecology and evolution of Prodoxus species associated with Yucca and Hesperoyucca in this region could enhance our understanding of the hidden diversity within this group and complement existing information on the northern species (Smith & Leebens-Mack, 2024).
Acknowledgements
The authors are grateful to Lita Castañeda and Mario Salazar for their help with laboratory analysis, technical support, and assistance in the fieldwork. They also thank Alberto López Alemán for his valuable assistance in improving the English language of the manuscript. This study was supported financially by Secretaría de Ciencia, Humanidades, Tecnología e Innovación (Secihti) (CB-2014-01-238843, infra-2014-1-226339). The Rufford Foundation also provided financial support for a part of this study (RSG 13704-1) and the Jiji Foundation. The authors thank the Associate Editor and the anonymous reviewers for their valuable comments. The authors declare no conflicts of interest.
References
Álamo-Herrera, C. R., Arteaga, M. C., Bello-Bedoy, R., & Rosas-Pacheco, F. (2022). Pollen dispersal and genetic diversity of Yucca valida (Asparagaceae), a plant involved in an obligate pollination mutualism. Biological Journal of the Linnean Society, 136, 364–374. https://doi.org/10.1093/biolinnean/blac031
Álamo-Herrera, C. R., Arteaga, M. C., & Bello-Bedoy, R. (2024). Genetic diversity and phenotypic variation in a parasitoid wasp involved in the yucca – yucca moth interaction. Revista Mexicana de Biodiversidad, 95, e955461. https://doi.org/10.22201/ib.20078706e.2024.95.5461
Alemán, A., Arteaga, M. C., Gasca-Pineda, J., & Bello-Bedoy, R. (2024). Divergent lineages in a young species: the case of Datilillo (Yucca valida), a broadly distributed plant from the Baja California Peninsula. American Journal of Botany, 111, e16385. https://doi.org/10.1002/ajb2.16385
Althoff, D. M., Segraves, K. A., Leebens-Mack, J., & Pellmyr, O. (2006). Patterns of speciation in the yucca moths: parallel species radiations within the Tegeticula yuccasella species complex. Systematic Biology, 55, 398–410. https://doi.org/10.1080/10635150600697325
Althoff, D. M., Svensson, G. P., & Pellmyr, O. (2007). The influence of interaction type and feeding location on the phylogeographic structure of the yucca moth community associated with Hesperoyucca whipplei. Molecular Phylogenetics and Evolution, 43, 398–406. https://doi.org/10.1016/j.ympev.2006.10.015
Althoff, D. M., Segraves, K. A., Smith, C. I., Leebens-Mack, J., & Pellmyr, O. (2012). Geographic isolation trumps coevolution as a driver of yucca and yucca moth diversification. Molecular Phylogenetics and Evolution, 62, 898–906. https://doi.org/10.1016/j.ympev.2011.11.024
Arteaga, M. C., Bello-Bedoy, R., León-de la Luz, J. L., Delgadillo, J., & Domínguez, R. (2015). Phenotypic variation of flowering and vegetative morphological traits along the distribution for the endemic species Yucca capensis (Agavaceae). Botanical Sciences, 93, 765–770. https://doi.org/10.17129/botsci.214
Arteaga, M. C., Bello-Bedoy, R., & Gasca-Pineda, J. (2020). Hybridization between yuccas from Baja California: Genomic and environmental patterns. Frontiers in Plant Science, 11, 685. https://doi.org/10.3389/fpls.2020.00685
Bandelt, H. J., Forster, P., & Röhl, A. (1999). Median-joining networks for inferring intraspecific phylogenies. Molecular Biology and Evolution, 16, 37–48. https://doi.org/10.1093/oxfordjournals.molbev.a026036
Bouckaert, R., Heled, J., Kühnert, D., Vaughan, T., Wu, C. H., Xie, D. et al. (2014). BEAST 2: a software platform for Bayesian evolutionary analysis. Plos Computational Biology, 10, e1003537. https://doi.org/10.1371/journal.pcbi.1003537
Darriba, D., Taboada, G. L., Doallo, R., & Posada, D. (2012). jModelTest 2: more models, new heuristics and high-performance computing. Nature Methods, 9, 772. https://doi.org/10.1038/nmeth.2109
De la Rosa-Conroy, L., Gasca-Pineda, J., Bello-Bedoy, R., Eguiarte, L. E., & Arteaga, M. C. (2020). Genetic patterns and changes in availability of suitable habitat support a colonisation history of a North American perennial plant. Plant Biology, 22, 233–242. https://doi.org/10.1111/plb.13053
Dolby, G. A., Bennett, S. E. K., Lira-Noriega, A., Wilder, B. T., & Munguia-Vega, A. (2015). Assessing the geological and climatic forcing of biodiversity and evolution surrounding the Gulf of California. Journal of the Southwest, 57, 391–455.
Driscoe, A. L., Nice, C. C., Busbee, R. W., Hood, G. R., Egan, S. P., & Ott, J. R. (2019). Host plant associations and geography interact to shape diversification in a specialist insect herbivore. Molecular Ecology, 28, 4197–4211. https://doi.org/10.1111/mec.15220
Drummond, A. J., Rambaut, A., Shapiro, B. E. T. H., & Pybus, O. G. (2005). Bayesian coalescent inference of past population dynamics from molecular sequences. Molecular Biology and Evolution, 22, 1185–1192. https://doi.org/10.1093/molbev/msi103
Drummond, C. S., Xue, H. J., Yoder, J. B., & Pellmyr, O. (2010). Host-associated divergence and incipient speciation in the yucca moth Prodoxus coloradensis (Lepidoptera: Prodoxidae) on three species of host plants. Heredity, 105, 183–196. https://doi.org/10.1038/hdy.2009.154
Engelmann, G. (1872). The flower of yucca and its fertilization. Bulletin of the Torrey Botanical Club, 3, 33.
Excoffier, L., Laval, G., & Schneider, S. (2005). Arlequin (version 3.0): an integrated software package for population genetics data analysis. Evolutionary Bioinformatics Online, 2005, 47–50. https://doi.org/10.1177/117693430500100003
Fick, S. E., & Hijmans, R. J. (2017). WorldClim 2: new 1-km spatial resolution climate surfaces for global land areas. International Journal of Climatology, 37, 4302–4315. https://doi.org/10.1002/joc.5086
Futuyma, D. J. (2000). Some current approaches to the evolution of plant–herbivore interactions. Plant Species Biology, 15, 1–9. https://doi.org/10.1046/j.1442-1984.2000.00029.x
Gloss, A. D., Dittrich, A. C. N., Goldman-Huertas, B., & Whiteman, N. K. (2013). Maintenance of genetic diversity through plant–herbivore interactions. Current Opinion in Plant Biology, 16, 443–450. https://doi.org/10.1016/j.pbi.2013.06.002
Gloss, A. D., Groen, S. C., & Whiteman, N. K. (2016). A genomic perspective on the generation and maintenance of genetic diversity in herbivorous insects. Annual Review of Ecology, Evolution, and Systematics, 47, 165–187. https://doi.org/10.1146/annurev-ecolsys-121415-032220
Leebens-Mack, J., Pellmyr, O., & Brock, M. (1998). Host specificity and the genetic structure of two yucca moth species in a yucca hybrid zone. Evolution, 52, 1376–1382. https://doi.org/10.1111/j.1558-5646.1998.tb02019.x
Leebens-Mack, J., & Pellmyr, O. (2004). Patterns of genetic structure among populations of an oligophagous pollinating Yucca moth (Tegeticula yuccasella). Journal of Heredity, 95, 127–135. https://doi.org/10.1093/jhered/esh025
Lenz, L. W. (1998). Yucca capensis (Agavaceae, Yuccoideae), a new species from Baja California Sur, Mexico. Cactus and Succulent Journal, 70, 289–296.
Liu, S., Jiang, N., Xue, D., Cheng, R., Qu, Y., Li, X. et al. (2016). Evolutionary history of Apocheima cinerarius (Lepidoptera: Geometridae), a female flightless moth in northern China. Zoologica Scripta, 45, 160–174. https://doi.org/10.1111/zsc.12147
Nakamura, H., Teshima, K., & Tachida, H. (2018). Effects of cyclic changes in population size on neutral genetic diversity. Ecology and evolution, 8, 9362–9371. https://doi.org/10.1002/ece3.4436
Papadopoulou, A., Anastasiou, I., & Vogler, A. P. (2010). Revisiting the insect mitochondrial molecular clock: the mid-Aegean trench calibration. Molecular Biology and Evolution, 27, 1659–1672. https://doi.org/10.1093/molbev/msq051
Pellmyr, O. (2003). Yuccas, yucca moths, and coevolution: a review. Annals of the Missouri Botanical Garden, 90, 35–55.
Pellmyr, O., Balcazar-Lara, M., Segraves, K. A., Althoff, D. M., & Littlefield, R. J. (2008). Phylogeny of the pollinating yucca moths, with revision of Mexican species (Tegeticula and Parategeticula; Lepidoptera, Prodoxidae). Zoological Journal of the Linnean Society, 152, 297–314. https://doi.org/10.1111/j.1096-3642.2007.00361.x
Phillips, S. J., Anderson, R. P., & Schapire, R. E. (2006). Maximum entropy modeling of species geographic distributions. Ecological Modelling, 190, 231–259. https://doi.org/10.1016/j.ecolmodel.2005.03.026
Rambaut, A., Drummond, A. J., Xie, D., Baele, G., & Suchard, M. A. (2018). Posterior summarization in Bayesian phylogenetics using Tracer 1.7. Systematic Biology, 67, 901–904. https://doi.org/10.1093/sysbio/syy032
Rogers, A. R., & Harpending, H. (1992). Population growth makes waves in the distribution of pairwise genetic differences. Molecular Biology and Evolution, 9, 552–569. https://doi.org/10.1093/oxfordjournals.molbev.a040727
Rozas, J., Sánchez-Del Barrio, J. C., Messeguer, X., & Rozas, R. (2003). DnaSP, DNA polymorphism analyses by the coalescent and other methods. Bioinformatics, 19, 2496–2497. https://doi.org/10.1093/bioinformatics/btg359
Segraves, K. A., & Pellmyr, O. (2001). Phylogeography of the yucca moth Tegeticula maculata: the role of historical biogeography in reconciling high genetic structure with limited speciation. Molecular Ecology, 10, 1247–1253. https://doi.org/10.1046/j.1365-294X.2001.01275.x
Smith, C. I., Godsoe, W. K., Tank, S., Yoder, J. B., & Pellmyr, O. (2008). Distinguishing coevolution from covicariance in an obligate pollination mutualism: asynchronous divergence in Joshua tree and its pollinators. Evolution, 62, 2676–2687. https://doi.org/10.1111/j.1558-5646.2008.00500.x
Smith, C. I., Drummond, C. S., Godsoe, W., Yoder, J. B., & Pellmyr, O. (2009). Host specificity and reproductive success of yucca moths (Tegeticula spp. Lepidoptera: Prodoxidae) mirror patterns of gene flow between host plant varieties of the Joshua tree (Yucca brevifolia: Agavaceae). Molecular Ecology, 18, 5218–5229. https://doi.org/10.1111/j.1365-294X.2009.04428.x
Smith, C. I., Tank, S., Godsoe, W., Levenick, J., Strand, E., Esque, T. et al. (2011). Comparative phylogeography of a coevolved community: concerted population expansions in Joshua trees and four yucca moths. Plos One, 6, e25628. https://doi.org/10.1371/journal.pone.0025628
Smith, C. I., & Leebens-Mack, J. H. (2024). 150 Years of coevolution research: evolution and ecology of yucca moths (Prodoxidae) and their hosts. Annual Review of Entomology, 69, 375–391. https://doi.org/10.1146/annurev-ento-022723-104346
Tamura, K., Dudley, J., Nei, M., & Kumar, S. (2007). MEGA4: molecular evolutionary genetics analysis (MEGA) software version 4.0. Molecular Biology and Evolution, 24, 1596–1599. https://doi.org/10.1093/molbev/msm092
Turner, R. M., Bowers, J. E., & Burgess, T. L. (1995). Sonoran Desert plants: an ecological atlas. Tucson: University of Arizona Press.
Revision of Hermeniawith descriptions of five new species and a new genus (Polychaeta: Polynoidae)
Revisión de Hermenia con descripciones de cinco nuevas especies y un género nuevo (Polychaeta: Polynoidae)
Patricia Salazar-Silva a, *, Sergio I. Salazar-Vallejo b
a Tecnológico Nacional de México, Instituto Tecnológico de Bahía de Banderas, Crucero a Punta de Mita, 63734 Bahía de Banderas, Nayarit, Mexico
b El Colegio de la Frontera Sur, Unidad Chetumal, Depto. Sistemática y Ecología Acuática, Av. Centenario Km 5.5, 77019 Chetumal, Quintana Roo, Mexico
*Corresponding author: salazarsilva01@yahoo.com (P. Salazar-Silva)
Received: 04 March 2025; accepted: 23 July 2025
http://zoobank.org/urn:lsid:zoobank.org:pub:08AE4922-3010-4327-B8AA-9753DD0AE1D1
Abstract
Hermenia Grube & Örsted (in Grube, 1856) contains 3 described species, with H. verruculosa Grube & Örsted (in Grube, 1856) as its type species and distributed in the Grand Caribbean. H. acantholepis (Grube, 1876) from the Philippines, and H. neoverruculosa Pettibone, 1975 from the Cargados Carajos, Republic of Mauritius. After the revision of type and non-type specimens, differences were observed in body shape, pigmentation pattern, prostomium, size of elytra, dorsal tubercles, and ventral papillae. Type material for the 3 species in the genus was revised, and Hermenia is redefined, its species are redescribed, and 4 species are described: H. chuarae sp. nov. from Indonesia, H. mezianei sp. nov. from Vietnam, H. treadwelli sp. nov. from the Grand Caribbean, and H. wehei sp. nov. from the Arabian Sea. On the other hand, Lepidonotus hermenioides Amoureux, 1974, described from Madagascar, resembles Hermenia species by having papillate integument, but differs by having elytra with long fimbriae. Parahermenia gen. nov. is proposed for including it, and Parahermenia piotrowskiae sp. nov. from Philippines. Identification keys for all species of Hermenia and Parahermenia were included.
Keywords: Caribbean; Lepidonotus; Parahermenia; Scaleworms; Taxonomy
Resumen
Hermenia Grube et Örsted (in Grube, 1856) contiene 3 especies descritas, con H. verruculosa Grube et Örsted (in Grube, 1856) como la especie tipo y distribuida en el Gran Caribe, H. acantholepis (Grube, 1876) de Filipinas y H. neoverruculosa Pettibone, 1975 de Cargados Carajos, República de Mauricio. Después de la revisión del material tipo y de ejemplares no tipo, se observaron diferencias en la forma del cuerpo, en el patrón de pigmentación, prostomium, tamaño de los élitros, tubérculos dorsales y papilas ventrales. El material tipo para las 3 especies fue revisado y Hermenia es redefinida, sus especies son redescritas y se describen 4 nuevas: H. chuarae sp. nov. de Indonesia, H. mezianei sp. nov. de Vietnam, H. treadwelli sp. nov. del Gran Caribe y H. wehei sp. nov. del mar Arábigo. Por otro lado, Lepidonotus hermenioides, descrita de Madagascar, es similar a las especies de Hermenia por tener integumento papilado, pero difiere por tener élitros con fimbria larga. Parahermenia gen. nov. se propone para incluir a ésta y a Parahermenia piotrowskiae sp. nov. de Filipinas. Se proveen claves de identificación para las especies de Hermenia y Parahermenia.
Palabras clave: Caribe; Lepidonotus; Parahermenia; Escamosos; Taxonomía
Introduction
Grube (1850: 36) separated the species of Polynoe Savigny in Lamarck, 1818 after the cover of the dorsum by elytra; in one group he placed all species with elytra large, completely covering dorsum, and in the other, he placed all species with smaller elytra, leaving most of dorsal surface uncovered. In his following paper (Grube, 1851: 120), he added the number of cephalic appendages and separated one species (Polynoe muricata Savigny in Lamarck, 1818) because it has only 4 (no median antenna), and for the other species, he continued using the size of elytra or cover of dorsal surface. In these groups, he used the type of elytra, and their number, sometimes with additional details, to separate similar species, a method that is still used for the whole family.
Kinberg (1856) proposed several new families and genera of scaleworms, and described many species collected during the “Eugenie Expedition”. For the Polynoidae, he proposed 5 genera and diagnosed them, including the elytral cover of the dorsal surface. In Halosydna Kinberg, 1856 the dorsum could be exposed, and in Hermadion Kinberg, 1856, the posterior segments were uncovered.
Hermenia Grube & Örsted in Grube, 1856 was proposed after the finding of an unusual polynoid scale worm because it has a few large elytrae along anterior chaetigers, and minute elytrae in median and posterior segments. The body is robust, and not easily to be broken in parts, as is the case for other polynoids; further, the surface of the body instead of being smooth bears tubercles or papillae, its elytra are fleshy, firmly attached in their elytrophores, and elytra surface has spiny non-sclerotized tubercles and digitiform papillae.
Grube & Örsted in Grube (1856) proposed Hermenia for a species seemingly without palps. Treadwell (1911) noted the palps and corrected the diagnosis for the genus, redescribed H. verruculosa, and regarded Polynoe nodosa Treadwell, 1901 (non P. nodosa Sars, 1861) as a junior synonym. Seidler (1923) redescribed H. verruculosa and redefined the genus to include Lepidonotus acantholepis (Grube, 1876), and redescribed the latter species in a posterior publication (Seidler, 1924).
Hermenia includes H. verruculosa Grube & Örsted in Grube, 1856 from the Caribbean Sea, H. acantholepis (Grube, 1876) from the Philippines, and H. neoverruculosa Pettibone, 1975 from the Seychelles, Indian Ocean. The most recent revision in the genus was carried out by Pettibone (1975) who added the third species: H. neoverruculosa. Pettibone separated the 3 species after the presence of dorsal tubercles or papillae, the number of accessory teeth in neurochaetae, presence of ventral papillae, and number of anterior larger elytrae.
The Caribbean Sea species, H. verruculosa, has been recorded in many localities along the Western Atlantic: New Jersey, Bahamas, Gulf of Mexico, Antilles, and Central to South America, from intertidal down to depths of 220 m (Pettibone, 1975; Salazar-Vallejo, 1996). It has been observed in cracks and interstices of coral rocks (Treadwell, 1911), and it has been reported as living with the brittle starfish Ophiocoma pumilla (now Ophiocomella pumilla [Lütken, 1856] after O’Hara et al., 2019) (Devaney, 1974; Pettibone, 1993). On the other hand, the Indian Ocean species, H. neoverruculosa, is apparently restricted to a few localities, whereas H. acantholepis, described from the Philippines, has been recorded in many localities in the Western Pacific, in coral fragments or on sand (Hanley & Burke, 1991).
During the study of available specimens previously identified as H. verruculosa from different localities along the Grand Caribbean, some were found, indicating the presence of another species. The most relevant differences were the presence of a nuchal lappet (missing in H. verruculosa), the shape of anterior dorsal tubercles, and the pigmentation pattern. First, the anterior margin of chaetiger 2 can be projected anteriorly in a semicircular nuchal lappet, or not projected at all. Second, in most specimens the dorsal tubercles are globular, and in one species they are depressed, being wider than long if seen from above, whereas in a single species they are modified into thin papillae. Third, there are 2 pigmentation patterns; in H. verruculosa there is a rectangular white spot in chaetigers 5 and 6, and in some specimens, there can be paler areas in dorsal surface of parapodia of chaetiger 6, sometimes becoming an inverted T-shaped spot, in the other pattern, the dorsum along first 5-6 chaetigers is completely white, or has an inverted triangular white area. Other species have rather homogeneous pigmentation along the body; further, the dorsal cirrostyle of median segments can be whitish (H. verruculosa), or brownish. These differences were used to separate similar species.
Other morphological features were found to be too variable such as body shape (rectangular vs. fusiform), parapodial size is modified after body contraction, and notochaetae are thin, denticulate capillaries that are usually broken, sometimes from the base, such that their number or presence was regarded as irrelevant for separating similar species.
To standardize these characters, type material of all the described species was examined. Consequently, Hermenia is redefined, H. verruculosa is restricted, and 3 new species are described, 1 from the Grand Caribbean, another one from the Sunda Strait, Indonesia, and a third one from Vietnam. A key to identify all species in Hermenia is also included.
One species, Lepidonotus hermenioides Amoureux, 1974 described from Madagascar, resembles Hermenia species by having papillate integument and neurochaetae with 2-3 additional teeth, but differs from those species by having large elytra in median and posterior segments, each with fimbriate elytra. The finding of another similar species from the Philippines made us propose Parahermenia gen. nov. for including them, and the additional species is described as H. piotrowskiae sp. nov.
Materials and methods
This study was based on the analysis of type material of H. verruculosa, H. acantholepis, and H. neoverruculosa. Further, the study of many specimens previously identified as H. verruculosa from different localities in the Grand Caribbean were examined from the University of Miami, mostly made during their cruises now well-known as University of Miami Deep Sea Expeditions, on board of RVs Gerda and John Elliot Pillsbury (1964-1975), and specimens from the Mexican Caribbean deposited in the Colección de Referencia, ECOSUR, Chetumal (ECOSUR).
Specimens were examined with stereoscopes and with compound microscopes. The length of the specimens was measured from the prostomial anterior margin to the posterior pygidial border, body width was measured in the widest part of the body, including parapodia but excluding chaetae. Body, segmental and parapodial terms follow Pettibone (1975). Segments without elytrae but provided with dorsal cirri are called cirrigers, whereas those having elytrae are called elytrigers.
Drawings were carried out with a camera lucida; some specimens were photographed with a digital camera, and a series of focal successions were compressed with HeliconFocus Ver. 8. Plates were generated with Paint Shop Pro ver. 2021.
To standardize the differences between species the following features were used: a) nuchal lappet over the prostomium, which is an anterior projections from segment 2 (Fig. 5A); b) expansion and anterior projection of the first 2 elytrophores; c) width of the second segment dorsally and in its anterior part, being regarded as narrow if shorter than prostomial width (Fig. 2A), or wide if being as long as, or larger than prostomial width (Fig. 4B); d) parapodial size in comparison to body width, being regarded as long if longer than half body-width, or short if smaller than half body width; e) presence of notochaetae; f) abundance and prominence of dorsal tubercles; g) abundance of ventral papillae; h) size of median and posterior segments elytrae, being regarded as tiny if smaller than dorsal segmental width; i) elytral insertion, being regarded as embedded if not lying on the surface; j) body shape being cylindrical roughly rectangular, fusiform depressed wider anteriorly, swollen wider anteriorly.
Known species are presented in chronological sequence, whereas the newly described taxa will be arranged alphabetically. The material belongs to the following institutions and museums: BMNH: The Natural History Museum, London, England. CAS: California Academy of Sciences, Invertebrate Zoology, San Francisco, California, U.S.A. ECOSUR: Colección de Referencia, El Colegio de la Frontera Sur, Chetumal, Quintana Roo, México. LACM: Natural History Museum of Los Angeles County, Allan Hancock Polychaete collection, Los Angeles, California, U.S.A. MNHN: National Museum of Natural History, Paris, France. MZB: Museum Zoologicum Bogoriense, National Research and Innovation Agency, Cibinong, Indonesia. SMF: Senckenberg Museum, Frankfurt, Germany. UF: Florida Museum of Natural History, University of Florida, Gainesville, Florida, U.S.A. UMML: Museum of Marine Invertebrates, Rosenstiel School of Atmospheric and Marine Sciences, University of Miami, Florida, U.S.A. USNM: National Museum of Natural History, Smithsonian Institution, Maryland, U.S.A. ZMH: Zoologischen Museum und Institut, Hamburg (now Leibniz Institute for the Analysis of Biodiversity Change), Germany. ZMUC: Museum of Zoology, University of Copenhagen, Denmark.
Descriptions
Family Polynoidae Kinberg, 1856
Subfamily Lepidonotinae Willey, 1902
Hermenia Grube & Örsted in Grube, 1856
Hermenia Grube & Örsted in Grube, 1856: 44; Baird, 1865: 200 (diagnosis); Seidler, 1923: 261-262, Fig. 6; Seidler, 1924: 94, Pettibone, 1975. 234 (diagnosis, key); Wehe, 2006: 78 (diagnosis); Salazar-Vallejo & Eibye-Jacobsen, 2012: 1397.
Diagnosis (modificated of Pettibone, 1975). Lepidonotinae with short body; integument tuberculate or papillate, venter smooth or papillate. Body with 26 segments, 12 pairs of elytrae on segments 2, 4, 5, 7, 9, 11, 13, 15, 17, 19, 21, 23. Elytra with globular macrotubercles and microtubercles, few papillae, fimbriae very short; first or first 3 elytra larger than median and posterior ones, most non-overlapping laterally or dorsally. Prostomium bilobed, with 2 pairs of eyes. Tentacular segment with few notochaetae and bulbous facial tubercle. Parapodia sub-biramous, notopodia small, neuropodia large. Notochaetae short, few to absent, slender, finely spinous. Neurochaetae stout, falcate, with 1-2 large accessory teeth. Dorsal cirri with cirrophores cylindrical, cirrostyles short. Ventral cirri short, subulate. Pharynx with 2 pairs of jaws and 11 pairs of marginal papillae. Nephridial papillae short, cylindrical from chaetigers 6-8.
Taxonomic summary
Type species. Hermenia verruculosa Grube & Örsted in Grube, 1856:44, by monotypy.
Distribution. Hermenia includes species from the Western Atlantic, the Indian and Western Pacific Oceans, that have been found mostly in shallow water mixed bottoms.
Remarks
Pettibone (1975) redefined Hermenia, but she did not regard the presence of the nuchal lappet as a distinctive character, although it is present in both H. acantholepis and H. neoverruculosa. The members of this genus are easily distinguished from other species belonging to lepidonotin genera provided with 12 pairs of elytra because, among other features, the reduction of the elytrae in medial and posterior segments, thick neurochaetae with separate accessory teeth, by having integument tuberculate or papillate with transverse lappet, and because the neurochaetae in segments 2 and 3 differ from those present in other segments (see key to genera below).
Hermenia resembles Parahermenia gen. nov. by having a tuberculate or papillate integument, neurochaetae subdistally smooth with tips bi- or tridentate. They differ because in Hermenia the first and last pair of elytra may overlap laterally, with most others being reduced, and by having minute fimbria with abundant macrotubercles; whereas in Parahermenia the elytra overlap laterally and have very long fimbria with few macrotubercles.
Key to Lepidonotinae with 12 pairs of elytra (modified after Fauchald, 1977)
1 With branchial filaments on the elytrophores
……………………………………………………………………………… 2
– Branchial filaments absent
……………………………………………………………………………… 4
2 (1) With pseudelytrae on non-elytrophoral, alternating segments posteriorly
……………………………………………………………………………… Euphionella Monro, 1936
– Pseudelytrae absent
……………………………………………………………………………… 3
3 (2) Neurochaetae coarsely spinose
……………………………………………………………………………… Chaetacanthus Seidler, 1924
– Neurochaetae finely plumose
……………………………………………………………………………… Euphione McIntosh, 1885
4 (1) Notopodia present
……………………………………………………………………………… 5
– Notopodia absent
……………………………………………………………………………… Drieschia Michaelsen, 1892
5 (4) All neurochaetae similar in thickness, without bidentate hooks
……………………………………………………………………………… 6
– With 1 very large bidentate hook in the middle of each neuropodial fascicle
……………………………………………………………………………… Sheila Monro, 1930
6 (5) Neurochaetae subdistally denticulate
……………………………………………………………………………… 7
– Neurochaetae subdistally with 1 or 2 accessory teth
……………………………………………………………………………… 9
7 (6) Notochaetae of 2 kinds, lancet-shaped and tapering and serrated
……………………………………………………………………………… Thormora Baird, 1865
– Notochaetae all of 1 kind, usually slender and serrated
……………………………………………………………………………… 8
8 (7) Pseudelytrae on cirrigerous segments (non-elytrophoral segments)
……………………………………………………………………………… Dilepidonotus Hartman, 1967
– Pseudelytrae absent
……………………………………………………………………………… Lepidonotus Leach, 1816
9 (6) All elytra overlapping laterally, with long fimbriae and a few macrotubercles
……………………………………………………………………………… Parahermenia gen. nov.
– A few anterior elytra overlapping laterally, most elytra small, non-overlapping laterally, fimbria very short, macrotubercles abundant
……………………………………………………………………………… Hermenia Grube & Örsted in Grube, 1856
Hermenia verruculosa Grube & Örsted in Grube, 1856 restricted
(Figs. 1, 2)
Hermenia verruculosa Grube & Örsted in Grube, 1856: 44-45; Treadwell, 1911: 9-11, Figs. 23-26 (partim; Dry Tortugas, Florida, dead coral rock); Seidler, 1923: 261, Fig. 6 (Saint Thomas); Seidler, 1924: 95-96 (Antillas: Barbados and Saint Thomas); Hartman, 1939: 4 (Old providence Island, Colombia, reef in tide pool); Ebbs, 1966: 500, Fig. 4 (Margot Fish Shoal, Florida in coral debris and red coral patch); Pettibone, 1975: 235-238, Figs 1, 2 (synonyms, partim; Gulf of Mexico Florida, off Lousiana, Yucatán, Mexico, low water to 223 m).
Polynoe nodosa Treadwell, 1902: 187, Figs 8, 9 (non Sars, 1861; Fajardo, Puerto rico, Saint Thomas, 20-23 m, coral bottom).
Lepidonotus verruculosus: Horst, 1922: 198 (Caracas bay, Curazao in coral Porites).
Diagnosis. Hermenia with body subrectangular; first pair of elytra bigger, covering prostomium, remaining elytra tiny; dorsal integument with abundant globular tubercles, in segments 4 and 5 forming a pale; segment 2 not projected over prostomium as nuchal lappet; neurochaetae with 1 accessory tooth.
Description. Holotype (ZMUC-1101), complete, body dorsally curved, wider anteriorly, 25 mm long, 6.5 mm wide, 26 segments. Dorsum brownish with transverse pale and dark bands, each segment with dark or pale series of tubercles, densely packed, larger in anterior and posterior segments. Venter smooth or pilose, with abundant thin micropapillae. Segments 5 and 6 with globular pale or colorless papillae arranged in small diffuse pale spots, 1 per segment.
Prostomium bilobed (Fig. 1A), partially retracted into segment 2; facial tubercle round; eyes colorless, 2 pairs, dorsolateral, subdermal, posterior eyes faded color, hidden by peristomium. Median antenna with ceratophore prominent, annulate, inserted frontally, ceratostyle smooth, thin subdistally swollen, with a darker band, tip thin. Lateral antennae with ceratophores cylindrical, ceratostyles thick, smooth, half as long as median antenna, subdistally swollen, tips long. Palps lost.
Tentacular segment indistinct dorsally, without chaetae. Tentaculophores thick, cylindrical. Tentacular cirri unequal to each other, dorsal ones larger, as long as median antenna. Second segment, chaetiger, narrow mid-dorsally with a prominent globular tubercle (Fig. 2A, B), without nuchal lappet. Elytrophores not expanded, nor extended anteriorly (Fig. 1A).
First pair of elytra largest, colorless (Fig. 1B), completely cover prostomium; elytral surface with abundant macrotubercles and cylindrical papillae; macrotubercles wart-like, semispherical, spinulose, larger towards margin, concentrated posteriorly; papillae thick, shorter along elytral margin (Fig. 1C, D), slightly swollen distally (Fig. 1E); other elytra circular, of similar size, strongly adhered in the back, elytral surface with 4 large semispherical macrotubercles, spinulose, and scattered cylindrical papillae, elytral margins with abundant thick, digitate papillae (Fig. 1F).
Parapodia short, as long as half body width; dorsally with traverse arrays of globular tubercles along notopodium, remaining surface with arrays of papillae; ventrally with transverse pleats and scattered tiny papillae (Fig. 1G). Notopodia reduced, acicular lobe small, covered by a small papilla, without notochaetae. Neuropodia slantingly truncated, dorsally furrowed, prechaetal lobe longer than postchaetal one, distally with some papillae.
Cirrigerous segments with cylindrical, thick, short cirrophores, cirrostyles smooth, short, with some filiform papillae, not reaching neurochaetal tips. Ventral cirri with cirrophores thick, very short, cirrostyles thick, short, tapered. Nephridial papillae from segment 7, progressively larger posteriorly, becoming thick, long, cylindrical papillae.
Neurochaetae amber color. Segments 2 and 3 with neurochaetae thin and small; second segment with broken chaetae (after Pettibone [1975]) with long, entire tips and marginal large spines]. Segment 3 with 2 types of neurochaetae, upper ones with tips entire, marginally spinulose, lower ones bidentate, smooth. Following segments with thick neurochaetae, with a main tooth, thick, slightly falcate, tips sharp or blunt, and a single accessory tooth, thin, separated from the main one (Fig. 1H).
Posterior region tapered, truncate; pygidium with anus terminal, with 1 pair of translucent, long, subdistally swollen anal cirri.
Variation. The additional specimens have palps with abundant short papillae. In some specimens the pigmentation of the macrotubercles can be reddish, instead of dark brown, or light brown, but in most specimens, there is a small white spot including unpigmented tubercles middorsally between segments 4 and 5, sometimes it can be completely reduced such that the dorsum is brownish, or there can be 2 smaller spots over parapodia (Fig. 2A).
The nuchal lappet is reduced (Fig. 2B). First elytron can be almost triangular (Fig. 2C), probably after being modified by pharynx eversion. The pharynx is a cylindrical tube with 11 pairs of marginal papillae (Fig. 2D-E), with lateral papillae being smallest.
Parapodia biramous (Fig. 2F), but notochaetae often few, fragile, usually broken; neurochaetae usually brownish, with a darker core, and with a single additional subdistal denticle (Fig. 2F, inset). The posterior region is barely tapered (Fig. 2G), elytra are of similar size as those present in preceding segments; pygidium truncate with anus between chaetigers 25 and 26.
A small specimen (LACM 14774; 7.5 mm long, 3 mm wide) has morphological features present in larger specimens, such as the body shape, the pharynx is exposed and has 11 pairs of marginal papillae, with the midlateral ones smaller. The first elytra are markedly larger than the following ones, and it has many globular macro- and microtubercles, with surface echinulate, the fimbriae are slightly larger than 2 times longer than wide. Further, the parapodia are biramous and their integument is papillate, with few notochaetae, mostly 1 per ramus, and they are denticulate along a single side. The neurochaetae have a single accessory denticle, looking bifurcate, and lower neurochaetae tend to be sharper.
Taxonomic summary
Type material. Caribbean Sea, Virgin Islands. Holotype of Hermenia verruculosa Grube & Örsted, 1856 (ZMUC-1101), St. John, U.S. Virgin Islands, 6 Jun. 1846, coll. Krøyer. Smaller syntype of Polynoe nodosa Treadwell, 1902 (USNM 16013), Saint Thomas, U.S. Virgin Islands, USFCS Fish Hawk, Sta. 6079, 37-42 m, 6 Feb. 1899 (15 mm long, 3.5 mm wide); larger syntype (USNM 16014), Puerto Rico, Fajardo, US Steamer Fish Hawk, Sta. unumb., depth not given, probably intertidal, 17 Jan. 1899 (bent ventrally; first pair of elytra and left parapodia of chaetigers 2, 3 and 15-18, and right parapodium of chaetiger 20 previously removed (1 elytron, and 3 posterior ones left in container); paler dorsal transverse bands in chaetigers 8 and 10; venter with abundant tiny papillae. Body 25 mm long, 6 mm wide, 27 chaetigers).

Distribution. Widely recorded along the tropical Western Atlantic, in shallow water, mixed, rocky, or coral bottoms. The indication for the Eastern Pacific (Salazar-Vallejo & Eibye-Jacobsen, 2012) was wrong. The record for the Arabian Sea (Wehe 2006) is shown below to belong to a different, newly described species (see below).
Additional material. Florida. One specimen (UF 1878), Gulf coast, N of St. Petersburg (28.59° N, 84.26° W), rock bottom, sponges, 29 m, 13 Mar. 2011, G. Paulay, M. Bemis & J. Moore, coll. complete, slightly bent ventrally; posterior end removed for molecular studies; no nuchal lappet; white spot rectangular, with paler areas in chaetiger 6; dorsal cirrostyles with swollen area white; anus position unknown; body 12 mm long, 3.5 mm wide, 26 chaetigers).
One specimen (UF 1879), same data as above (complete, slightly bent ventrally; pharynx exposed; first pair of elytra, and right parapodium of chaetiger 14 removed for observation (kept in container); no nuchal lappet; white spot rectangular, with paler areas in chaetiger 6; dorsal cirrostyles with swollen area white; anus terminal in chaetiger 26; body 21 mm long, 7 mm wide, 26 chaetigers).
One specimen (UF 1885), same data as above (complete, slightly bent ventrally; right elytron 1, and right parapodium of chaetiger 12 removed for observation (kept in container); no nuchal lappet; white spot rectangular, with paler areas in chaetiger 6; dorsal cirrostyles with swollen area white; anus terminal in chaetiger 26; body 24 mm long, 6 mm wide, 26 chaetigers).
Two specimens (USNM 17733), Dry Tortugas, Jun. -Jul. 1914, A.L. Treadwell, coll. (markedly bent ventrally; larger one with pharynx fully exposed, with 10-11 marginal papillae; body 25-30 mm long, 4.5-6.0 mm wide, 26 chaetigers).
Two specimens (USNM 46913), Tortugas,1930, W.L Schmitt, coll. (only 1 specimen in container; bent ventrally; chaetiger 6 with 3 white spots dorsally; 2 parapodia and first elytron from another specimen in container); body 30 mm long, 8 mm wide, 26 chaetigers).
Caribbean Sea. One specimen (ECOSUR 1309), University of Miami, Cruise P6806, RV Pillsbury, Southern Caribbean, off NE Venezuela, Sta. 709 (11°08.8’ N, 62°46.1’ W), 46 m, 19 Jul. 1968 (complete, anterior region almost completely brownish, with 1 middorsal small white spot in chaetiger 5, 2 laterals in chaetiger 6; no nuchal lappet; anus terminal in chaetiger 26; body 28 mm long, 6 mm wide, 26 chaetigers).
One specimen (ECOSUR 1310), University of Miami, Cruise P6802, RV Pillsbury, Nortwestern Caribbean, off Honduras, Sta. 629 (15°58’ N, 86°09’ W), 40 m, 21 Mar. 1968 (complete, anterior region with 1 middorsal small white spot in chaetiger 5, 2 laterals in chaetiger 6; no nuchal lappet; anus terminal in chaetiger 26; body 26 mm long, 7 mm wide, 26 chaetigers).
One specimen (ECOSUR 1311), Majagual, reef lagoon, rocks, 4 m, 6 Jun. 1998, M.A. Ruiz-Zárate, coll. (bent ventrally; with small narrow middorsal white and 2 lateral white spots in chaetiger 6; no nuchal lappet; anus terminal in chaetiger 26; body 18 mm long, 5 mm wide, 26 chaetigers).
Three specimens (ECOSUR 1316), Majagual, reef lagoon, rocks, 4 m, 6 Jun. 1998, M.A. Ruiz-Zárate, coll. (juveniles, bent ventrally, 1 with pharynx exposed, 10 pairs of marginal papillae; with wide middorsal white spot in chaetigers 4-5, and 2 lateral white spots in chaetiger 6; no nuchal lappet; anus terminal in chaetiger 26; body 8.1-8.5 mm long, 2.0-2.1 mm wide, 26 chaetigers).
One specimen (ECOSUR 3289), Majagual, Quintana Roo, México, reef lagoon, 4 m 6 Jun. 1998, M.A. Ruiz-Zárate, coll. (bent ventrally; pharynx exposed, 11 pairs of marginal papillae; 3 round white spots in chaetiger 6, anus terminal in chaetiger 26; body 13 mm long, 4 mm wide, 26 chaetigers).
One specimen (ECOSUR 3289), Punta Nizuc, Quintana Roo, México, 4 m, 10 Feb. 2001, P. Salazar-Silva, coll. (bent ventrally; with white middorsal band in chaetiger 5, wide transverse band in chaetiger 6; no nuchal lappet; anus terminal in chaetiger 26; body 11 mm long, 3 mm wide, 26 chaetigers).
One specimen (LACM 14763), British Virgin Islands, Guana Island, Muskmelon Bay, off Crab Cove (18.48° N, 64.57° W), reef slope coral heads, 10-16 m, 13 Jul. 2000, G. Hendler, T. Zimmerman, J. Martin & R. Ware, coll. (bent ventrally, with salt particles adsorbed on body; chaetigers 5-6 with a median rectangular white spot, and 2 smaller lateral ones on chaetiger 6; body 20 mm long, 6 mm wide, 26 chaetigers).
Two specimens (LACM 14774), British Virgin Islands, Guana Island, Pelican Ghut (18°28’36” N, 64°33’31” W), 6-12 m, G. Hendler, coll. (body 7.5-18.0 mm long, 3.0-6.5 mm wide, 26 chaetigers; smaller specimen features included in variation).
One specimen (LACM A44-39), Tortuga Island, RV Velero III, Sta. 44-39 (11°02’30” N, 65°14’45” W to 11°03’30” N, 65°14’00” W), 38-40 m, 21 Apr. 1939 (bent ventrally; chaetiger 6 with a median and 2 lateral white spots; body 23 mm long, 6 mm wide, 26 chaetigers).
Two specimens (LACM 147856), British White Bay, ARMS (18.3° N, 64.37° W), 9 m, 15 Jul. 2000, J. Zimmerman, J. Martin & R. Ware, coll. (almost colorless; both without nuchal hood, pharynx exposed, with 10 pairs of marginal papillae, body 6-8 mm long, 1.5-2.0 mm wide, 25-26 chaetigers).
One specimen (UMML 22.729a), University of Miami, Cruise P7006, Hispanola and Jamaica, RV Pillsbury, Sta. 1198 (17°49.4’ N, 76°12.3’ W), 29-37 m, 4 Jul. 1970 (bent ventrally; with 3 white spots, 1 in chaetigers 5-6, 2 laterals in chaetiger 6; without nuchal lappet; anus terminal in chaetiger 26; body 20 mm long, 6 mm wide, 26 chaetigers).
One specimen (UMML P979), University of Miami, Cruise P6907, Antillean Arc, Leeward Islands, RV Pillsbury, Sta. 979 (17°51’ N, 62°39’ W), 37 m, 22 Jul. 1969 (anterior fragment; without nuchal lappet; colorless, 10 mm long, 5 mm wide, 16 chaetigers).
One specimen (USNM 46916) Ascención Bay, Quintana Roo, Mexico, Central Part of Nicchehabin Reef., 1.2-1.8 m, Apr. 1960, E.L. Bousfield, coll. (slightly twisted; brownish, white spot in chaetiger 6; posterior nephridial lobes brownish; body 19.5 mm long, 5 mm wide, 26 chaetigers).
One specimen (USNM 46917), Ascensión Bay, Nicchehabin Reef., Quintana Roo, México, 21-19 m, 13 Apr. 1960, E.L. Bousfield, coll. (bent ventrally; dorsum with 3 white spots in chaetiger 6, chaetiger 5 with a median spot; body 28 mm long, 6 mm wide, 26 chaetigers).
One specimen (USNM 46920) Majito Reef, Puerto Rico, 0.3-0.6 m, 9 Mar. 1967, M.E. Rice, coll. (bent laterally; median segments swollen; body 20 mm long, 6 mm wide, 26 chaetigers).
Two specimens (USNM 50112), Carrie Bow Cay, Belize, 30 m, 11 Jun. 1972, M.E. Rice, coll. (bent ventrally; pharynx fully everted, 11-12 marginal papillae; body 16-21 mm long, 4-7 mm wide, 25-26 chaetigers).
One specimen (ZMUC-1102), St. Croix Island, H. Riise, coll. (no further data; prostomium retracted into second segment, without palps; body 19 mm long, 6.5 mm wide, 25 chaetigers).
Remarks
Hermenia verruculosa Grube & Örsted in Grube, 1856 differs from other species in the genus (see Hermenia key below)because it has a subrectangular dorsal white spot; segment 2 wide among the elytrophores, not projected on the prostomium as a nuchal lappet, elytrophores not expanded, dorsum with prominent globular densely packed tubercles; between segments 4 and 5 there is a dorsal small spot diffuse to well defined of unpigmented tubercles; venter wrinkled with abundant short thin papillae; elytra fleshy, rounded, attached firmly. The first pair of elytra is larger than the middle and posterior ones. All elytra with macrotubercles semispherical, spinous, unpigmented on the first pair of elytra, on subsequent elytrae reddish. Parapodia short, neuropodia diagonally truncate with an incipient prechaetal lobe. Neurochaetae with 1 accessory tooth and notochaetae absent in most of the parapodia. Pharynx everted with 11 pairs of marginal papillae and 2 pairs of jaws. The dorsal spot of unpigmented tubercles (Fig. 2F) is herein considered as distinctive of H. verruculosa.
Hermenia verruculosa has been recorded from different localities in the Grand Caribbean (Fauvel, 1953b; Horst, 1922; Bellan, 1964); however, the characterizations were short, often without illustrations, and the specimens are not available. Ebbs (1966) recorded 2 specimens from a coral patch of Margot Fish Shoal, Miami, Florida, as H. verruculosa, and his description referred red pigmentation; these specimens are colorless now, and the tubercles are white. On the other hand, part of Ebbs’ material corresponds with the features of H. verruculosa (UMML 150), whereas another lot (UMML 22:149) includes what we regard as an undescribed species described below.
Hermenia acantholepis (Grube, 1876) restricted
(Figs. 3, 4)
Polynoe acantholepis Grube, 1876: 61.
Polynoe (Lepidonotus) acantholepis: Grube, 1878: 24, Pl. 2, Fig. 1 (Samoa y Philippines).
Lepidonotus acantholepis: Herdman & Hornell, 1903: 25 (Ceylan, currently Sri Lanka, 82-92 m, in coral); Fauvel, 1922: 490-491, Fig. 1 (Houtman Abrolhos, Indian Ocean, in coral reef).
Hermenia acantholepis: Seidler, 1923: 262; Seidler, 1924: 94-95 (North West of Madagascar); Monro, 1924: 39-40, Figs 2-3 (Goto Island, Japon); Pruvot, 1930: 11-13, Pl. 1, Figs 27-32 (syn.); Fauvel, 1932a: 16-17 (Indian Ocean); Fauvel, 1947: 16-17, Fig. 12 (syn.), New Caledonia, South Pacific; Fauvel, 1953a: 38, Fig. 14a, b (Ceylan currently Sri Lanka); Pettibone, 1975: 239-241, Fig. 3 (partim, Fig. 4 corresponds to H. mezianei sp. nov., see below); Hanley & Burke, 1991: 62-64, Fig. 19A-G (Chesterfield Islands, New Caledonia, coral shallow water to 69 m, coral sand).
Diagnosis. Hermenia with body subrectangular; first and last pairs of elytra larger, overlapping, first 3 pairs larger than following ones; median elytra circular covering adjacent segments; elytral tubercles globular, with a basal hump; dorsal integument white, almost smooth, with thin papillae; segment 2 briefly projected over prostomium as a small nuchal lappet; neurochaetae tridentate.
Description. Holotype (ZMH 504) complete, mature female; dorsum wrinkled, globular tubercles scarce, whitish; venter wrinkled, without papillae; body depressed, 24 mm long, 6.5 mm wide, 26 chaetigers.
Prostomium in bad condition, wider than long, partially covered by segment 2; facial tubercle round; 2 pairs of eyes, both almost unpigmented. Median antenna with ceratophore cylindrical, long, inserted frontally; ceratostyle long, thick, surface smooth, without papillae; subdistally swollen, tip filiform. Lateral antennae with ceratophores thin, ceratostyle shorter, similar in shape to median antenna. Palps thick, without pigmentation, surface with rows of short papillae.
Tentacular segment not visible dorsally, tentaculophores thick, without chaetae; tentacular cirri similar in shape to median antennae. Segment 2 with nuchal lappet. Elytrophores expanded, not covering tentaculophores.
Elytra small, not overlapped laterally nor covering dorsum; elytra of anterior and posterior segments larger than elytra of middle segments, first pair of elytra round (Fig. 3C), second pair reniform (Fig. 3A); median and posterior elytra round (Fig. 3E), covering at least half width of adjacent segments. Elytral surface with tubercles and abundant filiform papillae. Tubercles abundant, ovoid, spinous, basally bulbous, projected beyond elytral margin (Fig. 3B, D). Elytra of median and posterior segments with marginal macrotubercles thinner, compressed (Fig. 3E-F), mostly brownish.
Parapodia with notopodia reduced, cylindrical ridge over neuropodia. Neuropodia thick, surface with transverse wrinkled rows, prechaetal lobe truncate, slightly larger than postchaetal lobe, distally with short papillae.
Dorsal cirri short, not longer than neuropodia, expanded subdistally, tips short filiform, similar in shape to antennae. Cirrophore cylindrical, short. Ventral cirri short, basally thick, tapered into fine tips, surface smooth. Nephridial papillae thick, short, distally blunt along chaetigers 6-25.
Notochaetae of anterior and middle segments short, scarce, upper region long, tapered into fine tip, absent in posterior segments. Neurochaetae thick, with striae; upper region short; tips thick, slightly curved, with 2 short, subdistal accessory teeth (Fig. 3G).
Posterior region tapered, blunt; pygidium with anus dorsal between chaetigers 25 and 26, anal cirri missing (thin, distally swollen, as long as last segment).
Variation. One specimen recently collected from Indonesia (UF 41) has a body almost pure white with brownish elytra (Fig. 4A); first 3 pairs of elytra larger than median ones, progressively smaller, such that the third is about twice as large as the fourth. The prostomium is white, with darker ceratophores and a basal medial region of ceratostyles, with a small dark band before swollen areas (Fig. 4B); eyes are black, of similar size, anterior eyes in the widest prostomial area, posterior ones towards the posterior margin. The nuchal lappet is truncate, well defined (Fig. 4B). The elytra have brownish macrotubercles, larger ones are marginal, with a basal swollen area, and globular microtubercles, more abundant along the inner anterior region (Fig. 4C). Parapodia biramous with small notochaetae (Fig. 4D, inset). Neurochaetae brownish with darker core, each with 2 subdistal teeth, often of similar size but different width (Fig. 4E). The posterior region is slightly tapered, with last pair of elytra slightly larger than preceding ones (Fig. 4F); anus dorsal, in posterior margin of chaetiger 25, anal cirri thin, resembling dorsal cirri, as long as last segment.
Taxonomic summary
Type material. Holotype of Polynoe acantholepis Grube, 1876 (ZMH 504), Upolu, Philippines.
Distribution. Indian Ocean and Western Pacific (Horst, 1917; Monro, 1939; Hanley & Burke, 1991; Imajima, 1997), in shallow mixed bottoms.
Additional material. Sri Lanka. One specimen (BMNH 1973:12), Talili, 73 m, no further data (dorsum wrinkled, with few tiny tubercles; venter with small papillae; first and second pair of elytra covering prostomium; elytral macrotubercles projected beyond margin, other elytra with marginal papillae filiform; notopodia small, with an acicular papilla).
Maldives. One specimen (BMNH 1941.4.4.195), Felidu, J.S. Gardiner, coll. (dorsum pale color, wrinkled with few papillae; notopodia short lobes; dorsal papillae short, compressed; venter with abundant short papillae; elytra of middle segments smaller than those of anterior segments, but larger than posterior ones).
Andaman Sea. Two specimens (LACM 2831963), juveniles, International Indian Ocean Expedition, RV Anton Bruun, Sta. 29 (11°23’ N, 93°31’ E), 55-40 m, 28 Mar. 1963 (1 dried-out; complete, whitish, cephalic appendages lost; elytra mostly white, barely compressed; nuchal lappet indistinct after eversion of pharynx; 11 pairs of marginal papillae; left parapodium of chaetiger 12 removed for observation kept in container; body 8 mm long, 2.5 mm wide, 26 chaetigers).
Indonesia. One specimen (UF 41), Sulawesi (Celebes Island), southern outer barrier reef (0.49° S, 122.07° E), 1-3 m, 22 Sep. 1999, G. Paulay, coll. (bent ventrally; body white, corrugated, pilose; elytra brownish, marginal macrotubercles L-shaped, internal lobe shorter; first elytra and left parapodium of chaetiger 14 removed for observation, kept in container; nuchal lappet distinct; dorsal cirrostyles with swollen area white; anus in chaetiger 25; body 23 mm long, 6 mm wide, 26 chaetigers).
Philippines. One specimen (CAS 187233), Hearst Philippine Biodiversity Expedition 2011, Luzon, Batangas Province, Mabini (Calumpan Peninsula), Maricaban Strait, Arthur’s Rock (13.70° N, 120.87° E), 16 m, 10 May 2011, A. Hermosillo, coll. (bent ventrally; body dirty white, first elytra whitish, all others brownish; first 3 and last elytra with marginal macrotubercles compressed, basally expanded (L-shaped); nuchal lappet short; dorsal cirrostyles with swollen areas white; anus in chaetiger 25; body 43 mm long, 9 mm wide, 26 chaetigers).
One specimen (CAS 214669), Verde Island Passage Expedition 2015, Mindoro, Oriental Mindoro Province, Puerto Galera, Shipyard Dive Site (13.52° N, 120.96° E), 6.5-16.0 m, sandy mud, 12 Apr. 2015, C. Piotrowski, coll. (partially dehydrated; middorsal area grayish, laterally white, elytra elytra brownish; first 3 and last elytra with marginal macrotubercles compressed, basally expanded (L-shaped); nuchal lappet distinct; dorsal cirrostyles with swollen areas white; anus between chaetigers 25-26; body 18.5 mm long, 5 mm wide, 26 chaetigers).
Papua New Guinea. One specimen (MNHN IA 2015-1924), Kavieng 2014 Expedition, New Ireland, RV Alis, Sta. DW4492 (02°25’23.99’’ S, 149°57’35.42’’ E), 112-140 m, 6 Sep. 2014 (bent ventrally, dried-out; first left elytron without macrotubercles along anterior inner area; median and posterior elytra circular, last pair larger than previous one; body 37 mm long, 7 mm wide, 26 chaetigers).
Coral Sea. One specimen (MNHN IA 2023-19), Cruise CORAIL 2, RV Coriolis, Sta. DW31 (19°24’51.59’’ S, 158°45’1.81’’ E), 57 m, 23 Jul. 1988, B. Richer de Forges, coll. (complete, slightly bent ventrally, first left elytron without macrotubercles along anterior inner area; median and posterior elytra circular, last 2 pairs larger than previous ones; body 22 mm long, 5.5 mm wide, 26 chaetigers).
Japan. One specimen (BMNH 1925.1.28.4-5), Goto Island, no further data (dorsum wrinkled without tubercles or papillae; dorsum smooth; notopodia cylindrical ridges; coelom with numerous oocytes).
New Caledonia. One specimen (MNHN IA 2023-57), Musorstom Cruise LAGON, Yaté Sector, RV Vauban, Sta. DW642 (21°54’12.0024’’ S, 166°42’12.0204’’ E), 44-47 m, 7 Aug. 1986, B. Richer de Forges, coll. (complete, slightly bent ventrally, first left elytron without macrotubercles along anterior inner area; median and posterior elytra circular, last pair larger than previous ones; body 21 mm long, 6 mm wide, 26 chaetigers).
One specimen (MNHN IA 2023-58), Musorstom Cruise LAGON, Yaté Sector, RV Vauban, Sta. DW737 (22°08’23.99’’ S, 166°59’06’’ E), 49-50 m, 12 Aug. 1986, B. Richer de Forges, coll. (complete, slightly bent ventrally, first left elytron without macrotubercles along anterior inner area; median and posterior elytra circular, last pair larger than previous ones; body 17.5 mm long, 5.5 mm wide, 26 chaetigers).
Remarks
Hermenia acantholepis differs from H. verruculosa by having a depressed body; segment 2 wide projected on the prostomium as a short nuchal lappet; dorsum with transverse wrinkled ridges, tubercles small, flattened, scarce. Venter smooth, without papillae. Elytra with 2 kinds of tubercles, bulbous-ovoid basally and larger around margin. In earlier species diagnoses the marginal macrotubercles have been regarded as ovoid, but they are roughly bottle-shaped, with a basal swollen area; H. acantholepis is also distinguished by its neurochaetae because they have 2 subdistal accessory teeth, and notochaetae are present along most notopodia.
Pruvot (1930: 11) and Fauvel (1947: 17) noted that H. acantholepis has a milky white body, with brownish elytra, and dorsal cirri with brownish bases. Pruvot (1930: 13) also noted 2 varieties: the western one found from Sri-Lanka to the Philippines with very small elytra (matching H. neoverruculosa Pettibone, 1975, see below), and an eastern one, found in New Caledonia and Samoa, with larger elytra (matching the current H. acantholepis).
Hermenia neoverruculosa Pettibone, 1975
(Figs. 5, 6)
Hermenia acantholepis: Fauvel, 1932b: 132 (Gulf of Suez, Read Sea); Fauvel, 1933: 41 (Gulf of Suez, Read Sea); Wehe, 2006: 79, Fig. 13a-c. (non Grube, 1876; Red Sea).
Hermenia neoverruculosa Pettibone, 1975:242-245, Figs 5-6 (syn.).
Diagnosis. Hermenia with body subrectangular; first and second pair of elytra overlapping; dorsal integument with abundant globular tubercles, often with black core; segment 2 with nuchal lappet wide, round; median and posterior elytra with black macrotubercles; neurochaetae tridentate.
Description. Holotype (BMNH 1941.4.4.197) complete, body depressed, subrectangular, 30 mm long, 6.5 cm wide, 26 chaetigers. Dorsum with transverse rows of globular tubercles with darker cores, larger along anterior and posterior segments. Tubercles light brown, except on segments 3-4, where some tubercles are whitish, without pigmentation, forming a small, diffuse middorsal spot. Venter pale, wrinkled, pubescent.
Prostomium bilobed (Fig. 5A), wider than long, not retracted into segment 2; facial tubercle round, pale. Eyes black; anterior eyes directed anteriorly, on the widest part of the prostomium, subdermal; posterior eyes near posterior margin, darker, slightly larger Fig. 6B). Median antenna with ceratophore inserted frontally, thick, surface with papillae, ceratostyle tubular, 2 times as long as prostomium length, subdistally swollen, tip filiform, surface smooth, with dark bands; lateral antennae with ceratophores cylindrical, short, about as long as prostomium; ceratostyles shorter, resembling median antenna. Palps missing.
Tentacular segment not visible dorsally, tentaculophores thin, long, with scarce, short chaetae. Tentacular cirri thin, long, resembling antennae, surface smooth.
Segment 2 narrow with a wide rounded nuchal lappet. First pair of elytrophores reaching bases of tentaculophores. Nephridial papillae not visible along anterior segments, distinct from chaetiger 7, longer along median and posterior segments.
First 2 pairs of elytra larger than following ones; elytron 3 slightly larger than elytron 4; first pair of elytra overlapped dorsally; elytral surface with macrotubercles blackish (Fig. 5B), or pale, and papillae; macrotubercles scattered, ovoid, short to elongate, blunt, spinous, micropapillae cylindrical, abundant (Fig. 5C), thicker along margin. Elytra of median and posterior segments small (Fig. 5D), macrotubercles black, with abundant cylindrical papillae on surface and along margins (Fig. 5E).
Parapodia with notopodia reduced to acicular lobe, acicular papilla present (Fig. 5F). Neuropodia thick, robust, distally with small papillae, and small prechaetal and postchaetal lobes with long papillae, postchaetal lobe larger than prechaetal one; dorsal surface with transverse rows of globular pale tubercles. Dorsal cirri thin, smooth, short, not reaching neurochaetae tips, same shape as antennae; cirrophore short, cylindrical. Ventral cirri short, thin, not reaching the lower tip of the neurochaetal lobe.
Notochaetae thin, short, 2-3, laterally denticulate capillaries. Neurochaetae thick, upper region short, main fang long, thick, slightly curved, more curved in posterior segments, accessory teeth short, 1/4 as long as main fang. Second parapodia with thin neurochaetae, tips entire with 3 long spines. Median parapodia with neurochaetae often with 2 accessory teeth, sometimes 1 broken (Fig. 5G).
Posterior region tapered, truncate; pygidium with anus dorsal, anal cirri lost.
Variation. One specimen (UF 3985) has body grayish, dorsal tubercles with cores slightly darker, better defined in intersegmental areas; first elytra pale with macrotubercles blackish, other elytra grayish with dark macrotubercles; first and second pair of elytra of similar size, second pair about 3 times as large as third pair (Fig. 6A); following elytra small, round, with black macrotubercles.
Prostomium pale, with ceratophores blackish with pigmentation extended along anterior prostomial areas, ceratostyles brownish, with a wide brownish band before enlarged pale area, tips pale (Fig. 6B); nuchal lappet barely developed, truncate. First elytron (Fig. 6C) with marginal macrotubercles lobate, not basally expanded, inner macrotubercles globular, shorter, blackish and paler ones (Fig. 6C).
Parapodia biramous (Fig. 6D) with few notochaetae (Fig. 6D, inset); neurochaetae abundant, brownish and paler, with darker cores (Fig. 6E), each with 2 fragile, tapered accessory teeth, sometimes with a single denticle left (Fig. 6E, inset), or all broken. Posterior region tapered; last pair of elytra as large as preceding ones, with black macrotubercles; anus dorsal between chaetigers 25-26, anal cirri thin, delicate, as long as last chaetiger (Fig. 6F).
Taxonomic summary
Type material. Republic of Mauritius. Holotype of Hermenia neoverruculosa Pettibone, 1975 (BMNH 1941.4.4.197), Cargados Carajos, 55 m, Sea Lark Expedition, 28 Aug. 1905, J.S. Gardiner, coll.

Distribution. Indian Ocean to Western Pacific, in shallow water mixed bottoms.
Additional material. Red Sea. One specimen (MNHN A403), Mission Dollfus en Égypte 1927-1929, Sta. 24, ‘bloc des vermets’ (probably intertidal Dendropoma vermetid mass, see Ben-Eliahu 1975), 30 Dec. 1928, R.P. Dollfus, coll. (complete, bent ventrally, partially dehydrated, integument damaged, middorsally eroded, with fine sediment particles laterally; dorsum with tubercles; all elytra on site, with fine sediment particles, median and posterior elytra circular; anus between chaetigers 25-26; body 14.5 mm long, 4 mm wide, 26 chaetigers).

One specimen (MNHN A894), Mission Dollfus en Égypte 1927-1929, Sta. 24, ‘bloc des vermets’ (probably intertidal Dendropoma vermetid mass, see Ben-Eliahu 1975), 30 Dec. 1928, R.P. Dollfus, coll. (complete, breaking in 2 parts, integument damaged, almost completely detached from body wall; dorsum with tubercles; 2 anterior elytra and right parapodium of chaetiger 12 previously removed, kept in container; most elytra on site, median and posterior ones circular; anus between chaetigers 25-26; body 18.5 mm, long, 5 mm wide, 26 chaetigers).
Papua New Guinea. One specimen (MNHN IA 2017-3596), Papua Niugini Expedition, W. Wongat Island, Sta. PR108 (05°08’04.44” S, 145°49’29.21” E), 2-30 m, 26 Nov. 2012 (complete, bent ventrally; all elytra on site, median elytra circular, smaller than segment width; anterior end markedly retracted, nuchal lappet not seen; dorsal tubercles globular, with black core; venter pubescent; anus dorsal between chaetigers 25-26; body 45 mm long, 8 mm wide, 26 chaetigers).
One specimen (UF 3985), Madang Province, BilBil Island (5.2962° S, 145.7822° E), outer reef, 13 m, 12 Nov. 2012, B. Faure, R. Ibik & P.-H. Kuo, coll. (complete, slightly bent laterally; first pair of elytra and right parapodia of chaetigers 10 and 12 removed for observation, kept in container; nuchal lappet distinct; dorsal tubercles globular with black core; venter pubescent; anus dorsal between chaetigers 25-26; body 40 mm long, 7 mm wide, 26 chaetigers).
One specimen (UF 3986), Madang Province, University Road (5.22° S, 145.79° E), 15 m, 12 Nov. 2012, B. Faure, R. Ibik & P.-H. Kuo, coll. (complete, juvenile, markedly bent ventrally after removal of left parapodia of chaetigers 11-16; not dissected to avoid further damage; elytra 2-6 with black spot in insertion area; elytra of median and posterior chaetigers with macrotubercles pale and black; nuchal lappet short; dorsal tubercles globular with black core, better defined intersegmentally; venter pubescent; anus dorsal between chaetigers 25-26; exposed enteron with globular, longer than wide caeca; body 14 mm long, 2.5 mm wide, 26 chaetigers).
Remarks
Hermenia neoverruculosa resembles H. verruculosa because both have dorsal globular tubercles, elytra with spinous macrotubercles and papillae. However, H. neoverruculosa differs from H. verruculosa in that its dorsal tubercles are shorter and densely packed, its venter is smooth, segment 2 is dorsally narrow between the first pair of elytrophores, and projected as a nuchal lappet over the prostomium, notochaetae are small in most parapodia, and neurochaetae have mostly 2 accessory teeth.
Hermenia neoverruculosa had been recorded only in the Indian Ocean, including D’arros Island (Monro, 1924:40), and the Seychelles (Monro, 1939:169). Pettibone (1975) recorded both materials and the features match the species.
Hermenia chuarae sp. nov.
(Fig. 7)
http://zoobank.org/urn:lsid:zoobank.org:act:59B0411C-59EB-4533-BAC3-5DE3698C850F
Hermenia acantholepis: Chuar et al., 2021: 64-65, Fig. 3G (non Grube, 1876 (Sunda Strait, Indonesia, 448-469 m, on sediment).
Diagnosis. Hermenia with body subrectangular; first and second pair of elytra overlapping; dorsal integument with abundant depressed tubercles, with pale core; segment 2 with nuchal lappet wide; median and posterior elytra with pale and brownish macrotubercles; neurochaetae tridentate.
Description. Holotype (MMMM 000) complete, body depressed, subrectangular, slightly bent laterally, 37 mm long, 6 mm wide, 26 chaetigers. Dorsum with transverse rows of depressed tubercles (Fig. 7A), core pale, of similar size along body; tubercles brownish, darker basally. Venter pale, wrinkled, with abundant filiform papillae.
Prostomium bilobed, about as wide as long, slightly retracted into segment 2; facial tubercle not visible dorsally, pale, with a median globular tubercle. Eyes black; anterior eyes about 2 times as large as posterior ones, directed anteriorly, on the widest part of the prostomium (Fig. 7B); posterior eyes near posterior margin. Antennae, palps, and tentacular cirri directed posteriorly. Median antenna with ceratophore papillate, inserted frontally, 2 times as wide and longer than laterals, ceratostyle about 2 times as long as prostomium, thin, subdistally swollen, swollen region pale, darker along it, tip thin, dark; lateral antennae with ceratostyles 1.5 times as long as prostomium, shorter than median one, with similar pigmentation pattern. Palps tapered, papillate, subdistally with a black ring, tips pale.
Tentacular segment indistinct dorsally, tentaculophores thin, long, with a single short chaeta. Tentacular cirri longer than median antenna, shorter than palps, with a similar pigmentation pattern to the antennae.
Segment 2 narrow with a wide rounded nuchal lappet. First pair of elytrophores reaching bases of tentaculophores. Nephridial papillae not visible along anterior segments, distinct from chaetiger 7, longer along median and posterior segments, often with marginal papillae.
First 3 pairs of elytra larger than the following ones, elytron 3 slightly larger than twice elytron 4. First pair of elytra overlapped dorsally; elytral surface with macrotubercles white and brownish (Fig. 7C), and papillae indistinct; macrotubercles short, globular to elongate, blunt. Elytra of median and posterior segments small, macrotubercles pale or brownish, with abundant papillae along margins.
Parapodia with notopodia reduced to acicular lobe (Fig. 7D), acicular papilla present. Dorsal cirri thin, smooth, short, not reaching neurochaete tips, resembling antennae; cirrophore short, cylindrical. Neuropodia thick, robust, distally with small papillae, and small prechaetal and postchaetal lobes with long papillae; prechaetal lobe larger than postchaetal one. Ventral cirri short, tapered, reaching the lower tip of the neurochaetal lobe.
Notochaetae thin, short, 3-4 laterally denticulate capillaries (Fig. 7D, inset). Neurochaetae thick, upper region short, main fang long, slightly falcate, accessory teeth short, 1/3 as long as main fang (Fig. 7E). Median parapodia with neurochaetae often with 2 accessory teeth, sometimes 1 denticle broken.
Posterior region tapered, truncate (Fig. 7F); pygidium with anus dorsal, anal cirri long, resembling antennae, about as long as last 3 chaetigers.
Taxonomic summary
Type material. Sunda Strait, Indonesia. Holotype (MZB Pol. 300), South Java Deep-Sea Biodiversity Expedition 2018, RV Baruna Jaya VIII, Sta. DW17 (06°07.33’ S, 105°00.76’ E to 06°07.22’ S, 105°00.86’ E), 448-469 m, 26 Mar. 2018.
Distribution. Only known from the Sunda Strait, Indonesia, in bathyal depths.
Etymology. This species is named after Miss Cheah Hoay Chuar. She is a polychaete taxonomist at the National University of Singapore, in recognition of her involvement in the South Java Deep Sea Biodiversity Expedition 2018, and because she kindly allowed us to study the holotype for this species.
Remarks
Hermenia chuarae sp. nov. resembles H. neoverruculosa, originally described from the Cargados Carajos Archipelago, Republic of Mauritius, Indian Ocean. Both species have a dorsum grayish with transverse rows of tubercles and elytral macrotubercles globular, non-L-shaped. However, they differ because H. chuarae has dorsal tubercles depressed, without black cores, and the median and posterior elytra have pale and brownish macrotubercles, whereas in H. neoverruculosa the dorsal tubercles are globular with black cores, and its median and posterior elytra have black macrotubercles.
On the other hand, the holotype of H. chuarae is the deepest record for any Hermenia species, being collected in sediments at 448-469 m depth.
Hermenia mezianei sp. nov.
(Fig. 8)
http://zoobank.org/urn:lsid:zoobank.org:act:500F2A6D-2A63-44F5-8DAB-EDE4646B7DCB
Hermenia acantholepis: Fauvel, 1935: 281, 286 (Vietnam, Pacific Ocean); Fauvel, 1939: 256 (Vietnam, Pacific Ocean); Dawydoff, 1952: 90 (non Grube, 1876; Vietnam, Pacific Ocean).
Diagnosis. Hermenia with body blunt fusiform; only first pair of elytra overlapping, first 3 pairs larger than following ones; median elytra oval barely covering adjacent segments; elytral tubercles globular, with a basal hump; dorsal integument white, barely rugose anteriorly, with thin papillae; segment 2 barely projected over prostomium as a small nuchal lappet; neurochaetae 2 accessory teeth.
Description. Holotype (MNHN A398) complete, dorsum wrinkled, almost smooth, a few sparse tubercles and larger papillae anteriorly (Fig. 8A); left elytra 1, right elytra 1, 2, and right parapodia of chaetigers 2, 14, and 15 previously dissected (kept in container), venter smooth, without papillae; body depressed, 13.5 mm long, 4.5 mm wide, 26 chaetigers.
Prostomium partially retracted into following segment, subhexagonal, wider than long (Fig. 8B); facial tubercle not visible dorsally, low; 2 pairs of black eyes of similar size. Median antenna with ceratophore 2 times as wide and slightly longer than laterals, ceratostyle short, tapered (probably in regeneration); right lateral ceratostyle smooth, tapered, subdistally wider, longer than palps. Palps barely papillate.
Tentacular segment not visible dorsally, tentaculophores thick, without chaetae; right tentacular cirri asymmetrical, ventral ceratostyle one 2 times as long as dorsal one (probably in regeneration). Segment 2 was previously slightly dissected, with a short nuchal lappet. Elytrophores not covering tentaculopohores.
Elytra small, not overlapped laterally nor covering dorsum: first 3 elytra larger than median ones, elytra 1 and 2 of similar size, elytron 3 about half their size, elytron 4 and following ones slightly smaller than elytron 3, oval, wider than long, barely covering adjacent segments. Elytra surface with tubercles and sparse filiform papillae (Fig. 8C). Tubercles abundant, ovoid, spinous, some basally bulbous, most globular, projected beyond elytral margins; marginal tubercles progressively smaller in median and posterior segments, pale or brownish.
Parapodia with notopodia reduced, blunt conical ridge over neuropodia (Fig. 8D). Neuropodia thick, surface with transverse wrinkled rows, prechaetal and postchaetal lobes truncate, of similar size, with margin papillate.
Dorsal cirri long, reaching upper neurochaetae, subdistally swollen, brownish, tips long filiform, resembling antennae and tentacular cirri. Cirrophore very short, cylindrical. Ventral cirri short, tapered, surface smooth. Nephridial papillae globular, short, along chaetigers 6-25.

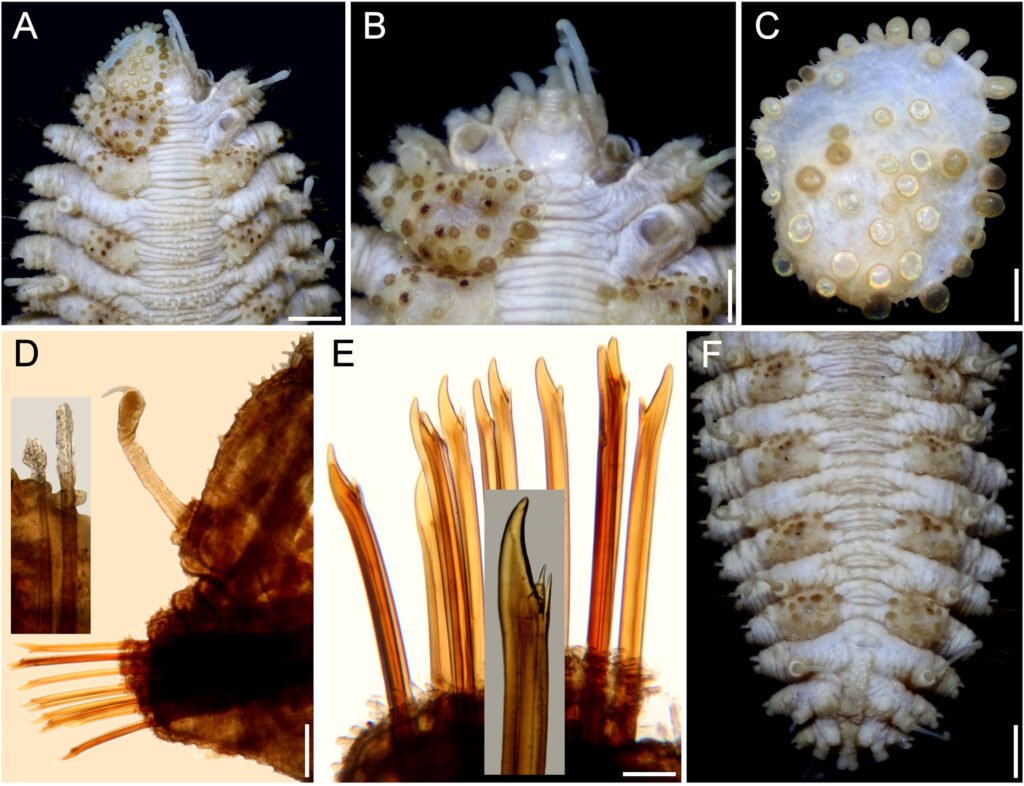
Notochaetae short, scarce, fragile, tips broken (Fig. 8D, inset), with impurities, marginally denticulate. Neurochaetae thick, smooth, upper region short; tips thick, slightly falcate, with 2 short, subdistal accessory teeth (Fig. 8E).
Posterior region tapered, blunt (Fig. 8F); last pair of elytra of similar size as preceding ones, pygidium with anus dorsal between chaetigers 25 and 26, anal cirri missing.
Taxonomic summary
Type material. Vietnam. Holotype (MNHN A398), Hón Lön Island (Honlohé, Ile Mamelles), By of Nha Trang, C. Dawydoff, coll. (no further data).
Distribution. Only known from Vietnam and Futuna Island (South Pacific Ocean), in substrates in platform-shelf depths.
Etymology. The specific epithet is after Dr. Tarik Meziane, curator of the Annelid collection in the Muséum National d’Histoire Naturelle, Paris, in recognition of the support he has kindly provided to our research projects.
Additional material. Futuna Island, Western Pacific, One specimen (MNHN IA 2023-45), Cruise MUSORSTOM 7, RV Alis, Sta. CP517 (14°13’23.99’’ S, 178°10’24.02’’ W), 223-235 m, 12 May 1992, P. Bouchet et al., coll. (partially dehydrated, markedly bent ventrally, integument tuberculate; first 3 pairs of elytra larger than following ones, third elytra about 2 times as large as fourth one; median and posterior elytra circular to oval, slightly wider than long, barely covering segment; last pair of elytra as large as previous one; not measured to avoid further damage).
Two specimens (MNHN IA 2023-48), Cruise MUSORSTOM 7, RV Alis, Sta. CP498 (14°18’54’’ S, 178°03’05.99’’ W), 105-160 m, 10 May 1992, P. Bouchet et al., coll. (integument tuberculate; first 3 pairs of elytra larger than following ones, third elytra about 2 times as large as fourth one; median and posterior elytra circular to oval, slightly wider than long, barely covering segment; last pair of elytra slightly larger than precedent one; body 18-28 mm long, 5.5-6.0 mm wide, 26 chaetigers).
Remarks
Hermenia mezianei sp. nov. resembles H. acantholepis restricted, by having a whitish, almost smooth integument, first 3 pairs of elytra larger than the following ones, each with marginal macrotubercles basally swollen, and trifid neurochaetae. Their main differences are the shape and size of the median and posterior elytra, the distribution of macrotubercles in the first elytra, and the type of macrotubercles in the median and posterior elytra. First, in H. mezianei the median and posterior elytra are oval, wider than long, barely covering adjacent segments, whereas in H. acantholepis they are circular and cover at least half of the adjacent segments. Second, the first elytra of H. mezianei have the anterior inner area with round macrotubercles, whereas there are no macrotubercles in the same area in H. acantholepis, only microtubercles. Third, macrotubercles in median and posterior elytra in H. mezianei are short, globular, mostly pale, barely projected from the elytral margin, whereas in H. acantholepis they are digitate, almost all brownish, markedly projected from the elytral margin.
Hermenia treadwelli sp. nov.
(Figs. 9, 10)
http://zoobank.org/urn:lsid:zoobank.org:act:9588D5E3-1895-4086-AD94-A650503CB7F7
Hermenia verruculosa: Treadwell, 1911: 9-11, Figs 23-26 (partim, nonGrube & Örsted in Grube, 1856; Dry Tortugas, Florida in dead coral); Pettibone, 1975: 235-238, Figs 1, 2 (syn., partim; only his specimens completely white along anterior chaetigers.
Diagnosis. Hermenia with body fusiform; segment 2 with short nuchal lappet; first pair of elytra bigger, not overlapping, other elytra tiny; dorsal integument with abundant dorsal tubercles, paler ones forming an inverted T-shaped spot in segments 5 or 5 anterior segments white, with pale tubercles; segment 2 with short projection over prostomium as a nuchal lappet; neurochaetae bidentate.
Description. Holotype (ECOSUR 1396) with body robust, depressed, fusiform, widest medially, 24 mm long, 6.5 mm wide, 26 segments. Dorsum covered with globular tubercles of different sizes, some larger than the others, larger in anterior and posterior segments, tubercles dark brown in anterior segments, paler towards posterior part in cirrigerous segments, giving an appearance of banding of dark segments alternating with pale ones. First 5 segments almost completely white (Fig. 9A). Venter with thick digitiform papillae.
Prostomium bilobed (Fig. 9B), retracted into the second segment. Two pairs of dark eyes, anterior pair on the widest part of the prostomium; posterior eyes covered by second segment. Palps robust with papillae, tapered, tips filiform (left palp lost). Three antennae, median antennae with ceratophore inserted frontally on prostomial lobe, about 2 times as wide as laterals, cylindrical, darker than prostomium, ceratostyles lost. Pharynx not everted.
First pair of elytra largest, round (Fig. 9C), covering prostomium, following elytra smaller; first pair of elytra with papillae digitiform, short, thick on the surface and along margin; elytral surface with many macrotubercles, present on elytrophore mark, larger towards margin. Elytra of median and posterior segments with abundant digitiform papillae on surface and along margin; macrotubercles thick, hemispherical, spinous.
Tentacular segment not visible dorsally; tentaculophores short, without chaetae; tentacular cirri thick, similar in shape and length as the median antenna. Segment 2 narrow between the first pairs of elytrophores, projected on the prostomium as a nuchal lappet. First pair of elytrophores expanded.
Parapodia biramous, truncate, dorsal surface with rows of globular tubercles (Fig, 9D). Notopodia short, acicular ridge covered with 1 blunt papilla (Fig. 10D, inset left). Neuropodia thick with prechaetal and postchaetal lobes reduced, each with abundant marginal papillae. Cirrigerous segments with dorsal cirri short, not surpassing neuropodia tips, surface smooth, similar in shape to antennae; cirrophore short, thick, with papillae; ventral cirri thin, short. Nephridial papillae visible from segment 7, thick, and becoming larger on posterior segments.
Notochaetae absent. Neurochaetae of median and posterior segments thick (Fig. 9D, insets above), upper region short with a long tooth, slightly curved, and a secondary straight denticle distant from each other. Parapodia of the second segment with neurochaetae thinner, of 2 kinds, some with long spines, tips entire, others with spines only subdistally.
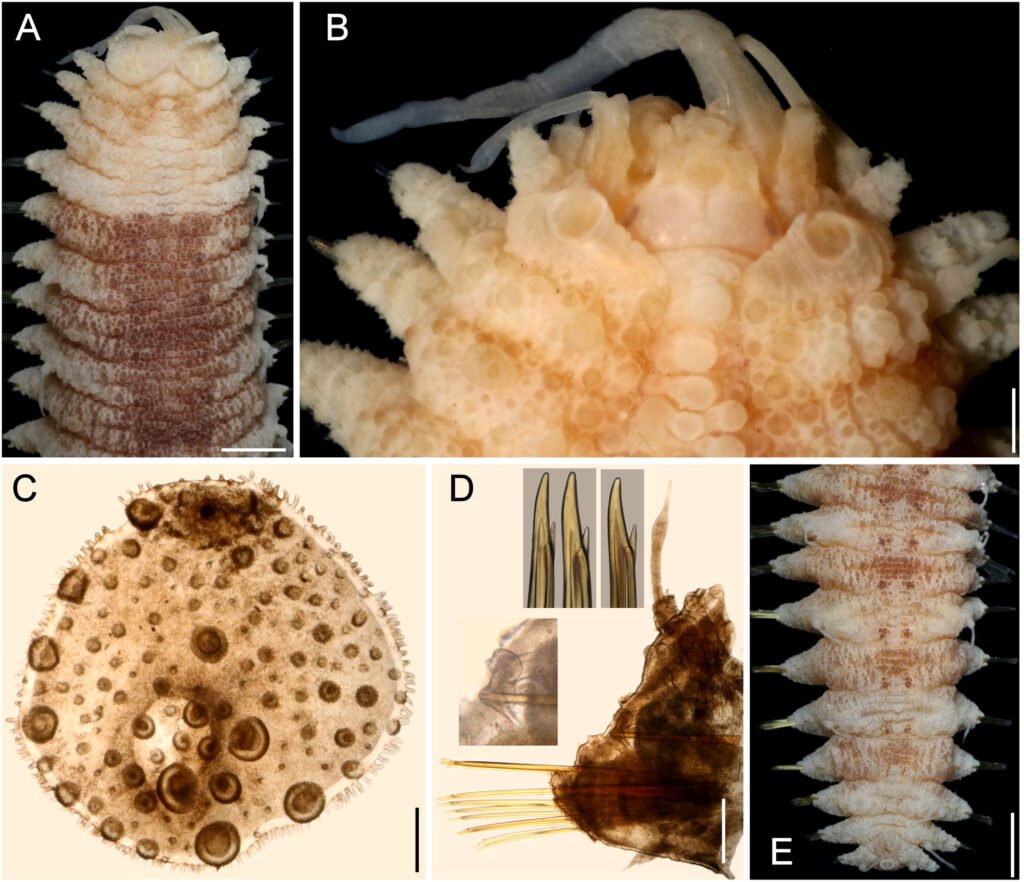
Posterior region pale, truncate (Fig. 9E); last pair of elytra as large as preceding ones, pygidum terminal, dorsal anus, anal cirri lost.
Variation. Other specimens have first elytra pale, and an anterior, inverted triangle spot along the dorsum of the chaetigers 3-5 (Fig. 10A). The prostomium retains dark-reddish pigmentation along ceratophores, and the median antenna ceratostyle, palps, and tentacular cirri are brownish, with pale swollen areas (Fig. 10B). The nuchal lappet is better defined as well.
The first elytron has globular macrotubercles, mostly brownish, with abundant marginal papillae and pale microtubercles along its surface (Fig. 10C). Parapodia biramous (Fig. 10D) with barely projected notopodia (Fig. 10D, left inset), and dorsal cirri with conical cirrophore (Fig. 10D, upper inset). Notochaetae broken and lost. Neurochaetae abundant, brownish (Fig. 10E), with 1 subdistal accessory denticle (Fig. 10E, insets).
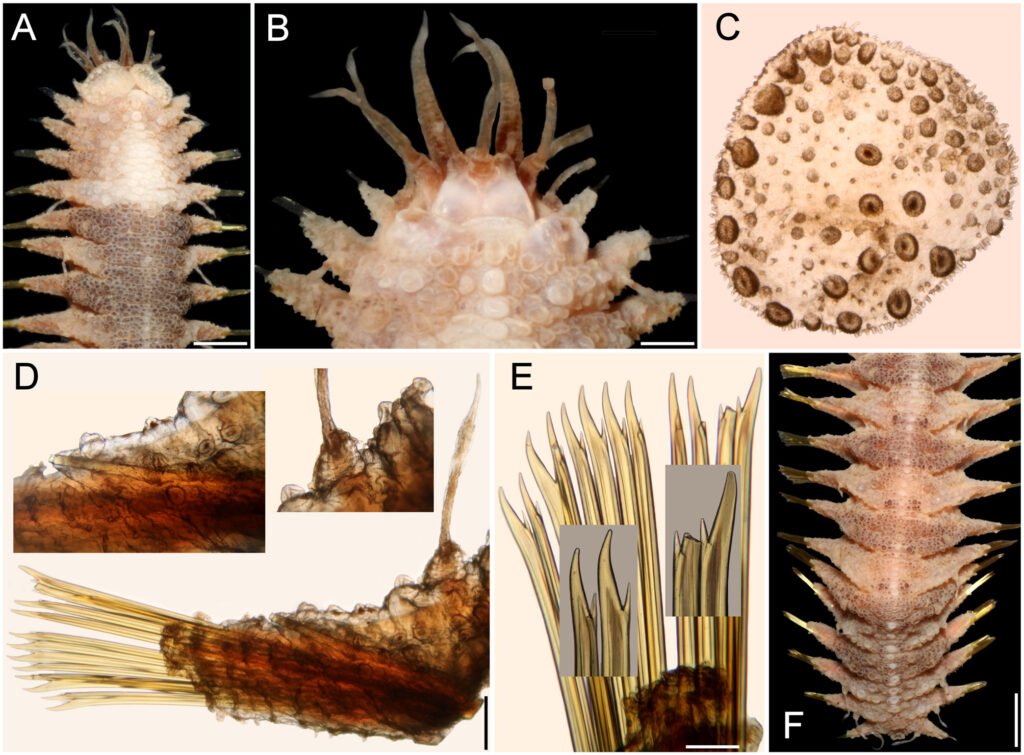
Posterior region banded (Fig. 10F), elytrigerous chaetigers with dorsal surface darker, cirrigerous chaetigers paler; pygidium with anus dorsal, between chaetigers 25-26, anal cirri lost.
Taxonomic summary
Type material. Caribbean Sea. Holotype (ECOSUR 1396), Chinchorro Bank, Cayo Norte, R/V Edwin Link, Sta. 2774 (18º45.63’ N, 87º15.84’ W), 55 m, August, 1990 E. Escobar, L. Soto, J.L. Villalobos, coll.
Distribution. Western Tropical Atlantic (Bahamas to Venezuela), in shallow mixed bottoms.
Etymology. The species is named to honor the late Dr. Aaron Treadwell in recognition of his contributions to the knowledge of different families of polychaetes.
Additional material. Northwestern Atlantic. One specimen (USNM 3296), USFCS Albatross, Sta. 2246 (39°56’45” N, 70°20’30” W), off New Jersey, U.S.A., Sta. 188, 220 m, 26 Sep. 1884 (posterior region bent ventrally; dorsum of chaetigers 2-5 with a discontinuous inverted T-shaped white spot; body 22 mm long, 5.5 mm wide, 26 chaetigers).
Bahamas. One specimen (USNM 16492), Holding Key, Andros Island, 13 May 1912, P. Bartsch, coll. (slightly bent laterally; chaetigers 2-5 with dorsum almost completely white; body 20 mm long, 5 mm wide, 26 chaetigers).
One specimen (USNM 46921), outer reef, point north of Clarencetown, Long Island, 15 May 1970, C. Riser, coll. (chaetigers 4-5 with a wide, inverted T-shaped white band, interrupted laterally; median and posterior nephridial lobes brownish; body 28.5 mm long, 5.5 mm wide, 26 chaetigers).
Florida. One specimen (USNM 17722), Tortugas, 1914, A.L. Treadwell, coll. (dorsum brownish, with inverted T-shaped wide band in chaetigers 4-5; body nephridial lobes from chaetiger 8, progressively larger posteriorly; 34 mm long, 7 mm wide, 26 chaetigers).
Caribbean Sea. One specimen (ECOSUR 9), Majagual, Quintana Roo, México, 6 Jun. 1998, M.A. Ruiz-Zárate, coll. (juvenile; slightly twisted; no dissected to avoid further damage; without nuchal lappet; anterior region with inverted T-shaped white spot along chaetigers 4-5; anus in chaetiger 26; body 11 mm long, 3 mm wide, 26 chaetigers).
One specimen (ECOSUR 1305), Majagual, Quintana Roo, México, 6 Jun. 1998, M.A. Ruíz-Zárate, coll. (juvenile, complete; bent ventrally; chaetigers 1-5 completely white; nuchal lappet distinct; anus terminal in chaetiger 26; body 11 mm long, 3.5 mm wide, 26 chaetigers).
One specimen (ECOSUR 1312), Xcacel, Quintana Roo, México, 17 Apr. 1996, S.I. Salazar-Vallejo & L.F. Carrera-Parra, coll. (complete; bent ventrally; pharynx exposed, 10 pairs of marginal papillae; chaetigers 1-5 completely white; nuchal lappet short; anus terminal in chaetiger 26; body 18 mm long, 5 mm wide, 26 chaetigers).
One specimen (ECOSUR 1318), Venezuela, Margarita Island, Los Testigos, seagrasses, 6 Jun. 1961 (partially dehydrated; anterior region with inverted triangle white spot along chaetigers 4-5; nuchal lappet distinct; anus in chaetiger 26; body 19 mm long, 5 mm wide, 26 chaetigers).
Two specimens (ECOSUR 1394), Punta Nizuc, Cancún, Quintana Roo, coral rocks, 1 Sep. 1997, S.I. Salazar-Vallejo, L.F. Carrera-Parra & M.A. Ruiz-Zárate, coll. (complete, fusiform; first 5 chaetigers almost completely white; nuchal lappet short; anus terminal in chaetiger 26; body 20-27 mm long, 5.5-7.5 mm wide, chaetigers).
One specimen (ECOSUR 1395), Majagual, Quintana Roo, reef lagoon, rocks, 18 Mar. 2002, S.I. Salazar-Vallejo, coll. (complete, fusiform; first 5 chaetigers almost completely white; nuchal lappet distinct; anus terminal in chaetiger 26; body 28.5 mm long, 7 mm wide, chaetigers).
One specimen (ECOSUR 2429), Punta Herradura, Quintana Roo, México, 28 Oct. 1997, P. Salazar-Silva, coll. (bent laterally; first right elytron and left parapodium of chaetiger 5 previously removed, kept in container; anterior region with inverted T-shaped white spot along chaetigers 4-5; anus prolapsed, in chaetiger 26; body 21 mm long, 5.5 mm wide, 26 chaetigers).
One specimen (LACM 14762), British Virgin Islands, Guana Island, Beef Island, long white beach at far west end of airport property, (18.448° N, 64.541° W), coral rubble, 1-2 m, snorkel, hand, 23 Jul 2000. T. Zimmerman, T. Haney, R. Ware, D. Cadien, coll. (barely bent ventrally, with salt particles adsorbed on body; chaetigers 4-5 with an inverted T-shaped white band; body 26.5 mm long, 7 mm wide, 26 chaetigers).
One specimen (LACM 147883), Jamaica, Saint Ann Bay, Bull Reef (18.735° N, 77.289° W), coral rubble and algae, 8 m, 2 Jun. 2006, K. Rawlinson, M. Bolanos, A. DuPont, A. Allan, J. Dunn & L. Harris, coll. (slightly bent ventrally, pharynx partially exposed; chaetigers 2-5 completely white dorsally; body 14.5 mm long, 3.5 mm wide, 26 chaetigers).
Three specimens (LACM 147910), British Virgin Islands, Guana Island, White Bay, ARMS (18.3° N, 64.37° W), 9 m, 15 Jul. 2000, J. Zimmerman, J. Martin & R. Ware, coll. (smaller ones almost colorless, largest bent ventrally, with an inverted whitish T-shaped spot along anterior chaetigers; body 7-26 mm long, 2.0-6.5 mm wide, 26 chaetigers).
One specimen (UMML 22.729b), University of Miami, Cruise P7006, Hispanola and Jamaica, RV Pillsbury, Sta. 1198 (17°49.4’ N, 76°12.3’ W), 29-37 m, 4 Jul. 1970 (bent ventrally; smaller one with inverted T-shaped white spot in chaetigers 2-5, anus terminal in chaetiger 26; body 15 mm long, 5 mm wide, 26 chaetigers).
One specimen (UMML P341), University of Miami, Cruise 6607, Panama to Venezuela, R/V Pillsbury, Sta. 341 (09°02’ N, 77°02’ W), 44 m, 9 Jul. 1966 (anterior region with an inverted T-shaped spot in chaetigers 2-5; first left elytron and left parapodia of chaetigers 3, 4, 13 previously removed (elytron and 1 parapodium kept in container); anus in chaetiger 26; body 15 mm long, 5 mm wide, 26 chaetigers).
Three specimens (UMML P1148), University of Miami, Cruise P7006, Hispanola and Jamaica, RV Pillsbury, Sta. 1148 (20°00’ N, 71°41’ W), 38 m, 1 Jan. 1970 (anterior region with inverted T-shaped spot in chaetigers 2-5; 1 specimen with anterior end dissected, several parapodia and elytra previously remove, kept in container; nuchal lappet distinct; dorsal cirri completely pale; body 21-24 mm long, 6-7 mm wide, 26 chaetigers).
One specimen (UMML P1330), RV Pillsbury, Cruise P7101, Central America, off Nicaragua, Sta. 1330 (11°51’ N, 83°27’ W), 24 m, 28 Jan. 1971 (soft, bent ventrally; chaetigers 4-5 with an inverted triangle white spot; nuchal lappet indistinct; anus terminal in chaetiger 26; body 21 mm long, 6 mm wide, 26 chaetigers).
One specimen (USNM 951), off Colon, Panama, USFSS Albatross, Sta. 2146 (9°32’00” N, 79°54’30” W), 61 m, 2 Apr. 1884, 61 m. (bent ventrally and laterally; chaetigers 4-5 with an inverted T-shaped dorsal white band; body 26 mm long, 7.5 mm wide, 26 chaetigers).
One specimen (USNM 20489), Old Providence, Colombia, coll. W.L. Schmitt, coll., 6 Aug. 1938, shore, reef and tide pool. (bent ventrally, chaetigers 2-5 with an inverted T-shaped white band; body 22 mm long, 4 mm wide, 27 chaetigers).
One specimen (USNM 46912) St. James Island, near St. Thomas, 4.5 m, 10 Jul. 1915, C.R. Shoemaker, coll. (body slightly bent laterally; dorsum of chaetigers 4-5 with a discontinuous inverted T-shaped white spot; anterior chaetigers with notochaetae; body 21 mm long, 5.5 mm wide, 26 chaetigers).
One specimen (USNM 46915), Barbuda, around Spanish Point, in beach wrack, fossil coral and live Porites, Sta. 112-58, 28 Apr. 1958, W. L. Schmitt, coll. (chaetigers 4-5 with a wide, inverted T-shaped white band; left parapodia of chaetigers 22-23 broken; breaking in posterior end; body 25 mm long, 6 mm wide, 27 chaetigers).
One specimen (USNM 46919), off Loggerhead Key, Dry Tortugas, Florida, 6 m, matrix of brain coral, 19 Aug. 19 1966, R. F Cressey, coll., & donor, (only 1 of the 2 specimens identified by M. H. Pettibone: wider slightly bent ventrally, thinner with first few chaetigers almost completely white, wider with 3 white spots in chaetiger 6 and a median on in chaetigers 4-5; body 16-18 mm long, 5.0-5.5 mm wide, 26 chaetigers).
One specimen (USNM 46922), U.S. Virgin Islands, 90 m, Sep. 1970, J. Clark, coll. (chaetigers 4-5 with a wide, inverted T-shaped white band; many chaetae broken; posterior end bent ventrally; median and posterior nephridial lobes brownish; body 21.5 mm long, 6.5 mm wide, 26 chaetigers).
No location. USNM 34285 (2), w/o field data, 11 Jan. 1966. (soft, barely pigmented, 1 with a dorsal white spot, the other with a discontinuous inverted T-shaped white spot in chaetigers 4-5; smaller specimen with several left posterior parapodia previously removed, some in container; body 24-28 mm long, 6-7 mm wide, 26 chaetigers).
Remarks
Hermenia treadwelli sp. nov. is distinguished by having a body fusiform, depressed, wider medially, venter wrinkled, with papillae thick, abundant; nuchal lappet thick, shorter than in H. neoverruculosa. Dorsum with globular tubercles of different sizes, some prominent giving a crowded appearance, parapodia short; from the second pair of elytrophores, elytra smaller, fleshy, barely covering elytrophores, firmly attached.
Hermenia treadwelli differs from H. verruculosa by having a body depressed, fusiform, venter with papillae thick, long, segment 2 with a well-developed nuchal lappet, on anterior part of the dorsum with a T-shaped spot with pale tubercles, extended along almost the whole segment 6, and middorsal areas of segment 5, whereas in H. verruculosa the body is subrectangular, venter with papillae thin, short, without nuchal lappet on prostomium, and the small spot of unpigmented tubercles is small and extended middorsally along segments 5-6.
Further, H. treadwelli differs from H. neoverruculosa because its nuchal lappet is shorter, neurochaetae only have 1 accessory tooth, the venter has papillae instead of being smooth, and neurochaetae with 2 accessory teeth.
After the body shape, being depressed, and retracted prostomium, venter with thick papillae, H. treadwelli body indicates a species with reduced mobility in comparison with H. verruculosa or H. neoverruculosa.
Hermenia wehei sp. nov.
(Fig. 11)
http://zoobank.org/urn:lsid:zoobank.org:act:08AE4922-3010-4327-B8AA-9753DD0AE1D1
Hermenia verruculosa: Wehe, 2006: 80-81, Pl. 1, Fig. d (non Grube & Örsted in Grube, 1856; Socotra Island, Indian Ocean, 8-10 m).
Diagnosis. Hermenia with body subrectangular; first pair of elytra bigger, cover prostomium, remaining elytra tiny; dorsal integument with abundant globular tubercles, without larger ones along middline; neurochaetae with 2 o 3 accessory teeth.
Description. Holotype (SMF 136089) complete, bent ventrally (Fig. 11A), slightly wider anteriorly, 26.5 mm long, 18 mm wide, 26 chaetigers. Dorsum almost without pigmentation (pale with elytrigerous segments brownish, cirrigerous whitish; elytra and a few middorsal tubercles brownish); dorsal tubercles medium sized, larger middorsal tubercles missing. Venter almost smooth.
Prostomium markedly retracted into segment 2 (Fig. 11B); facial tubercle not seen; eyes not seen. Median ceratophore slightly wider and longer than laterals; left lateral antenna present, almost half as long as palps. Palps bent ventrally, with abundant papillae in longitudinal rows.
Tentacular segment indistinct dorsally. Tentaculophores thick, cylindrical, without chaetae. Tentacular cirri subequal, almost as long as palps. Second segment narrow middorsal, without large globular tubercles, without nuchal lappet; elytrophores not expanded anteriorly; right ventral cirrus lost, left one about 6 times longer than the following one.
First pair of elytra largest, pale, completely cover prostomium; elytral surface with abundant macrotubercles markedly surpassing elytral margins (Fig. 11C), and smaller microtubercles; macrotubercles globular, longer than wide, surface spinulose, present along all elytral surface, some papillae present along inner margin. Following elytra of similar size, circular, not removed to avoid further damage, with macro- and microtubercles, mostly globular.

Parapodia short, 1/4 as long as body width; dorsally with transverse rows of globular tubercles along notopodia, remaining surface with papillae, ventrally with transverse pleats and scattered papillae. Notopodia reduced, acicular lobe small, with an apical papilla. Neuropodia truncate, dorsally furrowed, prechaetal lobe slightly larger than postchaetal one, with some distal papillae.
Cirrigerous segments with cylindrical, thick, short cirrophores, cirrostyles smooth, long, almost smooth, reaching the neuropodial tip (Fig. 11D). Nephridial papillae low, blunt, from chaetiger 12, continued to chaetiger 25, progressively larger.
Neurochaetae amber color. Most body segments with 2-3 notochaetae, curved, marginally denticulate. Neurochaetae decreasing in size and width ventrally, most with 2 accessory teeth, on lower position (Fig. 11E).
Posterior region tapered, truncate; pygidium with anus terminal, anal cirri lost (Fig. 11F).
Taxonomic summary
Type material. Arabian Sea. Holotype (SMF 136089, Socotra Island, Sta. 726, 8-10 m, 9 Apr. 2000, T. Wehe, coll.
Distribution. Known only from Socotra Island, Arabian Sea, and from Saya de Malha Bank, Indian Ocean; in subtidal substrates (8-26 m).
Etymology. The specific epithet is after Dr. Thomas Wehe, in recognition of his relevant publications on scaleworms, and especially because he collected the holotype.
Additional material: Indian Ocean, Saya de Malha Bank.One specimen (MNHN IA 2017-4879), Saya Expedition, RV Agulhas II, Sta. YS08 (09°41.9’ S, 60°51’ E), 26 m, 8 Nov. 2022, S. Hourdez, coll. (complete, bent ventrally, with ill-defined transverse bands in elytrigerous segments; first elytra pair markedly larger than following ones, barely covering prostomial sides; pharynx everted, 4 mm long, with 10 pairs of terminal papillae; right parapodium of chaetiger 15 removed for observation; neurochaetae with a single accessory denticle (rarely double); body 19 mm long, 5 mm wide, 26 chaetigers).
One specimen (MNHN 2021-727), Saya Expedition, RV Agulhas II, Sta. YS08 (16°50’ S, 59°31.3’ E), 17 m, 20 Nov. 2022, S. Hourdez, coll. (complete, bent ventrally with ill-defined transverse bands in elytrigerous segments; first elytra pair markedly larger than following ones, barely covering prostomial sides; left parapodia of chaetigers 16-19 removed for molecular studies; body 23 mm long, 6.5 mm wide, 26 chaetigers).
Remarks
Hermenia wehei sp. nov. belongs to the group of species (H. verruculosa, H. treadwelli) having only the first pair of elytra markedly larger than the following ones. However, H. wehei separates from the other species regarding differences in the first elytra, the development of middorsal tubercles, and in neurochaetal tips. Thus, H. wehei has the first elytra with macrotubercles clearly projected beyond the margin (against barely projected), medium-sized middorsal tubercles (against having larger ones middorsally), and its neurochaetae are bidentate or tridentate (against only bidentate in the other species).
Key to species of Hermenia Grube & Örsted in Grube, 1856
1 First 2-3 anterior pairs of elytra markedly larger than posterior ones; neurochaetae of median segments with 2 accessory teeth
………………………………………………………………………………2
– Only first pair of elytra markedly larger than posterior ones; neurochaetae of median segments with 1 accessory denticle, rarely with 2 accessory teeth
………………………………………………………………………………5
2 (1) Dorsum whitish, papillate or barely tuberculate; elytral macrotubercles globular, basally swollen or L-shaped (inner projection smaller)
………………………………………………………………………………3
– Dorsum grayish, clearly tuberculate; tubercles arranged in transverse series; elytral macrotubercles globular, no L-shaped
………………………………………………………………………………4
3 (2) Median and posterior elytra covering up to half of adjacent segments, elytra round, last one slightly larger than previous one
………………………………………………………………………………H. acantholepis (Grube, 1876) Philippines
– Median and posterior elytra barely covering adjacent segments, elytra oval to circular, last elytra as large as previous one
………………………………………………………………………………H. mezianei sp. nov. Vietnam
4 (2) Dorsum with rows of globular tubercles, each with blackish core; median and posterior elytra with black macrotubercles
………………………………………………………………………………H. neoverruculosa Pettibone, 1975 Cargados Carajos, Indian Ocean
– Dorsum with rows of depressed tubercles, each with pale core; median and posterior elytra with pale and brownish macrotubercles
………………………………………………………………………………H. chuarae sp. nov. Indonesia
5 (1) Neurochaetae with a single accessory tooth; middorsal surface with large globular tubercles; first left elytron with macrotubercles barely projected beyond margin
………………………………………………………………………………6
– Neurochaetae with 1-2 accessory teeth; middorsal surface without large globular tubercles; first left elytron with macrotubercles distinctly projected beyond margin
………………………………………………………………………………H. wehei sp. nov. Arabian Sea
6 (5) Segment 2 with nuchal lappet over prostomium; dorsal cirrostyle of median segments with swollen area brownish (fresh specimens with chaetigers 1-5 almost completely white, or with a large white inverted triangular spot over the dorsum of the chaetigers 3-5)
………………………………………………………………………………H. treadwelli sp. nov. Grand Caribbean Sea
– Segment 2 without nuchal lappet over prostomium; dorsal cirrostyle of median segments with swollen area whitish (fresh specimens with a middorsal white rectangular spot, or dorsum completely dark along chaetigers 5-6, sometimes separated by brownish area into 3 spots)
………………………………………………………………………………H. verruculosa Grube & Örsted in Grube, 1856 U.S. Virgin Islands, Caribbean Sea
Parahermenia gen. nov.
http://zoobank.org/urn:lsid:zoobank.org:act:5604018E-9BC0-4D3C-908F-1E355BAEFE7A
Diagnosis. Lepidonotinae with short body; integument papillate, venter papillate. Body with 26 segments, 12 pairs of elytra on segments 2, 4, 5, 7, 9, 11, 13, 15, 17, 19, 21, 23. Prostomium bilobed, with 2 pairs of eyes. Elytra with globular macrotubercles and microtubercles, abundant papillae, fimbriae very long; elytra of similar size along body, overlapping laterally, not dorsally. Tentacular segment with few notochaetae and bulbous facial tubercle. Parapodia subbiramous, notopodia small, neuropodia large. Notochaetae long, slender, finely spinous. Neurochaetae stout, falcate, with 1-2 large accessory teeth. Dorsal cirri with cirrophores cylindrical, cirrostyles short. Ventral cirri short, subulate. Pharynx with 2 pairs of jaws and 9 pairs of marginal papillae. Nephridial papillae short, cylindrical, from chaetigers 6-8.
Taxonomic summary
Type species. Lepidonotus hermenioides Amoureux, 1974.
Distribution. Parahermenia includes 2 species found in shallow water sediments, 1 from Madagascar, the other from the Philippines.
Etymology. Parahermenia is made by combining the stem genus name Hermenia with the Greek prefix para-, meaning “besides, nearby” (Brown 1954: 587), and this new name indicates the affinities to the older genus group name. Gender: Feminine.
Remarks
Pettibone (1977:40) noted that there are 3 genera with long filiform papillae on the distal end of neuropodia. She proposed 2 new genera, Lepidonopsis Pettibone, 1977 and Parahalosydnopsis Pettibone, 1977, which, together with Halosydnopsis Uschakov & Wu, 1959, were the only genera provided with filiform papillae. The number of pairs of elytra and neurochaetal tips separates these 3 genera because Lepidonopsis has 12 pairs and bidentate neurochaetae, Halosydnopsis has 17 and unidentate neurochaetae, and Parahalosydnopsis has about 30 pairs and unidentate neurochaetae. These 3 genera have small denticles in series along the pectinate area.
Lepidonotus hermenioides does not belong in Lepidonopsis although it has 12 pairs of elytra, because each elytron is provided with very long fimbria, only 2 macrotubercles in the insertion area and the elytra do not overlaped dorsally, whereas in Lepidonopsis the elytra overlap covering the body, have short fimbriae and different types of microtubercles (Salazar-Silva & Carrera-Parra, 2014). On the other hand, L. hermenioides does not fall within Hermenia despite sharing a papillate integument, and neurochaetae with 1-2 accessory teeth. The main differences are in the elytra. In L. hermenioides elytra are of similar size along the body, overlapping laterally but not dorsally, and their margin has abundant long fimbriae, whereas in the species of Hermenia median and posterior segments have elytra that do not overlap laterally or dorsally, and fimbriae are minute.
Amoureux (1974) avoided proposing a new genus because he only had a single specimen, and it was regarded as a very small juvenile. He might have suspected some ontogenetic modifications that would complicate separating the new group. We agree this and the new species described below include small specimens; however, after the study of many specimens of different size, we do not find evidence that elytra would be so drastically modified such that they would become progressively smaller during growth, or that the elytral margins would be modified from having very long fimbriae to becoming progressively reduced to become minute.
Parahermenia hermenioidea (Amoureux, 1974) comb. nov.
(Fig. 12)
Lepidonotus hermenioides Amoureux, 1974: 131, Fig. 1.
Diagnosis. Parahermenia with body papillate, elytra with long filamentous papillae, without macrotubercles or spines, margin fimbriate; neurochaetae with 2-3 accessory large teeth.
Description. Holotype (MNHN POLY TYPE 1275) complete, brownish, with abundant papillae along body (Fig. 12A); pharynx fully exposed; elytra paler with long fimbriae, and filaments on elytral surface; first pair of elytra on site, right elytra 10 and 12 on site; right parapodia of chaetigers 10, 12, 14, and left parapodia of chaetigers 13 and 14 previously removed (1 elytron and 1 parapodium in container). Body depressed, wider anteriorly, 3.5 mm long (pharynx 0.8 mm long), 1.2 mm wide (without chaetae), 23 chaetigers.
Prostomium oval, as long as wide; only left lateral antenna, ceratostyle on site, short, all other lost (Fig. 12B); median antenna ceratophore about twice wider than laterals. Palps lost. Eyes indistinct. Pharynx cylindrical, grayish, with 9 pairs of marginal papillae.
Facial tubercle globular, small. Tentacular segment indistinct dorsally; second segment chaetiger without nuchal lappet, dorsum without tubercles; tentacular cirri present on left side, 4-5 times longer than left antenna, subdistally swollen, with 4-5 chaetae.
Ventral cirri of chaetiger 2 are about 3-4 times longer than the following ones, surpassing neurochaetal tips. Nephridial lobes barely developed, not visible.
Elytra oval, with 2 globular macrotubercles in the insertion area, each with small conical papillae; microtubercles conical over elytral surface, and long filaments on surface, margin with long fimbriae, filaments 1/2-1/3 elytral length (Fig. 12C).
Parapodia biramous. Notopodia reduced, low round lobes. Dorsal cirri resembling tentacular cirri, barely swollen subdistally. Notochaetae thin, with a single series of fine denticles (Fig. 12D).
Neuropodia larger, prechaetal and postchaetal lobes blunt, papillate. Neurochaetae barely swollen subdistally, unidentate, with 2-3 large subdistal accessory teeth (Fig. 12E).
Posterior region truncate; pygidum with anus terminal, anal cirri tapered, resembling dorsal cirri.

Taxonomic summary
Type material. Madagascar. Holotype of Lepidonotus hermenioides Amoureux, 1974 (MNHN POLY TYPE 1275), Nosy Tanikely, Madagascar, comb-dredge (faubertage) 1, 15 m, 6 Apr. 1960, G. Cherbonnier, leg.
Distribution. Only known from a single locality in Madagascar, in shallow rocky bottoms.
Remarks
Parahermenia hermenioidea (Amoureux, 1974) comb. nov. needed an alteration of the specific name for matching the genus gender. This is regarded as an adjective and, after Brown (1954: 483), the suffix –oid, –oides, –odes, –oideus, –a, –um, are derived from the Greek suffix –eides meaning “like, resembling, having the form of”. He also indicated that –oideus, –a, –um have their gender indicated by the adjectival endings –us, –a, –um.
Amoureux (1974) noted that the neurochaetae of Lepidonotus hermenioides resembles those present in Hermenia, and this explains the specific name. The generic affinities are problematic because L. hermenioides does not match Hermenia by having median and posterior elytra larger, with long fimbriae and long papillae along its surface, instead of large globular macro- and microtubercles and short fimbriae, as revised Pettibone (1975).
Parahermenia piotrowskiae sp. nov.
(Fig. 13)
http://zoobank.org/urn:lsid:zoobank.org:808A56BB-18
7E-4E60-97D9-DF4308793F2E
Diagnosis. Parahermenia with body papillate, elytra with globular macrotubercles and a single spine, and few filamentous papillae, margin fimbriate; neurochaetae with 2-3 accessory large teeth.
Description. Holotype (CAS 217917), complete, markedly bent ventrally, with abundant papillae along body; dorsum and vente white, first and last pairs of elytra whitish, first with 3 colorless macrotubercles, arranged in a triangle, last elytra with a single colorless globular macrotubercle; other elytra with globular brownish macrotubercles centrally, and long filamentous fimbriae. Second pair of elytra with 3 large globular central macrotubercles, arranged in a triangle (Fig. 13A), following elytra with smaller macrotubercles, arranged in an oblique row or a triangle (Fig. 13B). Some left median and posterior elytra detached; right elytron 6 and left parapodium of chaetiger 12 removed for observation (all kept in container). Body depressed, slightly tapered posteriorly, 6 mm long, 2 mm wide, 26 chaetigers.
Prostomium hexagonal, about as long as wide; median ceratophore 2 times as large and long as laterals, median ceratostyle lost; lateral ceratostyles tapered, subdistally swollen, 1/5 as long as palps, with reddish bands below and over swollen area, tips reddish. Palps papillate, with 2 longitudinal lateral reddish bands, middorsal area pale, with a subdistal reddish band, tips darker. Eyes black, of similar size, anterior eyes in wider prostomial area, posterior ones close to posterior margin.
Facial tubercle globular, small. Tentacular segment indistinct dorsally; second chaetiger without nuchal lappet, dorsum with transverse reddish bands, without tubercles; tentacular cirri resembling lateral antennae in pigmentation, slightly shorter than palps, chaetae not seen.
Ventral cirri of chaetiger 2 lost. Nephridial lobes not seen. Median elytra oval, overlapping laterally, not dorsally. Each with 2-3 large, central reddish or brownish globular macrotubercles, some with 4 (Fig. 13C), others with 2-3 macrotubercles, a single large falcate spine external to globular macrotubercles, and sparse papillae, most short (about as long as wide), a few 4-6 times longer than wide; fimbriae with cylindrical filaments, some distally swollen, 1/10-1/7 elytral length.
Parapodia biramous (Fig. 13D). Dorsal cirri resembling tentacular cirri, barely swollen subdistally, with brownish bands before and after a swollen dark area (Fig. 13D), ceratostyle with small papillae. Notochaetae thin, with a single series of fine denticles (Fig. 13D, inset below).
Neuropodia larger, prechaetal and postchaetal lobes blunt, papillate. Neurochaetae barely swollen subdistally, unidentate, with 2 large subdistal teeth (Fig. 13E).
Posterior region truncate; pygidium with anus terminal (Fig. 13F), anal cirri resembling dorsal cirri.
Taxonomic summary
Type material. Philippine Islands. Holotype (CAS 217917), Verde Island Passage Expedition 2016, Luzon Island, Batangas Province, Tingloy, Maricaban Island, Devil’s Point (13.65°N, 120.84°E), coral rubble, 40 m, 20 Apr. 2016, C. Piotrowski, coll.
Distribution. Only known from a subtidal locality (40 m) in Luzon Island, Philippines.
Etymology. This species is being named after M.Sc. Christina N. Piotrowski, Collection Manager of the Invertebrate Zoology collections in the California Academy of Sciences, in recognition of her support of our research activities, and because of her sampling efforts in many tropical localities of the world, especially because she collected the holotype.
Remarks
Parahermenia piotrowskiae sp. nov. resembles P. hermenioidea, the other only known species in the genus, because both have small bodies, and their elytra have long filamentous fimbriae. Their main differences are in the elytral ornamentation. In P. piotrowskiae elytra have globular, usually pigmented macrotubercles and a single spine, sparse filamentous papillae, and the fimbriae filaments are short, 1/10-1/7 as long as elytral length, whereas in P. hermenioidea the macrotubercles are markedly smaller, and there are no large spines, its filamentous papillae are long and abundant, and the fimbriae filaments are long, 1/2-1/3 of elytral length.
Key to species of Parahermenia gen. nov.
1 Median segments with elytral surface provided with large, reddish or brownish macrotubercles, a single large spine, papillation sparse; elytral fimbriae short (1/10-1/7 elytral length)
………………………………………………………………………………P. piotrowskiae sp. nov. Philippines
– Median segments with elytral surface provided with small, pale macrotubercles, without spines, papillation abundant; elytral fimbriae long (1/2-1/3 elytral length)
……………………………………………………………………………… P. hermenioidea (Amoureux, 1974) comb. nov. Madagascar.
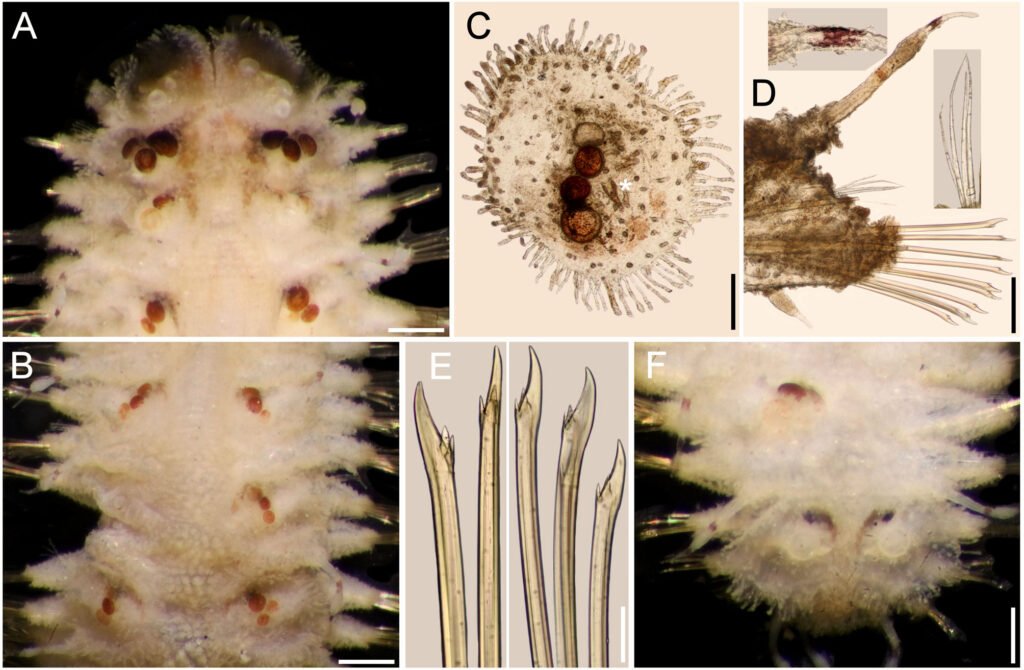
Discussion
The taxonomy of polynoid scaleworms has relied on morphological characters, such as the arrangement of cephalic appendages (subfamilies), and the features of the elytral and parapodial structures, including chaetae. Among Lepidonotinae, the traditional approach has been to use the number of pairs of elytra and other parapodial features for separating similar genera. In the group of lepidonotin genera provided with 12 pairs of elytra, the distinguishing features include the presence of some parapodial features (branchiae, pseudelytra) or types of chaetae. We have confirmed the distinguishing features of Hermenia such as the presence of reduced elytra along most body segments, and that neurochaetae have subdistal smooth regions, as opposed to most other genera having spinulose regions, and tips bi- or tridentate, opposed to being uni- or bidentate.
We have also refined the use of some distinguishing features for separating similar Hermenia species, and for newly describing other ones. They include the modifications of the nuchal lappet (distinctive, or indistinct), body integument (tuberculate, papillate, or both) and pigmentation, shape of elytra (mostly circular, some oval, wider than long), elytral size along body (similar to body end, or last pair larger than median ones), type of macrotubercles (most ovoid, a few basally swollen), and tips of neurochaetae (bi- or tridentate). Hermenia now includes 3 previously known species: H. verruculosa, described from the Caribbean Sea (type species), H. acantholepis, described from Philippines, and H. neoverruculoa, from Republic of Mauritius, and 4 new species: H. chuarae sp. nov. from Indonesia, H. mezianei sp. nov. from Vietnam, H. treadwelli sp. nov. from the Grand Caribbean, and H. wehei sp. nov. from the Arabian Sea and Indian Ocean.
Two other species, one already described, and another undescribed, resembled Hermenia species by having body papillate and neurochaetae subdistally smooth, but differed by having larger elytra overlapping laterally along body (restricted to a few anterior pairs in Hermenia), and elytra provided with long fimbria (short in Hermenia) and a few macrotubercles (very abundant in Hermenia). We proposed Parahermenia gen. nov. for these 2 species and separated them after the size of elytral fimbriae and number of macrotubercles. The new genus contains P. hermenioidea comb. nov. from Madagascar, and P. piotrowskiae sp. nov. from the Philippines. An interesting fact that no species of Hermenia or Parahermenia have been found along the Eastern Pacific or Western African coasts.
Acknowledgments
To Christina Piotrowski (CAS), Danny Eibye-Jacobsen and the late Mary Petersen (ZMUC), Leslie Harris (LACM), Marie-Louise Tritz and Ekin Tilic (SMF), Miranda Lowe and Emma Sherlock (BMNH), the late Nancy Voss and María Criales (UMML), Gisela Wegener and Angelika Brandt (ZMH), Fredrik Pleijel, Tarik Meziane, Laure Corbari and Stéphane Hourdez (MNHN), Gustav Paulay and Amanda Bemis (UF), kindly allowed us to study part of their collections. The then-known Consejo Nacional de Ciencia y Tecnología, México, funded the project Poliquetos del Gran Caribe (Conacyt 32529T), and provided a PhD scholarship to the first author (Conacyt 94497). The warm support by the late Kristian Fauchald and Len Hirsch, as well as Leslie Harris and David Ocker, Laure Corbari and Tarik Meziane, allowed us to make research visits to study their magnificent collections. Emilia González and Luis F. Carrera-Parra took care of the ECOSUR Reference Collection.
References
Amoureux, L. (1974). Annélides polychètes de Madagascar recueillies par G. Cherbonnier en 1960. Bulletin du Museum National d Histoire Naturelle, Paris, série 3, Zoologie, 217, 425–462.
Baird, W. (1865). Contributios towards a monograph of the species of Annelides belonging to the Aphroditacea, containing a list of the know species, and a description of some new species contained in the National Collection of the British Museum. Journal of the Linnean Society, Zoology, 8, 172–202. https://doi.org/10.1111/j.1096-3642.1865.tb02438.x
Bellan, G. (1964). Résultats scientifiques des campagnes de la Calypso dans l’Atlantique. Annélides polychetes. Annales de l’Institute Océanographique, 41, 301–314.
Ben-Eliahu, M. N. (1975). Polychaete cryptofauna from rims of similar intertidal vermetid reefs on the Mediterranean coast of Israel and in the Gulf of Elat: Sabellidae (Polychaeta Sedentaria). Israel Journal of Zoology, 24, 54–70. https://doi.org/10.1080/00212210.1975.10688411
Brown, R. W. (1954). Composition of scientific words: a manual of methods and a lexicon of materials for the practice of logotechnics. Baltimore: George W. King.
Chuar, C. H., Hadiyanto, H., & Lee, Y. L. (2021). Annotated checklist of polychaetes from deeper waters of the Sunda Strait and eastern Indian Ocean off southwest Java, Indonesia. Raffles Bulletin of Zoology, 36 (Supplement), 47–77. https://doi.org/10.26107/RBZ-2021-0030
Dawydoff, C. (1952). Contribution a l’étude des invertébrés de la faune marine benthique de l’Indochine. Bulletin Biologique de la France et de la Belgique, 37 (Supplement), 1–158.
Devaney, D. M. (1974). Shallow-water echinoderms from British Honduras, with a description of a new species of Ophiocoma (Ophiuroidea). Bulletin of Marine Science, 24, 122–164.
Ebbs, N. K. (1966). The coral-inhabiting polychaetes of the northern Florida reef tract, 1. Aphroditidae, Polynoidae, Amphinomidae, Eunicidae, and Lysaretidae. Bulletin of Marine Science, 16, 485–555.
Fauchald, K. (1977). The polychaeta worms: Definitions and keys to the orders, families and genera. Natural History Museum of Los Angeles County, Science Series, 28, 1–188.
Fauvel, P. (1922). Annélides polychètes de l’Archipel Hontman Abrolhos (Australie Occidentale) recueillies par M. le Prof. W.J. Dakin, F.L.S. Journal of the Linnean
Society,Zoology,34, 487–500. https://doi.org/10.1111/j.1096-3642.1922.tb01843.x
Fauvel, P. (1932a). Annelida Polychaeta of the Indian Museum, Calcutta. Memoirs of the Indian Museum, 12, 1–262.
Fauvel, P. (1932b). Mission Robert Ph. Dollfus en Égypte (Décembre 1927 – Mars 1929). Résumé analytique sur les polychètes. Bulletin de I institut d Égypte, 15, 131–144. https://doi.org/10.3406/bie.1932.3260
Fauvel, P. (1933). Mission Robert Ph. Dollfus en Égypte (Décembre 1927 – Mars 1929). Annélides polychètes. Bulletin de l’Institut d’Égypte, 21, 31–83.
Fauvel, P. (1935). Annélides polychètes de l’Annam. Memorie de lla Pontificia Accademia Romana dei Nuovi Lincei, série 3, 2, 279–354.
Fauvel, P. (1939). Annélides polychètes de l’Indochine recueillies par M. C. Dawydoff. Comment Pontificale Academie des Sciences, Civ Vaticana, 3, 243–368.
Fauvel, P. (1947). Faune de l’Empire Français, 8. Annélides polychètes de Nouvelle-Calédonie et des Iles Gambier. Paris: Office de la Recherche Scientifique Coloniale.
Fauvel, P. (1953a). The fauna of India including Pakiastan, Ceylon, Burma and Malaya: Annelida Polychaeta. Allhabad: Indian Press.
Fauvel, P. (1953b). Annélides polychétes de la Croisiére du Président Theodore Tissier aux Antilles. Bulletin de l’Institut Océanographique, Monaco, 1033, 1–23.
Grube, E. (1850). Die Familien der Anneliden. Archiv für Naturgeschichte, Berlin, 16, 249–364.
Grube, E. (1851). Uebersicht der Annelidengattungen und Arten zu ihrer vorläufigen Uterschidung. In Die Familien der Anneliden mit Angabe ihrer Gattungen und Arten. Ein systematischer Versuch (pp. 117–164). Berlin: Nicolai’schen Buchhandlung.
Grube, A. E. (1856). Annulata Örstediana Enumeratio Annulatorum, quae in itinere per Indiam occidentalem et Americam centralem annis 1845-1848 suscepto legit cl. A. S. Örsted, adjectis speciebus nonnullis a cl. H. Kröyer in itinere ad Americam meridionalem collectis. Havniae Part, 1, 44 – 62.
Grube, E. (1878). Annulata Semperiana. Memoires de l’Academie Imperiale des Sciences de St. Petersbourg, série 7, 25, 1–300.
Hanley, J. R., & Burke, M. (1991). Polychaeta Polynoidae: scaleworms of the Chesterfield Island and Fairway Reef, Coral Sea. In A. Crosnier (Ed.), Resultats des campagnes Musorstom 8 (pp. 9–82). Paris: Mémoires du Muséum National d’Histoire Naturelle.
Hartman, O. (1939). The polychaetous annelids collected on the Presidential Cruise of 1938. Smithsonian Miscellaneous Collections, 98, 1–22.
Herdman, W. A., & Hornell, J. (1903). Narrative with an outline of the investigation and details of the stations where observations were made. Report to the Government of Ceylon on the pearl oyster fisheries of the Gulf of Manaar, 1, 17–98.
Horst, R. (1917). Polychaeta Errantia of the Siboga-Expedition, 2. Aphroditidae and Chrysopetalidae. Siboga-Expeditie Monographie, Leyden, 24b, 1–140.
Horst, R. (1922). On some polychaetous annelids from Curaçao. Bijdragen tot de Dierkunde, 22, 193–201.
Imajima, M. (1997). Polychaetous annelids from Sagami Bay and Sagami Sea collected by the Emperor Showa of Japan and deposited at the Showa Memorial Institute, National Science Museum, Tokyo. Families Polynoidae and Acoetidae. National Science Museum, Tokyo Monographs, 13, 1–130.
Kinberg, J. G. H. (1856). Nya slägten och arter af Annelider. Animalia Annulata nova 1. Minus rite cognita recensuit. Öfversigt af Kongliga Vetenskaps-akademiens forhan-
dlingar, 12, 9–10, 381–388.
Monro, C. C. A. (1924). On the Polychaeta collected by H.M.S. ‘Alert’, 1881-1882. Families Polynoidae, Sigalionidae, and Eunicidae. Journal of the Linnean Society of London, Zoology, 36, 37–64. https://doi.org/10.1111/j.1096-3642.1924.tb02207.x
Monro, C. C. A. (1939). On some tropical polychaetes in the British Museum mostly collected by Dr. C. Crossland at Zanzibar, Tahiti, and the Marquesas, 1. Families Amphinomidae to Phyllodocidae. Annals and Magazine of Natural History, series 11, 4, 161–184.
O’Hara, T. D., Hugall, A. F., Cisternas, P. A., Boissin, E., Bribiesca-Contreras, G., Sellanes, J. et al. (2019). Phylogenomics, life history and morphological evolution of ophiocomid brittlestars. Molecular Phylogenetics and Evolution, 130, 67–80. https://doi.org/10.1016/j.ympev.2018.10.003
Pettibone, M. H. (1975). Review of the genus Hermenia, with a description of a new species (Polychaeta: Polynoidae: Lepidonotinae). Proceedings of the Biological Society of Washington, 88, 233–248.
Pettibone, M. H. (1977). Review of Halosydnopsis and related genera (Polychaeta: Polynoidae: Lepidonotinae). In D. J. Reish, & K. Fauchald (Ed.), Essays on polychaetous annelids in memory of Dr. Olga Hartman (pp. 39–62). The Allan Hancock Foundation, University of Southern California. Los Angeles.
Pettibone, M. H. (1993). Scaled polychaetes (Polynoidae) associated with ophiuroids and other invertebrates and review of species referred to Malmgrenia McIntosh and replaced by Malmgreniella Hartman, with descriptions of new taxa. Smithsonian Contributions to Zoology, 538, 1–92.
Pruvot, G. (1930). Annélides polychètes de Nouvelle-Calédonie recueillies par M. François. Archives de Zoologie Expérimentale et Générale, Paris, 70, 1–94.
Salazar-Silva, P., & Carrera-Parra, L. F. (2014). Revision of Lepidonopsis humilis (Augener, 1922) and description of L. barnichae sp nov (Annelida: Polychaeta: Polynoidae) based upon morphological and molecular characters. Zootaxa, 3790, 555–566. https://doi.org/10.11646/zootaxa.3790.4.4
Salazar-Vallejo, S. I. (1996). Lista de especies y bibliografía de poliquetos (Polychaeta) del Gran Caribe. Anales del Instituto de Biología UNAM, serie Zoología, 67, 11–50.
Salazar-Vallejo, S. I., & Eibye-Jacobsen, D. (2012). Annulata örstediana: Publication dates, composition and annotated taxonomic list, with some comments on Hemipodus (Polychaeta: Glyceridae). Revista de Biologia Tropical, 60, 1391–1402.
Seidler, H. J. (1923). Über neue und wenig bekannt Polychäten. Zoologischen. Anzeiger, Leipzig, 56, 254–264.
Seidler, H. J. (1924). Beitrage zur Kenntnis der Polynoiden, 1. Archiv für Naturgeschichte, 89A, 1–217.
Treadwell, A. L. (1911). Polychaetous annelids, from the Dry Tortugas, Florida. Bulletin of the American Museum of Natural History, 30, 1–12.
Wehe, T. (2006). Revision of the scale worms (Polychaeta: Aphroditoidea) occurring in the seas surrounding the Arabian Peninsula. Part 1. Polynoidae. Fauna Arabia, 22, 23–197.

Neoscelis septentrionalis, una especie nueva de Sonora, México (Coleoptera: Scarabaeidae: Cetoniinae)
Neoscelis septentrionalis, a new Mexican species from Sonora, Mexico (Coleoptera: Scarabaeidae: Cetoniinae)
Leonardo Delgado, Eder F. Mora-Aguilar * y Andrés Ramírez-Ponce
Instituto de Ecología, A.C., Red de Biodiversidad y Sistemática, Carretera Antigua a Coatepec Núm. 351, 91070 Xalapa, Veracruz, México
*Autor para correspondencia: eder.mora@inecol.mx (E.F. Mora-Aguilar)
Recibido: 3 marzo 2025; aceptado: 1 agosto 2025
http://zoobank.org/urn:lsid:zoobank.org:pub:C069AF28-A9C4-45C9-9F15-62F2537D6732
Resumen
Se describe una especie nueva mexicana de escarabajo del género Neoscelis Schoch, del estado de Sonora. Se compara a detalle con la especie más afín, N. longiclava Morón y Ratcliffe y se ilustran sus caracteres diagnósticos. Se incluye una clave para las 5 especies del género. Adicionalmente, se presenta el primer registro de N. dohrni (Westwood) para Zacatecas y se añaden datos sobre la variación de machos de N. longiclava.
Palabras clave: Taxonomía; Scarabaeoidea; México; Especie nueva
Abstract
A new Mexican species of the genus Neoscelis Schoch is described from the state of Sonora. The new species is compared in detail with the most similar species N. longiclava Morón y Ratcliffe, and its diagnostic characters are illustrated. A key to the 5 species of the genus is included. Additionally, the first record of N. dohrni (Westwood) for Zacatecas is presented, and new variation data of the males of N. longiclava are added.
Keywords: Taxonomy; Scarabaeoidea; Mexico; New species
Introducción
La tribu Goliathini Latreille, 1829 (Scarabaeidae: Cetoniinae) se distribuye principalmente en África y Asia, donde se conocen ca. 75 géneros y 521 especies de acuerdo con Krajcík (1998, 2012) y Schoolmeister (2024). En América se conocen solo 7 especies de 2 géneros endémicos de México, Ischnoscelis Burmeister, 1842 y Neoscelis Schoch, 1987 (Morón y Ratcliffe, 1989). Las relaciones filogenéticas de los goliatinos americanos no han sido esclarecidas; sin embargo, se ha mantenido la hipótesis que los relaciona con géneros del trópico asiático de la subtribu Rhomborhinina Westwood, 1842 (antes Coryphocerina Burmeister, 1842) (Morón y Ratcliffe, 1989).
Dentro del género Ischnoscelis se conocen 3 especies de Guerrero, Jalisco, México, Morelos y Sinaloa (Bouchard y Curoe, 2012; Curoe, 2013; Hernández-López et al., 2016). Para el género Neoscelis se han descrito 4 especies, N. coracina Mudge y Ratcliffe, 2003 del sur de Jalisco; N. dohrni (Westwood, 1855) de Aguascalientes, Colima, Durango, centro y norte de Jalisco, Nayarit y Sinaloa; N. hexakrotes García-Morales et al., 2019 de Guerrero y N. longiclava Morón y Ratcliffe, 1989 del oeste de Jalisco, descrito con 1 ejemplar macho (García-Morales et al., 2019; Morón y Ratcliffe, 1989, Mudge et al, 2003; Westwood, 1855). Con base en la revisión y comparación de especímenes de las especies del género Neoscelis en colecciones de México y EUA, describimos una especie de Neoscelis de Sonora.
Materiales y métodos
Se siguió la terminología y caracteres morfológicos usados por Morón y Ratcliffe (1989) y Mudge et al. (2003). La nomenclatura actualizada de las plantas siguió lo establecido en Tropicos (2025). Los ejemplares fueron estudiados con un estereomicroscopio Stemi SV6 Zeiss, las fotografías y medidas fueron tomadas con un estereomicroscopio Nikon SMZ25 y cámara DS-Fi2, las imágenes fueron editadas con el programa NIS-Elements software.
Se revisaron 32 ejemplares de especies de Neoscelis de la Colección Entomológica de la California Academy of Sciences (CASC), de la Colección Entomológica del Instituto de Ecología (IEXA) y de las colecciones particulares de Luis Leonardo Delgado Castillo (LLDC), Eder F. Mora-Aguilar (EMAC) y Andrés Ramírez Ponce (ARPC):
Neoscelis coracina Mudge y Raltcliffe (5): México: Jalisco, Ajijic (1 ♂, ARPC); Mazamitla, 1,720 m, 25-26-IX-2005, G. Nogueira col. (1 ♂, EMAC); idem excepto: 1,680 m, 02-X-2005 (2 ♀♀, EMAC); Puerta el Zapatero, 10-2009, fruit trap, Jesus Aguilar leg. (1 ♀ ARPC)
Neoscelis dohrni (Westwood) (10): México: Jalisco, Ixtlahuacán de los Membrillos, sierra El Travesaño, 29-IX-1995, 1,650 m, (4♂♂, IEXA); San Cristóbal de la Barranca, Mesa de los Caballos, 26-VII-2006, 1,671 m, D. Jimeno col. (1 ♂, EMAC); idemexcepto: 23-VII-2006 (1 ♂, EMAC); San Miguel de Hidalgo, 830 m snm, 15-IX-2003, G. Nogueira col (1 ♂, EMAC); Talpa de Allende, Los Sauces, VI-2012, 1,470 m, pino-encino, Fam. Jimeno-Sevilla cols. (1 ♂, EMAC); Tequila, volcán de Tequila, 17-X-2008, 1,380 m (2♂♂, IEXA).
Neoscelis hexakrotes García-Morales et al. (14): México: Guerrero,Acahuizotla, 1,120 m, VII/2017, local collector (3 ♂♂ paratipo, ARPC); Chilpancingo, 1.5 km NE de Acahuizotla, bosque tropical subcaducifolio, 915 m, X/2006, J. Juan López col (1 ♂ holotipo, 1 ♀ paratipo, ARPC); San Roque Mochitlán, 2-X-2018, L. Zacarías L. (1 ♂ paratipo ARPC); idemexcepto: 8-10/IX/2019, (2 ♂♂, 1 ♀ paratipo, ARPC); 18-20/IX/2019, L. Zacarías L. (5 ♂♂ paratipo, ARPC)
Neoscelis longiclava Morón y Ratcliffe (3): México: Jalisco,Estación de Biología Chamela, 7-IX-1989, E. Ramírez. R. A col (3 ♂♂, CAS, IEXA, LLDC).
Utilizamos el concepto filogenético de especie: “Una especie es la agregación más pequeña de poblaciones (sexual) o linajes (asexual) diagnosticables por una combinación única de estados de carácter” (Wheeler y Platnick, 2000). El mapa fue generado utilizando el sitio web SimpleMappr (Shorthouse 2010).
Resultados
Neoscelis septentrionalis sp. nov. Delgado, Mora-Aguilar y Ramírez-Ponce
(figs. 1A-F, 2)
http://zoobank.org/urn:lsid:zoobank.org:act:87061B5E-4746-4671-867C-57FA3873B69D
Diagnosis. Esta especie se diferencia fácilmente de las 4 especies conocidas del género Neoscelis por la combinación de 2 caracteres: maza antenal más larga que los artejos antenales precedentes y protarsómeros con sedas densas en forma de parches en su cara ventral.
Descripción. Holotipo macho: longitud desde ápice del clípeo al ápice de élitros: 19.8 mm, longitud del pronoto 6.6 mm, ancho del pronoto: 7.6 mm, longitud de élitros 12.0 mm, ancho humeral 9.3 mm. Color dorsal y ventral verde obscuro con tonalidades verde metálicas (fig. 1A).
Cabeza con cuerno clipeal corto, aproximadamente el doble de largo que ancho y ampliamente bifurcado casi desde su base. Clípeo rugoso-punteado, moderadamente ensanchado hacia el ápice; con tubérculo preapical a cada lado, divergentes; cóncavo a cada lado de la cresta media longitudinal, sobresaliente ampliamente. Frente irregularmente aplanada a ligeramente cóncava, con puntuación densamente rugosa y setígera, menos marcada hacia el vértice; sedas amarillentas de longitud moderada. Distancia interocular igual a 2.7 veces el ancho interocular de un ojo. Antenas con 10 antenómeros, maza antenal claramente más larga que 7 antenómeros anteriores, longitud del club antenal poco más de un tercio más largo que resto de antenómeros, menos de 3 veces más larga que ancha en vista dorsal (1:0.35) y 3 veces más larga que ancha en vista lateral (fig. 1B-C).
Pronoto semi-hexagonal, lados con reborde marginal, borde basal suavemente sinuado frente al escutelo. Ángulos anteriores, medios y posteriores obtusos y redondeados. Superficie pronotal con puntos setígeros de tamaño moderado y volviéndose rugosos, más grandes y densos hacia los márgenes; sedas pálido-amarillentas erectas, más cortas y finas que las que presenta en la cabeza. Escutelo ligeramente convexo, superficie con escultura similar a la del pronoto. Mesepímero densamente punteado y setígero, sedas finas y de longitud moderada.
Proceso meso-metasternal ligeramente prolongado y redondeado en vista ventral, cara anterior en 80° del plano ventral en vista lateral y con sedas largas y conspicuas. Meso y metasterno con sedas abundantes y largas, mayormente hacia la parte externa, puntuación rugosa transversal.
Protibia larga, esbelta y moderadamente curva, con 2 dientes preapicales pequeños. Protarsómeros ligeramente más largos en longitud que la protibia (incluyendo uñas), con sedas ventrales conspicuas, densas, café amarillentas (carece de los primeros 3 protarsómeros izquierdos (fig. 1A, D). Meso y metatibias esbeltas, ligeramente curvas, con tarsómeros respectivos más largos que su longitud. Meso y metatarsómeros con algunas sedas ventrales hacia el ápice.
Élitros con 3 costillas moderadamente elevadas y lisas, ápices poco marcados, superficiales, intervalos entre ellas con puntos densos de tamaño medio; superficie junto al margen, densa y finamente rugosa, transversalmente a partir del tercio apical.
Esternitos abdominales brillantes, con puntos pequeños y moderadamente densos, con sedas largas y poco densas hacia los lados. Pigidio moderadamente convexo en vista lateral, superficie fina y densamente estrigulada, con sedas pálidas, cortas, erectas y densas.
Genital masculino con parámeros subparalelos en la base, que se ensancha gradualmente hacia el ápice, siendo más ancho en la porción subapical (vista frontal) (fig. 1E); curvados uniformemente, con el ápice doblado ligeramente hacia la porción ventral (vista lateral) (fig. 1F).
Hembra. Desconocida.
Resumen taxonómico
Material tipo. Holotipo ♂, primera etiqueta poco legible: “Est???lla [Estrella] / Sonora, Mex. / X-2-33 [1933] // Coll. Jentry [Gentry] // Van Dyke coll” // “Neoscelis / septentrionalis Delgado et al. 2025 / Holotipo ♂”. El ejemplar se encuentra depositado en la CASC.
Etimología. El nombre de esta especie refiere al Septentrión, el hemisferio norte, ya que esta especie es la que se distribuye más al norte de las especies conocidas de Neoscelis.
Distribución. Tomando en cuenta los datos legibles de la etiqueta, a pesar de su mal estado de conservación, logramos deducir que se hace referencia a la localidad “La Estrella” o “Cañón Estrella”, asociando la fecha de colecta y colector que en ella aparecen. Howard S. Gentry, fue un botánico californiano y colector del holotipo; realizó sus primeras colectas botánicas para su obra “Rio Mayo Plants” (Gentry, 1942) en otoño de 1933. Este trabajo se realizó en el valle del Río Mayo, particularmente entre el río Cedros y el arroyo Guajaray. La localidad La Estrella fue una de las casi 120 localidades enlistadas en su trabajo. Por su parte, Edwin Van Dyke, profesor de entomología de la Universidad de Berkeley, y parte del staff curatorial de la California Academy of Science, donó su colección particular a esta última (Evans y Hogue, 2004).
Esta especie solo se conoce de la región del valle del Río Mayo de Sonora, en el distrito Sinaloense de la provincia de las Tierras Bajas del Pacífico (Morrone, 2017, 2019). En esta región se presenta un mosaico paisajístico conformado por bosque espinoso dominado por Acacia cocliachantha Humb. y Bonpl. ex Willd. y Neltuma velutina (Wooton) Briton y Rose entre 0 y 450 m snm, bosque tropical caducifolio dominado por Lysiloma divaricatum (Jacq.) J.F. Macbr. y Bursera spp. y encinar, entre 300 y 900 m snm y de 900 a 1,100 m snm, respectivamente (Gentry, 1942; Martínez-Yrizar et al., 2010; Rzedowski, 1978). El Cañón Estrella se encuentra alrededor de 500 m de elevación, presenta una interfase entre el matorral y el bosque tropical caducifolio con sabinos (Taxodium mucronatum Ten.) y encinos-pastizal hacia las partes más altas (Gentry, 1942). El hallazgo de esta nueva especie extiende el rango geográfico de esta tribu alrededor del Trópico de Cáncer, en Sonora, México.
Comentarios taxonómicos
Neoscelis septentrionalis sp. nov presenta la mayor similitud con N. longiclava, especialmente por la longitud de la maza antenal (mayor a los artejos precedentes juntos) y la simplicidad de los ornamentos clipeales, así como el pronoto ligeramente convexo (fig. 1A, G). Ambas especies pueden diferenciarse fácilmente por la abundante vestidura de los protarsómeros en la cara ventral de N. septentrionalis sp. nov. (fig. 1D) —muy pocas sedas dispersas en N. longiclava (fig. 1J). Los cuernos clipeales distales y laterales son desarrollados en N. septentrionalis sp. nov (fig. 1A, B), discretos en N. longiclava (fig. 1G, H). La forma del proceso mesometasternal es de alrededor de 80° en vista lateral (45° en N. longiclava) y presenta coloración verde metálica (negra a verde oscuro brillante en N. longiclava [fig. 1G]). La forma de la maza antenal es más ancha en vista lateral (fig. 1C) y la forma de los parámeros ligeramente más angosta en el ápice (fig. 1E) respecto de N. longiclava (fig. 1I, K). Las hembras de ambas especies son desconocidas.

Respecto a las otras especies (N. coracina, N. dohrni y N. hexakrotes), se diferencian fácilmente porque éstas presentan maza antenal pequeña (menor a la longitud combinada de los artejos precedentes), ornamentos clipeales muy desarrollados y elevados, y pronoto fuertemente convexo.
El ejemplar citado por Morón y Ratcliffe (1989) como dudoso (“?”) y que señalan en su mapa de “Sonora: Estuilla” (localidad mal escrita), y reproducido por García-Morales et al. (2019) debe tratarse con toda seguridad de N. septentrionalis.
Neoscelis longiclava Morón y Ratcliffe (fig. 1G-L)
Variación (3♂♂). Longitud total: 18 mm (holotipo) 19.8-22.6 mm, ancho: 9.5 mm (holotipo), 9.7-10.4 mm, 10.2-10.6 mm. Largo de la cabeza: 3.8-4.3 mm, largo del pronoto: 6.3- 6.9 mm; ancho de pronoto: 7.8-8.1 mm, largo élitros: 12.0-13.6 mm.
Coloración dorsal y ventral negra brillante, o verde esmeralda oscuro, metálico, brillante, con cabeza y élitros más oscurecidos (fig. 1G). Maza antenal aproximadamente un tercio más larga que resto de antenómeros, más de 3 veces más larga que ancha en vista dorsal (1:0.29) y 2.5 veces más larga que ancha en vista lateral (fig. 1I). Cuernos clipeales anchos, con ápices redondeados con ancho variable, separación en ángulo obtuso, longitud de los cuernos igual o menor a la longitud de la base (más largos que la base y con ángulo de separación agudo). Base de los cuernos variable pudiendo ser rectangular (más ancha que larga) o subcuadrada (tan ancha como larga). Protarsómeros (incluyendo las uñas) tan largos como la protibia, con sedas pequeñas, poco visibles. Élitros con puntuación fina y densa, ápice de las costillas elitrales fuertemente marcados.
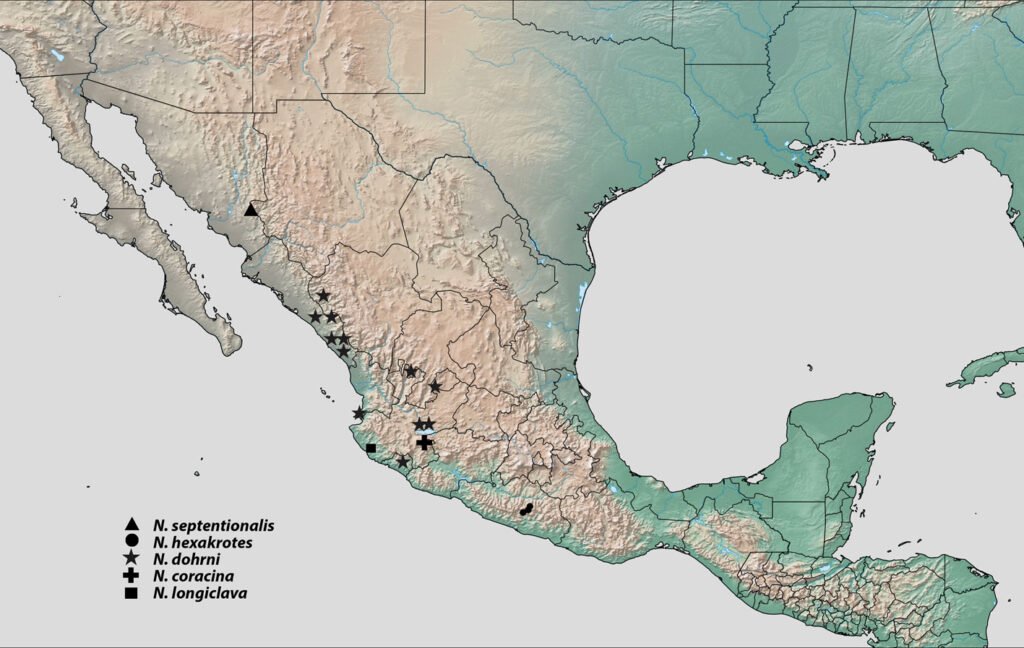
Clave para identificar los machos de las especies de Neoscelis (modificada de García-Morales et al., 2019)
1. Antenas con la maza antenal más larga que los antenómeros precedentes; ornamentos clipeales poco desarrollados; pronoto ligeramente convexo
……………………………………………………………………………… 2
1’. Antenas con la maza antenal más corta que los antenómeros precedentes; ornamentos clipeales notablemente desarrollados; pronoto fuertemente convexo
……………………………………………………………………………… 3
2. Superficie ventral de los protarsómeros con escasas sedas; maza antenal ancha, 2.5 veces más larga que ancha en vista lateral; coloración negra brillante o con ligera tonalidad esmeralda oscuro; oeste de Jalisco
……………………………………………………………………………… N. longiclava Morón y Ratcliffe
2’. Superficie ventral de los protarsómeros con sedas cortas y muy densas; maza antenal estrecha, 3 veces más larga que ancha en vista lateral; coloración verde obscura con tonalidades verde metálicas; Sonora
……………………………………………………………………………… N. septentrionalis sp. nov.
3. Frente convexa; metasterno y abdomen con abundantes sedas hacia la parte externa; Guerrero
……………………………………………………………………………… N. hexakrotes García-Morales, Ramírez-Ponce y Curoe
3’. Frente cóncava; metasterno y abdomen con escasas sedas hacia la parte externa; otros estados pero no en Guerrero
……………………………………………………………………………… 4
4. Carina preocular inclinada anteriormente, sin extenderse hacia la región central; cuerno cefálico profundamente bifurcado, ramas alargadas con tubérculo grande en cara posterior; color dorsal verde metálico, en ocasiones con reflejos cobrizos; de Colima a Durango
……………………………………………………………………………… N. dohrni (Westwood)
4’. Carina preocular extendiéndose hacia la región antero-central; cuerno cefálico moderadamente bifurcado, ramas subtriangulares sin tubérculo en cara posterior; color dorsal negro brillante, a veces con reflejos azul-verde; sur de Jalisco
……………………………………………………………………………… N. coracina Mudge y Ratcliffe
Nuevo registro de distribución de Neoscelis dohrni (Westwood). Se determinaron un macho y una hembra de N. dohrni etiquetados: “Tayahua, Zacatecas, México, 10-XI-1983, 22.0942° N, -102.8695° O, 1,700 m, S. Rosas col.”, ambos depositados en IEXA, que representan el primer registro de la especie para este estado.
Agradecimientos
A la Colección Entomológica de la California Academy of Sciences por el préstamo de ejemplares del género Neoscelis. Expresamos un reconocimiento especial al Dr. Leonardo Delgado (primer autor), quien lamentablemente falleció antes de la publicación de este trabajo, por sus valiosas contribuciones al estudio de los coleópteros de Latinoamérica.
Referencias
Bouchard, D. y Curoe, D. J. (2012). A new species of Ischnoscelis Burmeister, 1842 (Coleoptera, Cetoniinae, Goliatini). Coléoptères, 18, 1–8.
Curoe, D. J. (2013). Description of a third species of Ischnoscelis Burmeister (Coleoptera: Cetoniinae: Goliathini). Besouro, 23, 3–5.
Evans, A. V. y Hogue, J. N. (2004). Introduction to California beetles. Los Angeles, California: University of California Press.
García-Morales, L. J., Ramírez-Ponce, A., Curoe, D. J. y García-Jiménez, J. (2019). A new species of Neoscelis Schoch from Mexico (Coleoptera: Scarabaeidae: Cetoniinae: Goliathini) with new distributional records for the genus. Zootaxa, 4695, 122–130. https://doi.org/10.11646/zootaxa.4695.2.2
Gentry, H. S. (1942). Rio Mayo plants. A study of the flora and vegetation of the valley of the Rio Mayo. Washington, D.C.: Carnegie Institution of Washington. Publication 527.
Hernández-López, N., Delgado, L. y Burgos-Solorio, A. (2016). Description of the female of Ischnoscelis hoepfneri (Gory and Percheron) (Coleoptera: Scarabaeidae: Cetoniinae), notes on the species’ behavior, and a key the species of the genus. The Coleopterists Bulletin, 70, 387–390. https://doi.org/10.1649/0010-065X-70.2.387
Krajcik, M. (1998). Cetoniidae of the World. Catalogue-Part I. República Checa: Typos Studio Most.
Krajcik, M. (2012). Checklist of the world Scarabaeoidea. Anima.X. Supplement 5. República Checa: Plzen.
Martínez-Yrízar, A., Felger, R. S. y Búrquez, A. (2010). Los ecosistemas terrestres: un diverso capital natural. En F. E. Molina-Freaner y T. R. Van Devender (Eds.), Diversidad biológica de Sonora (pp. 129–156). México D.F.: UNAM-Conabio.
Morón, M. A. y Ratcliffe, B. C. (1989). A synopsis of the American Goliatini with description of a new Neoscelis from Mexico. The Coleopterists Bulletin, 43, 339–348.
Morrone, J. J. (2017). Neotropical biogeography: regionalization and evolution. Boca Raton, Florida: CRC Press. https://doi.org/10.1201/b21824
Morrone, J. J. (2019). Regionalización biogeográfica y evolución biótica de México: encrucijada de la biodiversidad del Nuevo Mundo. Revista Mexicana de Biodiversidad, 90, 1–68. https://doi.org/10.22201/ib.20078706e.2019.90.2980
Mudge, A. D., Ratcliffe, B. C., Westcott, R. L. y Noguera, F. A. (2003). A new species of Neoscelis from Jalisco, Mexico (Coleoptera: Scarabaeidae: Cetoniinae). Folia Heyrovskyana, 11, 143–154.
Rzedowski, J. (1978). Vegetación de México. México D. F.: Limusa.
Schoolmeister, P. (2025). World Scarabaeidae database (Version 2025-09-08). En O. Bánki, et al. (Eds.), Catalogue of life. Amsterdam, Países Bajos: Catalogue of Life Foundation. https://doi.org/10.48580/dgt98-38g
Shorthouse, D. P. (2010). SimpleMappr, an online tool to produce publication-quality point maps. Recuperado el 22 de mayo 2025, de: www.simplemappr.net
Tropicos (2025). Tropicos.org. Missouri Botanical Garden. Recuperado el 17 Ene 2025 de https://tropicos.org
Westwood, J. O. (1855). De Coleopteris Goliathidis novi mundo. Linnaea Entomologica, 10, 326–328.
Wheeler Q. D. y Platnick, N. I. (2000). The phylogenetic species concept (sensu Wheeler and Platnick). En Q. D. Wheeler y R. Meier (Eds.), Species concepts and phylogenetic theory: a debate (pp. 55–69). New York: Columbia University Press.
Macroalgae assemblages in different reef zones of a tropical urban beach in Brazil
Ensambles de macroalgas en diferentes zonas arrecifales de una playa urbana tropical en Brasil
Caio Ceza da Silva-Nunes a, c, d, e, *, Edilene Maria dos Santos Pestana b, c, Cibele Conceição dos Santos b, c, Lorena Pedreira Conceição a, c, José Marcos de Castro Nunes a, c
a Universidade Estadual de Feira de Santana, Departamento de Ciências Biológicas, Programa de Pós-Graduação em Botânica, Av. Transnordestina s/n, Novo Horizonte, CEP 44036-900, Feira de Santana, Bahia, Brazil
b Universidade Federal da Bahia, Instituto de Biologia, Programa de Pós-Graduação em Biodiversidade e Evolução, Rua Barão de Jeremoabo, 668 – Campus de Ondina, CEP: 40170-115, Salvador, Bahia, Brazil
c Universidade Federal da Bahia, Instituto de Biologia, Laboratório de Algas marinhas, Rua Barão de Jeremoabo, 668 – Campus de Ondina, CEP: 40170-115, Salvador, Bahia, Brazil
d Universidade Estadual do Sudoeste da Bahia, Departamento de Ciências Exatas e Naturais, Programa de Pós-Graduação em Ciências Ambientais, Campus Universitário “Juvino Oliveira”, BR 415, Km 04 CEP: 45.700-000 Itapetinga, Bahia, Brazil
e Colégio Estadual de Tempo Integral Antônio Batista, Rua Presidente Vargas, 83 CEP: 46380-076 Candiba, Bahia, Brazil
*Corresponding author: caiobio08@gmail.com (C.C. da Silva-Nunes)
Received: 26 February 2025; accepted: 01 August 2025
Abstract
Marine macroalgae are commonly used as bioindicators because they are sensitive to environmental changes. This study aims to verify the composition of macroalgae in the intertidal region in 3 reef zones on Itapuã beach, located in Salvador-Bahia, Brazil and which presents high tourist activity. The samples were obtained in July 2017, in the intertidal zone, in 3 reef regions: Protected Region (PR), Tide Pool Region (TP) and Frontal Region (FR). In each zone, 3 transects (20 m each) were placed, in which 5 squares measuring 20 x 20 cm were arranged at random points. Additionally, individuals were collected around each transect for qualitative analysis. Dry biomass was measured, and statistical tests were carried out to obtain diversity, equitability and similarity data. Fifty-two taxa were identified, 27 Rhodophyta, 16 Chlorophyta and 9 Phaeophyceae. TP had the highest species richness (34) and diversity; however, no significant differences were found in macroalgal biomass between the 3 reef zones analyzed. This study contributes to the understanding of the composition and structure of phytobenthic communities in intertidal regions of the Bahian coast.
Keywords: Bahia; Bioindicator; Brazil; Phytobenthos
Resumen
Las macroalgas marinas se utilizan comúnmente como bioindicadores porque son sensibles a los cambios ambientales. Este estudio tiene como objetivo verificar la composición de macroalgas en la zona intermareal en 3 regiones arrecifales de la playa de Itapuã, ubicada en Salvador-Bahía, Brasil y que tiene alta actividad turística. Las muestras fueron obtenidas en julio de 2017, en la zona intermareal, en 3 regiones arrecifales: región posterior (PR), pozas de marea (TP) y región frontal (FR). En cada región, se colocaron 3 transectos (de 20 m cada uno), donde se distribuyeron aleatoriamente 5 cuadrantes de 20 x 20cm. Además, se recolectaron individuos alrededor de cada transecto para análisis cualitativo. Se midió la biomasa seca y se realizaron pruebas estadísticas para obtener datos de diversidad, equitabilidad y similitud. Se identificaron 52 taxones: 27 Rhodophyta, 16 Chlorophyta y 9 Phaeophyceae. La TP presentó la mayor riqueza de especies (34) y diversidad; sin embargo, no se encontraron diferencias significativas en la biomasa de macroalgas entre las 3 regiones arrecifales. Este estudio contribuye a la comprensión de la composición y estructura de las comunidades fitobentónicas en zonas intermareales del litoral bahiano.
Palabras clave: Bahia; Bioindicador; Brasil; Fitobentos
Introduction
The State of Bahia has the longest coastline in Brazil, 1,103 km showing a great diversity of environments: sandy beaches, coral reefs, sandstone formations, rocky shores and mangroves (Nunes & Paula, 2004a). Furthermore, it is a region that presents a great diversity of substrates and geographical features, which is reflected in the diversity of marine flora (Nunes, 2005b). Nunes and Paula (2002) divided reef formations into 3 zones: Frontal Region (FR), Protected Region (PR) and Tide Pool Region (TP). The FR is a very hydrodynamic region, where direct collision of waves occurs. The PR is the region before the lagoon and after the reef top, it may have pools and is a region protected from direct wave collision. The TP is deep and may suffer greater wave action, forming pools on the reef top, or may suffer less or no wave action, forming pools on the reef plateau.
In recent decades, coastal areas have been undergoing an intense process of urban development, which has caused significant environmental pressures and impacts, especially in benthic communities in reef formations (Costa et al., 2012; Nascimento, 2013). In Salvador, these impacts come from urban expansion, real estate speculation, tourism, human activities, such as fishing and trampling, causing the degradation of the flora and a reduction in the richness and diversity of species, especially in the composition and structure of algal communities.
Marine macroalgal communities have a great ecological role, being, together with microalgae, at the base of the food chain as primary producers, being a source of food for a large part of the marine fauna. The presence of macroalgae along the coast is responsible for softening the impact of waves on the sea coast, and also plays the role of habitat for other organisms, such as animals or as a substrate for algae (Nunes, 2010; Pedrine, 2013).
When these organisms are exposed to some human interference, they are sensitive to changes in their habitat, such as the increase in the concentration of organic matter in the water, which leads to an increase in the biomass of some algal groups, or even the depletion of some nutrient that causes the disappearance of a certain species, working as bioindicators of environmental quality. In addition, the degradation of this community’s structure favors the emergence and persistence of more resistant and opportunistic species, as well as the exclusion of more fragile species (Nascimento, 2013; Nunes, 2010).
Functional groups of algae are based on similarities in their morphological and anatomical characteristics, in addition to their ecological characteristics. Steneck and Dethier (1994) grouped algae into 7 categories, these being microalgae, filamentous, foliaceous, cylindrical-corticate, coriaceous, articulated calcareous and encrusting. In the model proposed by Steneck and Dethier (1994), it is highlighted that environments of high productivity and low disturbance present high biomass and diversity of morphofunctional groups, providing an abundance of coriaceous and cylindrical-cortical algae, as they have a relatively large size and a longer life cycle.
Knowledge about macroalgae on the coast of Bahia has been expanded through taxonomic studies, with the metropolitan area of Salvador being one of the main regions of the State’s coast studied, for example: Altamirano and Nunes (1997), Amorin et al. (2006), Barreto et al. (2004), Macedo et al. (2009), Marins et al. (2008), Nunes (1998a, b, 1999, 2005a, b), Nunes and Guimarães (2008, 2009, 2010), Nunes and Paula (2000, 2001, 2002, 2004a, b, 2006), Nunes et al. (2005).
Studies regarding the community structure of marine macroalgae have been carried out on the Brazilian coast. The majority are concentrated in the subtidal zone, namely, Amado Filho et al. (2003), Figueiredo et al. (2004), Villaça et al. (2010) in Rio de Janeiro, Oliveira-Carvalho et al. (2003) in Pernambuco, and Horta et al. (2008) in Santa Catarina. And in the intertidal zone, Barbosa et al. (2008) in Espírito Santo, and Muñoz and Pereira (1997) in Pernambuco. In Bahia, there are studies by Caires et al. (2013), Costa et al. (2012), and Ferreira et al. (2022), who carried out studies in the intertidal zone, and Costa Jr. et al. (2002) and Marins et al. (2008) in the subtidal zone. Although the coast of Bahia is the longest in Brazil, studies aimed to understand the structure of phytobenthic communities are scarce, creating a gap in the knowledge of these communities.
The present study analyzes the intertidal phytobenthic community in 3 reef zones of Itapuã beach, aiming to identify and compare differences in the composition and structure of the species between the studied areas, using the Feldmann and Cheney indices as bioindicators of local environmental characteristics.
Materials and methods
Itapuã beach is located in Salvador, Bahia in Brazil (Fig. 1), and corresponds to 2 large rocky bodies, and has coarse sand beaches, many reefs and rocky bollards (Nunes, 1998b). The collection was carried out on July 11, 2017, in the intertidal region, during low spring tide. The intertidal region was compartmentalized into the 3 regions proposed by Nunes and Paula (2002). Nunes (2010) was followed for the collection protocol.
Three 20 m transects were placed in each zone and arranged parallel to the coastline. In each transect, 5 squares measuring 20 x 20 cm were previously drawn. All material contained in the square was removed using a spatula. For qualitative analysis, specimens were collected around each transect. The collected material was placed in plastic bags or polyethylene bottles, fixed with 4% formalin. All material from the collections was taken to the Marine Algae Laboratory (LAMAR), at the Biology Institute of the Federal University of Bahia, where it was analyzed using a stereomicroscope (Leica© – Zoom 2000) to separate and identify the epiphytic algae.
Taxon identification was done using an Olympus© – CX 22 microscope. Blades were assembled from freehand cuts, made with the aid of razor blades and box cutters. Calcareous algae were analyzed based on prior decalcification by immersing the samples in 0.6 M nitric acid. For specific identification, references commonly used in phycology were used (Dawes & Mathieson, 2008; Littler & Littler, 2000; Nunes, 2005b; Nunes & Guimarães, 2008; Nunes & Paula, 2000, 2001, 2002). For the arrangement of taxa, the taxonomic arrangement of Guiry and Guiry (2025) was mainly followed.
For the analysis of dry biomass, the infrageneric taxa were previously identified, separated and dried in an oven at 60 ºC for 48 hours and weighed until constant biomass was obtained. The results are presented in grams/m² of dry weight. The results for each reef zone were compared regarding the total number of taxa, and dry biomass using Shannon-Wiener diversity (H’) and Pielou equitability (J) indexes.
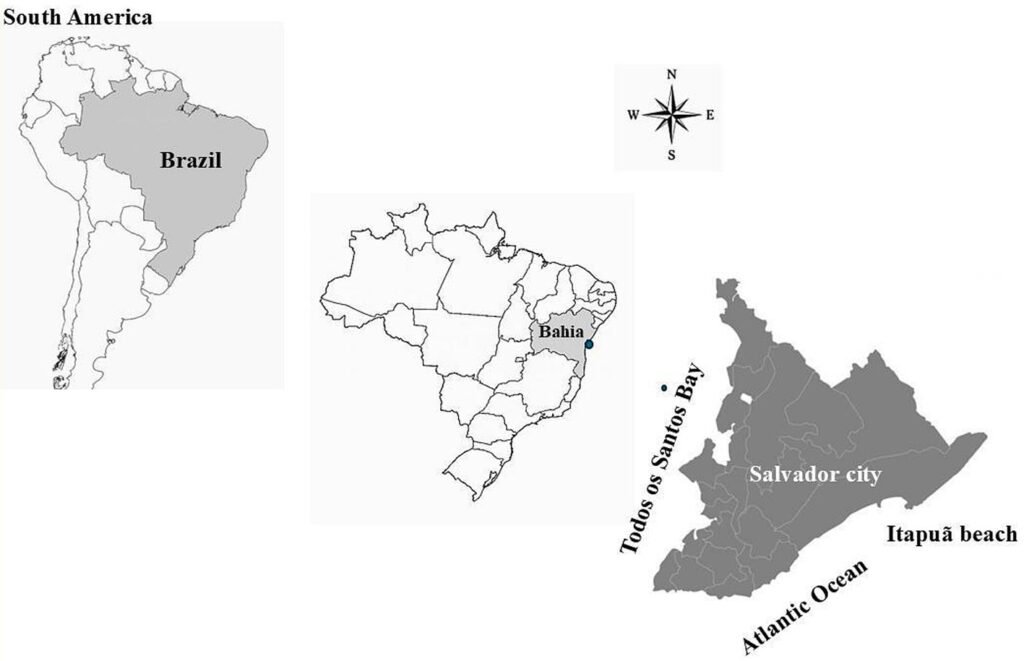
Using the macroalgae biomass data found in each reef zone, the Kruskal-Wallis test was performed to check whether there was a significant difference between the biomass recorded in the different zones. The Bray-Curtis similarity index was also calculated with the biomass data and Non-metric Multidimensional Scaling (nMDS) was performed using the transformed data. ANOSIM was performed for values in which there was a significant difference between the similarity results. The analyses were carried out using SigmaPlot 12 and Primer V6 software.
The species dominance index is used to assess how much one or a few species stand out in relation to the others within a biological community. It considers the proportion of individuals or the biomass of each species, allowing us to identify which of them exert the greatest influence on the structure and functioning of the ecosystem. The higher the value of the index, the greater the dominance, which indicates that a few species concentrate the majority of the individuals present. On the other hand, lower values reflect a more balanced and diverse community, with a lower concentration of individuals in a few species (Melo, 2008).
Simpson’s dominance index was used, calculated as D = ∑ (ni/N) 2, where ni is the number of individuals of a species and N is the total number of individuals of all species in the community. The value of D varies between 0 and 1, with values close to 1 indicating high dominance, that is, few species dominate the environment, while values close to 0 indicate greater diversity and balanced distribution among species.
The Feldmann index (obtained by dividing the number of species of Rhodophyta by that of Phaeophyceae [R/P]) and the Cheney index (adding the number of species of Rhodophyta to that of Chlorophyta, and dividing this value by the number of browns [R + C/ P]) (Figueiredo et al., 2008).
Results
Fifty-two taxa were identified, 27 (52) belonging to the Rhodophyta, 16 (31) Chlorophyta, and 9 (17) Phaeophyceae (Heterokontophyta). Table 1 presents the taxa, their distribution throughout the 3 zones of the reef and their respective morphotypes.
Among the zones, TP presented the greatest species richness (34) with Rhodophyta as the most representative group, 17 taxa, followed by Chlorophyta (11) and Phaeophyceae (6). Chaetomorpha minima, Gayliella dawsonii, Gracilaria ferox, Lejolisia mediterranea, Ptilothamnion speluncarum, and Stylonema alsidii were unique to this environment. PR was second in terms of species richness, such as Caulerpa chemnitzia, Caulerpa sertularioides, Cladophora corallinicola, Hypnea pseudomusciformis, Hypnea sp. 1, and Melanothamnus gorgoniae only occurred in this region. FR presented the lowest richness among the environments, such as Ceramium corniculatum, Codium taylori, Dictyopteris jamaicensis, Dictyopteris polypodioides, Sphacelaria tribuloides, and Wrangelia argus, all exclusive for the region. However, it is worth highlighting a greater representation of rhodophytes for the protected and FR.
Among the species found, 16 occurred in all regions such as Amansia multifida, Amphiroa anastomosans, Anadyomene stellata, Bryopsis pennata, Colpomenia sinuosa, Crouania attenuata, Dictyopteris delicatula, Dictyosphaeria versluysii, Dictyota mertensii, Gelidiella acerosa, Halimeda opuntia, Jania pedunculata var. adhaerens, Palisada perforata, Phyllodictyon anastomosans, Ulva flexuosa, and Ulva rigida.
Regarding the morphotypes, among the 52 taxa found, 19 were filamentous, 17 cylindrical-corticates, 9 foliaceous, 3 articulated calcareous, 3 encrusting and 1 coriaceous. Cylindrical-corticate algae were well represented in the 3 regions, predominating in the PR, followed by foliaceous algae; in the TP, the filamentous morphotype was the one with the highest representation, followed by cylindrical-corticated and foliaceous ones, the same pattern was observed in the FR.
The diversity of analysis based on the Shannon-Wiener index, showed that the TP is more diverse than the others, followed by the PR, and finally by the FR. Dominance values are inversely proportional to diversity values, which shows that in the FR, there is dominance of one or a few species over the others (Table 2).
In table 3, biomass data and the percentage of contribution of each macroalgae in the zone biomass are presented. The average total biomass of each zone was 108.01 g.m-2 in the FR, 48.83 g.m-2 in tide pool and 474.59 g.m-2 in the PR. The Kruskal-Wallis test showed that no significant differences were found in macroalgal biomass between the three reef zones analyzed (Kruskal-Wallis p = 0.869). However, the ordering of the sampling points according to the presence/absence of macroalgae (Fig. 2) shows the dispersion of samples in the 3 zones, mainly samples from the PR. The ANOSIM analysis (Table 4) shows that there was a significant difference between the FR and the TP, and between the FR and the PR, but there was no significant difference between the PR and the TP. The Feldmann and Cheney indices (Fig. 3) for the 3 reef regions studied were consistent with the flora of tropical and warm temperate regions.
Table 1
List of taxa found in the reef zones on Itapuã beach and their respective morphotypes: PR = Protected Region; TP = Tide Pool; FR = Frontal Region. Morphotypes: FT = filamentous; FC = foliaceous; CC = cylindrical corticates; CR = coriaceous; AC = articulated calcareous; E = encrusting. Presence (+) and absence (–).
| Taxa | PR | TP | FR | Morphotype |
| Chlorophyta | ||||
| Ulvophyceae | ||||
| Bryopsidales | ||||
| Bryopsidaceae | ||||
| Bryopsis pennata J.V. Lamouroux | + | + | + | FT |
| Caulerpaceae | ||||
| Caulerpa chemnitzia (Esper) J.V. Lamouroux | + | – | – | CC |
| C. racemosa (Forsskål) J. Agardh | + | + | + | CC |
| C. sertularioides (S.G. Gmelin) M. Howe | + | – | – | CC |
| Codiaceae | ||||
| Codium taylorii P.C. Silva | – | – | + | CC |
| Halimedaceae | ||||
| Halimeda opuntia (Linnaeus) J.V. Lamouroux | + | + | + | AC |
| Ulva flexuosa Wulfen | + | + | + | FC |
| U. rigida C. Agardh | + | + | + | FC |
| Cladophorales | ||||
| Anadyomenaceae | ||||
| Anadyomene stellata (Wulfen) C. Agardh | + | + | + | FC |
| Boodleaceae | ||||
| Phyllodictyon anastomosans (Harvey) Kraft & M.J. Wynne | + | + | + | FT |
| Cladophoraceae | ||||
| Chaetomorpha minima Collins & Hervey | – | + | – | FT |
| Cladophora corallinicola Sonder | + | – | – | FT |
| C. laetevirens (Dillwyn) Kützing | – | + | + | FT |
| C. vagabunda (Linnaeus) Hoek | + | – | + | FT |
| Siphonocladaceae | ||||
| Dictyosphaeria versluysii Weber Bosse | + | + | + | E |
| Valoniaceae | ||||
| Valonia aegagropila C.Agardh | + | + | – | CC |
| Heterokontophyta | ||||
| Phaeophyceae | ||||
| Dictyotales | ||||
| Dictyotaceae | ||||
| Dictyopteris delicatula J.V. Lamouroux | + | + | + | FC |
| D. jamaicensis W.R. Taylor | – | – | + | FC |
| D. polypodioides (De Candolle) J.V. Lamouroux | – | – | + | FC |
| Dictyota mertensii (C.Martius) Kützing | + | + | + | FC |
| Table 1. Continued | ||||
| Taxa | PR | TP | FR | Morphotype |
| Padina antillarum (Kützing) Piccone | + | + | – | FC |
| Spatoglossum schroederi (C. Agardh) Kützing | – | + | + | FC |
| Ectocarpales | ||||
| Scytosiphonaceae | ||||
| Colpomenia sinuosa (Mertens ex Roth) Derbès & Solier | + | + | + | E |
| Fucales | ||||
| Sargassaceae | ||||
| Sargassum polyceratium Montagne | – | + | + | CR |
| Sphacelariales | ||||
| Sphacelariaceae | ||||
| Sphacelaria tribuloides Meneghini | – | – | + | FT |
| Rhodophyta | ||||
| Florideophyceae | ||||
| Ceramiales | ||||
| Callithamniaceae | ||||
| Crouania attenuata (C. Agardh) J. Agardh | + | + | + | FT |
| Ceramiaceae | ||||
| Ceramium corniculatum Montagne | – | – | + | FT |
| Centroceras clavulatum (C. Agardh) Montagne | + | – | – | FT |
| Gayliella dawsonii (A.B. Joly) Barros-Barreto & F.P. Gomes | – | + | – | FT |
| Ceramothamnion brasiliensis (A.B. Joly) M.J. Wynne & C.W. Schneider | + | – | + | FT |
| Rhodomelaceae | ||||
| Alsidium triquetrum (S.G. Gmelin) Trevisan | – | + | – | CC |
| Amansia multifida J.V. Lamouroux | + | + | + | CC |
| Digenea simplex (Wulfen) C. Agardh | + | + | – | CC |
| Herposiphonia bipinnata M. Howe | – | + | – | FT |
| H. tenella (C. Agardh) Ambronn | – | + | – | FT |
| Melanothamnus gorgoniae (Harvey) Díaz-Tapia & Maggs | + | – | – | FT |
| Palisada perforata (Bory) K.W. Nam | + | + | + | CC |
| Wrangeliaceae | ||||
| Lejolisia mediterranea Bornet | – | + | – | FT |
| Ptilothamnion speluncarum (Collins & Hervey) D.L. Ballantine & M.J. Wynne | – | + | – | FT |
| Wrangelia argus (Montagne) Montagne | – | – | + | FT |
| Corallinales | ||||
| Corallinaceae incrustante | ||||
| Jania pedunculata var. adhaerens (J.V. Lamouroux) A.S. Harvey, Woelkerling & Reviers | + | + | + | AC |
| Corallinaceae incrustante | + | – | + | E |
| Lithophyllaceae | ||||
| Table 1. Continued | ||||
| Taxa | PR | TP | FR | Morphotype |
| Amphiroa anastomosans Weber-van Bosse | + | + | + | AC |
| Gelidiales | ||||
| Gelidiaceae | ||||
| Gelidium capense (S.G. Gmelin) P.C. Silva | + | – | + | CC |
| Gelidiellaceae | ||||
| Gelidiella acerosa (Forsskål) Feldmann & Hamel | + | + | + | CC |
| G. ligulata E.Y. Dawson | + | – | + | CC |
| Gigartinales | ||||
| Cystocloniaceae | ||||
| Hypnea pseudomusciformis Nauer, Cassano & M.C. Oliveira | + | – | – | CC |
| Hypnea sp.1 | + | – | – | CC |
| Gigartinaceae | ||||
| Chondracanthus acicularis (Roth) Fredericq | + | + | – | CC |
| Gracilariales | ||||
| Gracilariaceae | ||||
| Gracilaria ferox J. Agardh | – | + | – | CC |
| Nemaliales | ||||
| Galaxauraceae | ||||
| Galaxaura rugosa (J. Ellis & Solander) J.V. Lamouroux | – | + | + | CC |
| Stylonematophyceae | ||||
| Stylonematales | ||||
| Stylonemataceae | ||||
| Stylonema alsidii (Zanardini) K.M. Drew | – | + | – | FT |
| Total | 33 | 34 | 32 |
Discussion
Rhodophyta was the most representative group in the present study, followed by Chlorophyta and Phaeophyceae. This pattern has been observed in other studies on phytobenthic macroalgal communities as typical on the Brazilian coast (Braga et al., 2014; Costa et al., 2012; Ferreira et al., 2022). It should be noted that rhodophytes tend to dominate the environment in the absence of large brown algae in tropical environments, suggesting that there is competition between algae that use different strata in the phytobenthic community (Figueiredo et al., 2004). Furthermore, studies have confirmed the decline of Phaeophyceae at the same rate that Chlorophyta species have been increasing, which can be attributed to the negative effects of anthropogenic impact on the coastal environment (Oliveira & Qi, 2003; Scherner et al., 2013). Representatives of Phaeophyceae are more sensitive to pollutants (heavy metals and excess organic matter produced by sewage), which can even negatively affect the germination and cell division of these organisms (Kevekordes, 2001). Representatives of Chlorophyta, such as species of Ulva Linnaeus, have the capacity of benefit from contamination by pollutants, being considered opportunistic species (Scherner et al., 2012).
Table 2
Shannon-Wiener Diversity index, dominance and equitability of macroalgae in the intertidal region of Itapuã beach.
| Region | Shannon-Wiener | Dominance | Equability (J’) |
| Frontal Region | 3.466 | 0.0312 | 1 |
| Tide Pool | 3.526 | 0.0294 | 1 |
| Protected Region | 3.497 | 0.0303 | 1 |
The taxa that contributed most to the average biomass of macroalgae found on Itapuã beach were Amphiroa anastomosans, Digenea simplex, Gelidiella acerosa, and Sargassum polyceratium. It has been previously reported that algae with greater structural complexity, such as corticate algae, are better adapted to environments with greater hydrodynamics and luminosity, as this morphotype confers greater resistance to desiccation (Costa et al., 2012; Villaça et al., 2008).
Table 3
Biomass and percentage of contribution of macroalgae species in the constitution of biomass in each of the zones studied: FR = Frontal Region, TP = Tide Pool, and PR = Protected Region. Pi = Percentage of importance.
| Taxa | FR (g.m-2) | Pi | TP (g.m-2) | Pi (%) | PR (g.m-2) | Pi (%) |
| Amphyroa anastomosans | 70.07 | 64.87 | 15.76 | 32.28 | 31.59 | 6.66 |
| Anadyomene stellata | 1.41 | 1.31 | 1.76 | 3.61 | 0.00 | 0.00 |
| Bryopsis pennata | 0.00 | 0.00 | 0.20 | 0.42 | 0.00 | 0.00 |
| Caulerpa racemosa | 1.42 | 1.32 | 0.00 | 0.00 | 0.00 | 0.00 |
| C. sertularioides | 0.00 | 0.00 | 0.00 | 0.00 | 1.11 | 0.23 |
| Centroceras clavulatum | 0.00 | 0.00 | 0.00 | 0.00 | 0.25 | 0.05 |
| Chondracanthus acicularis | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 |
| Cladophora vagabunda | 0.00 | 0.00 | 0.00 | 0.00 | 0.15 | 0.03 |
| Colpomenia sinuosa | 0.16 | 0.15 | 0.08 | 0.15 | 0.00 | 0.00 |
| Dictyopteris delicatula | 5.42 | 5.01 | 5.48 | 11.22 | 0.00 | 0.00 |
| D. jamaicensis | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 |
| Dictyosphaeria versluysii | 0.67 | 0.62 | 0.25 | 0.51 | 0.09 | 0.02 |
| Dictyota mertensii | 0.00 | 0.00 | 0.00 | 0.00 | 1.13 | 0.24 |
| Digenea simplex | 0.00 | 0.00 | 0.97 | 1.98 | 430.92 | 90.80 |
| Gelidiellaceae | 0.00 | 0.00 | 2.00 | 4.09 | 0.00 | 0.00 |
| Gelidium sp. 1 | 0.00 | 0.00 | 0.94 | 1.92 | 0.19 | 0.04 |
| Gelidium sp. 2 | 0.00 | 0.00 | 0.00 | 0.00 | 0.09 | 0.02 |
| Gelidiella acerosa | 14.27 | 13.21 | 5.97 | 12.24 | 2.69 | 0.57 |
| G. ligulata | 0.71 | 0.66 | 0.00 | 0.00 | 0.00 | 0.00 |
| Halimeda opuntia | 0.31 | 0.29 | 2.14 | 4.38 | 0.48 | 0.10 |
| Hypnea sp. 1 | 0.40 | 0.37 | 0.00 | 0.00 | 0.00 | 0.00 |
| Hypnea sp. 2 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 |
| Jania pedunculata var. adhaerens | 7.60 | 7.03 | 0.00 | 0.00 | 0.27 | 0.06 |
| Lobophora variegata | 0.00 | 0.00 | 0.39 | 0.80 | 0.00 | 0.00 |
| Padina antillarum | 0.00 | 0.00 | 2.26 | 4.64 | 1.15 | 0.24 |
| Palisada perforata | 4.86 | 4.50 | 0.00 | 0.00 | 0.05 | 0.01 |
| Phyllodictyon anastomosans | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 |
| Sargassum polyceratium | 0.00 | 0.00 | 9.69 | 19.85 | 0.00 | 0.00 |
| Spatoglossum schroederi | 0.10 | 0.09 | 0.00 | 0.00 | 0.00 | 0.00 |
| Ulva rigida | 0.63 | 0.58 | 0.83 | 1.69 | 3.55 | 0.75 |
| Valonia aegagropila | 0.00 | 0.00 | 0.11 | 0.22 | 0.86 | 0.18 |
| Total | 108.01 | 48.83 | 474.59 |
The difference found in macroalgae biomass between reef zones can be explained by the hydrodynamics to which they are exposed on the reef, being wave action the main responsible for the spatial distribution of the community on the reef. The movement of waves helps to obtain nutrients, favoring the productivity of macroalgae and thus increasing biomass, however this only applies to those algae that have adaptations to resist the impact of waves, as they are also responsible for removing macroalgae from the substrate (Diez et al., 2003; Hurd et al., 1996; Leigh et al., 1987).
The highest biomasses were found in FR and PR. The FR is constantly submerged and this favors the establishment of algae, as they are submerged for longer, suffer less desiccation and are less exposed to direct sunlight. The lowest biomass values were recorded in the TP, a region that retains water during low tide, however the algae are subjected to intense solar radiation, high salinity and water temperature. The negative effects of these stressors result in lower biomass and also there is less hard substrate for fixation (Costa et al., 2012; Villaça et al., 2010).
Table 4
Results of ANOSIM tests for significance of differences between sample groups based on observed macroalgal species distribution data.
| Reef regions | (Global R= 0.613; significance level = 0.4%) | |
| Region pairs | Statistical R | Level |
| FR, TP | 0.815 | 10 |
| FR, PR | 0.667 | 10 |
| TP, PR | 0.481 | 20 |


The dominance of filamentous, cylindrical-corticate and foliaceous morphotypes was observed, corroborating the model by Orfanidis et al. (2001), which proposes that impacted environments should have a greater abundance of algae with intensely branched (corticated), laminar (foliaceous) and filamentous thallus, characterized by high growth rates and a short life cycle (annual). However, conserved environments would have an abundance of algae with thick thallus (coriaceous), articulated calcareous and encrusting, cacterized by low growth rates and a long life cycle (perennial), which reflected in the low representation of articulated, encrusting and coriaceous calcareous morphotypes. In a study carried out by Nascimento (2013) in Salvador and the North Coast of Bahia, the foliaceous and cylindrical-corticate morphotypes were more representative in both impacted and preserved environments, highlighting the predominance of these morphotypes.
The distribution of algae according to morphofunctional groups reflects the conditions of reef zones, since each taxa has its adaptive characteristics. Cylindrical-cortical algae were well represented in the 3 reef zones, with a predominance in the PR, as this morphology confers greater resistance to desiccation, helping these algae to establish themselves in different environments (Costa et al., 2012; Villaça et al., 2008). Tide pool region was dominated by filamentous algae, morphotype usually associated with benthic communities in early successional stages as they show rapid growth (Braga et al., 2014).
Regarding phytogeography, the Feldmann (F) and Cheney (C) indices found for the different regions of the Itapuã reef characterized the PR and the TP as tropical and the FR as warm temperate, according to the classification proposed by Horta et al. (2001). Factors such as the greater hydrodynamism in the FR may justify this classification in relation to its algal community, since the increase in the number of species of brown algae is noticeable as one approaches the FR. Bouzon et al. (2006), studying floristic and phytogeographic aspects of marine macroalgae in the Bays of Santa Catarina, attributed the variations in the Feldmann and Cheney indices to the exclusion of species of brown algae and the favoring of opportunistic species of red and green algae; in this study, lower values of these indices were recorded in the frontal zones and in the pool zone, where the richness of species of brown algae was greater.
Understanding the composition and structure of phytobenthic communities on the Brazilian coast, as well as on the coast of Bahia is extremely important. Despite the vast coastal extension, there are few studies referring to the algal community structure in intertidal zone as most studies focus on the subtidal zone. Also, most studies on the composition and structure of phytobenthic communities are restricted to the state of Rio de Janeiro. More studies should be carried out along the coast of Bahia to fill the gaps and alternating dry and rainy periods and/or comparing the reef zones of different beaches in the intertidal zone. The results suggest that the Itapuã reefs are in a good state of conservation.
Acknowledgements
CCSN, acknowledges the post-doctoral scholarship to the State University of Southwest Bahia (UESB) – Notice 266/2023.; CCSN, LPC, EMSP, to the Coordination for the Improvement of Higher Education Personnel – Brazil (CAPES), finance code 001; CCS, to the Bahia Research Support Foundation (FAPESB – T.O. B., No. BOL0416/2017) for a scholarship.; JMCN, to the National Council for Scientific and Technological Development (CNPq), Brazil for the research productivity fellowship (308261/2022-4).
References
Altamirano, M., & Nunes, J. M. C. (1997). Contribuciones al macrofitobentos del Município de Camaçari (Bahia, Brasil). Acta Botanica Malacitana, 22, 211–215. https://doi.org/10.24310/abm.v22i0.8639
Amado Filho, G. M., Barreto, M. B. B., Marins, B. V., Felix, C., & Reis, R. P. (2003). Estrutura das comunidades fitobentônicas do infralitoral da Baía de Sepetiba, RJ, Brasil. Revista Brasileira de Botânica, 26, 329–342. https://doi.org/10.1590/s0100-84042003000300006
Amorin, P. R. R., Moura, C. W. N., & Moniz-Brito, K. L. (2008). Estudo morfotaxonômico das espécies de Halimeda, Penicillus e Udotea (Bryopsidales, Chlorophyta) do Recife de Franja da Ilha de Itaparica, Bahia. Anais do XI Congresso Brasileiro de Ficologia & Simpósio Latino-Americano sobre Algas Nocivas. Rio de Janeiro: Museu Nacional. Série Livros.
Barbosa, S. O., Figueiredo, M. A. O., & Testa, V. (2008). Structure and dynamic of benthic communities dominated by macrophytes in praia de Jacaraípe, Espírito Santo, Brazil. Hoehnea, 35, 563–575. https://doi.org/10.1590/S2236-89062008000400008
Barreto, M. B. B., Brasileiro, P. S., Nunes, J. M. C., & Amado-Filho, G. M. (2008). Algas marinhas bentônicas do sublitoral das formações recifais da Baía de Todos os Santos, BA – 1. Novas ocorrências. Hoehnea, 31, 321–330.
Braga, A. C. S., Tâmega, F. T. S., Pedrini, A. G., & Muniz, R. A. (2014). Composição e estrutura da comunidade fitobentônica do infralitoral da praia de Itaipu, Niterói, Brasil: Subsídios para monitoramento e conservação. Iheringia. Série Botânica, 69, 267–276. https://isb.emnuvens.com.br/iheringia/article/view/90
Bouzon, J. L., Salles, J. P., Bouzon, Z., & Horta, P. A. (2006). Aspectos florísticos e fitogeográficos das macroalgas marinhas das baías da ilha de Santa Catarina, SC,Brasil. Insular, 35, 69–84.
Caires, T. A., Costa, I. O., Jesus, P. B. D., Matos, M. R. B. D., Pereira-Filho, G. H., & Nunes, J. M. D. C. (2013). Evaluation of the stocks of Hypnea musciformis (Rhodophyta: Gigartinales) on two beaches in Bahia, Brazil. Brazilian Journal of Oceanography, 61, 65–71. https://doi.org/10.1590/S1679-87592013000100007
Costa-Júnior, O. S., Attrill, M. J., Pedrini, A. G., & De Paula, J. C. (2002). Spatial and seasonal distribution of seaweeds on coral reefs from Southern Bahia, Brazil. Botanica Marina, 45, 346–355. https://doi.org/10.1515/BOT.2002.035
Costa, I. O., Caires, T. A., Pereira-Filho, G. H., & Nunes, J. M. C. (2012). Macroalgas bentônicas associadas a bancos de Hypnea musciformis (Wulfen) J.V. Lamour. (Rhodophyta-Gigartinales) em duas praias do litoral baiano. Acta Botanica Brasilica, 26, 493–507. https://doi.org/10.1590/S0102-33062012000200025
Dawes, C., & Mathieson, A. (2008). The seaweeds of Florida. Gainesville: University of Florida Press.
Diez, I., Santolaria, A., & Gorostiaga, J. M. (2003). The relationship of environmental factors to the structure and distribution of subtidal seaweed vegetation of the western Basque coast (N Spain). Estuarine, Coastal and Shelf Science, 56, 1041–1054. https://doi.org/10.1016/S0272-7714(02)00301-3
Ferreira, S. M. C., Lolis, L. A., Noga, P. M., Affe, H. M. J., & Nunes, J. M. C. (2022). A highly diverse phytobenthic community along a short coastal reef gradient in northeastern Brazil. Universitas Scientiarum, 27, 34–56. https://doi.org/10.11144/Javeriana.SC271.ahdp
Figueiredo, M. A. O., Barreto, M. B. B., & Reis, R. P. (2004). Caracterização das macroalgas nas comunidades marinhas da área de Proteção Ambiental de Cairuçú, Parati, RJ – Subsídios para futuros monitoramentos. Revista Brasileira de Botânica, 27, 11–17. https://doi.org/10.1590/S0100-84042004000100002
Figueiredo, M. A. O., Horta, P.A., Pedrini, A. G., & Nunes, J. M. C. (2008). Benthic marine algae of the coral reefs of Brazil: a literature review. Oecologia Brasiliensis, 12, 259–270. https://doi.org/10.4257/oeco.2008.1202.07
Guiry, M. D., & Guiry, G. M. (2025). AlgaeBase. World-wide electronic publication, University of Galway. Retrieved on February 21, 2025 from: https://www.algaebase.org
Horta, P. A., Amancio, E., Coimbra, C. S., & Oliveira, E. C. (2001). Considerações sobre a distribuição e origem da flora de macroalgas marinhas brasileiras. Hoehnea, 28, 243–265.
Horta, P. A., Salles, J. P., Bouzon, J., Scherner, F., Cabral, D., Bouzon, Z. L. et al. (2008). Composição e estrutura dos fitobentos do infralitoral da Reserva Biológica Marinha do Arvoredo, Santa Catarina, Brasil —implicações para a conservação. Oecologia Brasiliensis, 12, 51–57. https://doi.org/10.4257/oeco.2008.1202.06
Hurd, C. L., Harrison, P. J., & Druehl, L. D. (1996). Effect of seawater velocity on inorganic nitrogen uptake by morphologically distinct forms of Macrocystis integrifolia from wave-sheltered and exposed sites. Marine Biology, 126, 205–214. https://doi.org/10.1007/BF00347445
Kevekordes, K. (2001). Toxicity tests using developmental stages of Hormosira banksii (Phaeophyta) identify ammonium as a damaging component of secondary treated sewage effluent discharged into Bass Strait, Victoria, Australia. Marine Ecology Progress Series, 219, 139–148. https://doi.org/10.3354/meps219139
Leigh, E. G., Paine, R. T., Quinn, J. F., & Suchanek, T. H. (1987). Wave energy and intertidal productivity. Proceedings of the National Academy of Sciences, 84, 1314–1318. https://doi.org/10.1073/pnas.84.5.1314
Littler, D. S., & Littler, M. M. (2000). Caribbean reef plants. An identification guide to the reef plants of the Caribbean, Bahamas, Florida and Gulf of Mexico. Washington D.C.: OffShore Graphics.
Macedo, T. S., Varjão, A. S., Fernandes, L. L., Silva, D. F., Almeida, J. S., Messias, J. C. et al. (2009). Levantamento taxonômico e diversidade das macroalgas marinhas bentônicas da praia da Pituba, Salvador, Bahia. Revista Eletrônica de Biologia, 2, 29–39.
Marins, B. V., Brasileiro, P. A., Barreto, M. B. B., Nunes, J. M. C., Yoneshigue-Valentin, Y., & Amado-Filho, G. M. (2008). Subtidal benthic marine algae of the Todos os Santos Bay, Bahia State, Brazil. Oecologia Brasiliensis, 12, 229–242. https://doi.org/10.4257/oeco.2008.1202.05
Melo, A. S. (2008). O que ganhamos “confundindo” riqueza de espécies e equabilidade em um índice de diversidade? Biota Neotropica, 8, 21–27. https://doi.org/10.1590/s1676-06032008000300001
Muñoz, A. O. M., & Pereira, S. M. B. (1997). Caracterização quali-quantitativa das comunidades de macroalgas nas formações recifais da praia do Cupe Pernambuco, Brasil. Trabalhos Oceanográficos da Universidade Federal do Pernambuco, 25, 93–109. https://doi.org/10.5914/tropocean.v25i1.2731
Nascimento, O. S. (2013). Impactos da urbanização sobre a estrutura, cobertura de morfotipos funcionais e heterogeneidade química das comunidades de algas
recifais (M.Sc. Thesis). Instituto de Biologia, Universidade Federal da Bahia, Salvador.
Nunes, J. M. C. (1998a). Catálogo de algas marinhas bentônicas do Estado da Bahia. Acta Botanica Malacitana, 23, 5–21. https://doi.org/10.24310/abm.v23i0.8547
Nunes, J. M. C. (1998b). Rodofíceas marinhas bentônicas da orla oceânica de Salvador, Estado da Bahia, Brasil. Insula, 49, 27–37.
Nunes, J. M. C. (1999). Phaeophyta da Região Metropolitana de Salvador, Bahia, Brasil (M.Sc. Thesis). Instituto de Biociências, Universidade de São Paulo, São Paulo.
Nunes, J. M. C. (2005a). A família Liagoraceae (Rhodophyta, Nemaliales) no Estado da Bahia, Brasil. Hoehnea, 32, 429–444.
Nunes, J. M. C. (2005b). Rodofíceas marinhas bentônicas do Estado da Bahia, Brasil (Ph.D. Thesis). Instituto de Biociências, Universidade de São Paulo, São Paulo.
Nunes, J. M. C. (2010). Taxonomia morfológica: metodologia de trabalho. In A. G. Pedrini (Org.), Macroalgas: uma introdução à Taxonomia (pp. 53–70). Rio de Janeiro, Rio de Janeiro: Technical Books.
Nunes, J. M. C., & Guimarães, S. M. P. B. (2008). Novas referências de rodofíceas marinhas bentônicas para o litoral brasileiro. Biota Neotrópica, 8, 89–100. https://doi.org/10.1590/S1676-06032008000400008
Nunes, J. M. C., & Guimarães, S. M. P. B. (2009). Primeira referência de plantas gametofíticas em Spermothamnion nonatoi (Ceramiales, Rhodophyta). Rodriguésia, 60, 259–264. https://www.jstor.org/stable/23499987
Nunes, J. M. C., & Guimarães, S. M. P. B. (2010). Morfologia y toxonomía de Scinaia halliae (Scinaiaceae, Rhodophyta) em el litoral de Bahia y Espírito Santo, Brasil. Revista
de Biología Marina y Oceonografía, 45, 159–164. http://dx.doi.org/10.4067/S0718-19572010000100017
Nunes, J. M. C., & Paula, E. J. (2000). Estudos taxonômicos do gênero Padina Adanson (Dictyotaceae – Phaeophyta) no litoral do Estado da Bahia, Brasil. Acta Botanica Malaci-
tana, 26, 21–43. https://doi.org/10.24310/abm.v25i0.8470
Nunes, J. M. C., & Paula, E. J. (2001). O gênero Dictyota Laumouroux (Dictyotaceae – Phaeophyta) no litoral do Estado da Bahia, Brasil. Acta Botanica Malacitana, 26, 5–18. https://doi.org/10.24310/abm.v26i0.7375
Nunes, J. M. C., & Paula, E. J. (2002). Composição e distribuição das Phaeophyta nos Recifes da Região Metropolitana de Salvador, Bahia, Brasil. Iheringia, 57, 113–130.
Nunes, J. M. C., & Paula, E. J. (2004a). Chnoosporaceae, Scytosiphonaceae, Sporochnaceae e Sphacelariaceae (Phaeophyta) no Estado da Bahia, Brasil. Biotemas, 17, 7–28.
Nunes, J. M. C., & Paula, E. J. (2004b). Estudos taxonômicos de Ectocarpaceae e Ralfsiaceae (Phaeophyta) da Região Metropolitana de Salvador, Ba, Brasil. Acta Biologica Leopoldensia, 26, 37–50.
Nunes, J. M. C., & Paula, E. J. (2006). O gênero Dictyopteris J.V.Lamour. (Dictyotaceae – Phaeophyta) no estado da Bahia, Brasil. Hidrobiológica, 16, 251–258. https://hidro
biologica.izt.uam.mx/index.php/revHidro/article/view/1036
Nunes, J. M. C., Santos, A. C. C., & Santana, L. C. (2005). Novas ocorrências de algas marinhas bentônicas para o Estado da Bahia, Brasil. Iheringia Série Botânica,60,
99–106. https://isb.emnuvens.com.br/iheringia/article/view/209
Oliveira, E. C., & Qi, Y. (2003). Decadal changes in a polluted bay as seen from its seaweed flora: the case of Santos Bay in Brazil. AMBIO: A Journal of the Human Environment, 32, 403–405. https://doi.org/10.1579/0044-7447-32.6.403
Orfanidis, S., Panayotidis, P., & Stamatis, N. (2001). Ecological evaluation of transitional and coastal waters: a marine benthic macrophytes-based model. Mediterranean Marine Science, 2, 45–65. https://doi.org/10.12681/mms.266
Pedrini, A. G. (2013). Macroalgas (ocrófitas multicelulares) marinhas do Brasil. Rio de Janeiro, Rio de Janeiro: Technical Books.
Scherner, F., Barufi, J. B., & Horta, P. A. (2012). Photosynthetic response of two seaweed species along an urban pollution gradient: evidence of selection of pollution-tolerant species. Marine Pollution Bulletin, 64, 2380–2390. https://doi.org/10.1016/j.marpolbul.2012.08.012
Scherner, F., Horta, P. A., De Oliveira, E. C., Simonassi, J. C., Hall-Spencer, J. M., Chow, F. et al. (2013). Coastal urbanization leads to remarkable seaweed species loss and community shifts along the SW Atlantic. Marine Pollution Bulletin, 76, 106–115. https://doi.org/10.1016/j.marpolbul.2013.09.019
Steneck, R. S., & Dethier, M. N. (1994). A functional group approach to the structure of algal-dominated communities. Oikos, 69, 476-498. http://dx.doi.org/10.2307/3545860
Villaça, R., Yoneshigue-Valentin, Y., & Boudouresque, C. F. (2008). Estrutura da comunidade de macroalgas do infralitoral do lado exposto da ilha de Cabo Frio (Arraial do Cabo, RJ). Oecologia Brasiliensis, 12, 206–221. https://doi.org/10.4257/oeco.2008.1202.03
Villaça, R., Carvalhal-Fonseca, A., Jensen, V. K., & Knoppers, B. (2010). Species composition and distribution of macroalgae on Atol das Rocas, Brazil, SW Atlantic. Botanica Marina, 53, 113–122. https://doi.org/10.1515/BOT.2010.013
Zar, J. H. (2010). Biostatistical analysis. Upper Saddle River, New Jersey: Prentice Hall International.

El grupo pallida del género Phyllophaga (Coleoptera: Melolonthidae) en Oaxaca: nuevos registros y descripción de una especie nueva
The pallida group of Phyllophaga (Coleoptera: Melolonthidae) in Oaxaca: new records and description of a new species
Axel Teodoro Cortes-Dávila a, Salvador Lozano-Trejo a, Andrés Ramírez-Ponce b y Jesús Alberto Cruz-López c, *
a Instituto Tecnológico del Valle de Oaxaca, División de Estudios de Posgrado e Investigación, Ex-hacienda de Nazareno, Agencia de Policía de Nazareno Xoxo, Centro, 71233 Santa Cruz Xoxocotlán, Oaxaca, México
b Instituto de Ecología A.C., Red de Biodiversidad y Sistemática, Carretera Antigua a Coatepec 351, El Haya, 91070 Xalapa, Veracruz, México
c Instituto Nacional de Investigaciones Forestales, Agrícolas y Pecuarias, Campo Experimental Valles Centrales de Oaxaca, Melchor Ocampo Núm. 7, Santo Domingo Barrio Bajo, 68200 Villa de Etla, Oaxaca, México
*Autor para correspondencia: thelyphonidito@gmail.com (J.A. Cruz-López).
Recibido: 24 febrero 2025; aceptado: 23 julio 2025
http://zoobank.org/urn:lsid:zoobank.org:pub:927D7BB0-0BC3-4C5F-8EA1-E2F61BEC7254
Resumen
Uno de los grupos de especies pertenecientes al género Phyllophaga subgénero Phytalus que han recibido especial atención, es el grupo pallida. Actualmente, este grupo está conformado por 12 especies, de las cuales, ninguna se ha reportado para el estado de Oaxaca. En el presente trabajo, se reporta la presencia de este grupo para Oaxaca por primera vez, con base en la revisión del material depositado en el Laboratorio de Entomología Agrícola del Campo Experimental Valles Centrales de Oaxaca – INIFAP. Se aportan nuevos registros para Phyllophaga (Phytalus) dieteriana Deloya et Morón, 1998 y se describe a Phyllophaga (Phytalus) psittacina sp. nov., con base en ejemplares adultos de ambos sexos provenientes de Santa María Tecomavaca, en la Reserva de la Biosfera Tehuacán-Cuicatlán.
Palabras clave: Phytalus; Agave; Selva baja caducifolia; Reserva de la Biosfera Tehuacán-Cuicatlán
Abstract
One of the species groups belonging to the genus Phyllophaga, subgenus Phytalus that has received special attention is the pallida group. Currently, this group includes 12 species, of which none of them has been reported for Oaxaca State. Based on the review of the material deposited in the Laboratorio de Entomología Agrícola of the Campo Experimental Valles Centrales de Oaxaca – INIFAP, we report the presence of this group for the first time for Oaxaca. We provide new records for Phyllophaga (Phytalus) dieteriana Deloya & Morón, 1998, and the description of Phyllophaga (Phytalus) psittacina sp. nov., based on specimens of both sexes from Santa María Tecomavaca, in the Tehuacán-Cuicatlán Biosphere Reserve.
Keywords: Phytalus; Agave; Low deciduous forest; Tehuacán-Cuicatlán Biosphere Reserve
Introducción
El género Phyllophaga Harris, 1827 es uno de los grupos más diversos de la tribu Melolonthini (Coleoptera: Melolonthidae: Melolonthinae), agrupa a un total de 782 especies conocidas en América (Allsopp y Schoolmeesters, 2024). Actualmente, la taxonomía del género está sustentada en caracteres morfológicos externos, principalmente de la morfología de las uñas (Morón, 2015a, b). Sin embargo, estudios sobre la filogenia de Phyllophaga han demostrado que el esquema taxonómico actual debe ser redefinido con base en combinación de caracteres morfológicos que respalden grupos monofiléticos (Rivera-Gasperín y Morón, 2013, 2017a, b). En este sentido, el subgénero Phytalus Erichson, 1847 se reconoce por el ápice de las uñas profundamente hendido o bífido (Morón, 1986). Sin embargo, este subgénero es uno de los que requiere mayor atención taxonómica para el adecuado sustento de sus respectivos grupos. Actualmente, dentro de Phytalus se reconocen 8 grupos de especies, de los cuales solo 3 han sido revisados, teniendo en cuenta las aproximaciones filogenéticas mencionadas anteriormente (Morón, 2006, 2018).
Uno de éstos, el grupo pallida, está conformado por 12 especies distribuidas en los estados de Chihuahua, Colima, Guerrero, Jalisco, Michoacán, Morelos, Nayarit, Puebla, Sinaloa y Sonora, a lo largo de la vertiente del Pacífico, principalmente sobre la provincia biogeográfica Tierras Bajas de Pacífico sensu Morrone et al. (2017). Este grupo puede ser reconocido por tener cuerpo alargado, ovalado, con una longitud de 10 a 15 mm, color pardo rojizo a amarillo, pronoto ligeramente más ancho que la cabeza, antenas formadas por 10 artejos, con los antenómeros tercero al quinto cortos, maza antenal masculina más larga o de igual longitud que el funículo, pronoto y élitros glabros, espolones metatibiales articulados y dentículo inferior de las uñas tarsales de mayor o de similar longitud que el dentículo superior (Morón, 1986, 2006; Romero-López y Morón, 2017). Las especies de este grupo se han recolectado en matorrales xerófilos, bosques espinosos o en bosques tropicales caducifolios, generalmente a no más de 1,000 m snm; son especies poco abundantes, que son atraídas a la luz fluorescente blanca y luz negra (Morón, 2006; Romero-López y Morón, 2017).
De acuerdo con la lista de especies de Phyllophaga reportadas para Oaxaca (Hernández-Cruz et al., 2016), ninguna de las especies del grupo pallida ha sido registrada. Durante la revisión del material depositado en el Laboratorio de Entomología Agrícola del INIFAP – Oaxaca, se hallaron ejemplares de Phyllophaga (Phytalus) dieteriana Deloya et Morón, 1998, que representan el registro más austral para México. Adicionalmente, durante un muestreo de melolóntidos en la Reserva de la Biosfera de Tehuacán-Cuicatlán se recolectaron ejemplares de Phyllophaga (Phytalus) psittacina sp. nov., especie que es descrita a continuación con base en ejemplares de ambos sexos.
Materiales y métodos
Los ejemplares tipo se encuentran depositados en la Colección Entomológica del Instituto de Ecología, A.C. (IEXA), Xalapa, Veracruz y en la Colección Nacional de Insectos (CNIN), Instituto de Biología-UNAM, México. Material adicional se encuentra depositado en la Colección Entomológica del Campo Experimental Valles Centrales de Oaxaca (INIFAP). Los ejemplares de P. (Phytalus) psittacina sp. nov., fueron capturados mediante un muestreo aleatorio realizado durante agosto del 2023 y muestreos semanales realizados de febrero del 2024 a enero del 2025, en una parcela de agave espadín Agave angustifolia Haw. localizada en vegetación de selva baja caducifolia, bosque de cactáceas y matorral xerófilo. Para la captura de los ejemplares se utilizó una trampa de luz blanca led de impacto modificada para colectar escarabajos adultos, la cual se ubicó en el centro de la parcela de cultivo. La fuente de luz fue una lámpara de luz blanca led recargable de 36 W. La trampa utilizada se modificó a partir de la trampa de luz tipo embudo propuesta por Morón y Terrón (1988), cambiando al embudo por una pantalla de mica plástica transparente en forma de cruz, de 40 cm de diámetro por 50 cm de alto. El recipiente colector consistió en un bote circular de plástico con capacidad de 20 L, el cual contenía 10 L de solución de agua y jabón como lo sugiere Aragón-García et al. (2018), con la finalidad de romper la tensión superficial del agua y permitir que los ejemplares se hundan en la solución. La trampa estuvo activa desde las 19:00 hasta las 00:00 hrs. Los ejemplares de P. (Phytalus) dieteriana fueron capturados mediante un esfuerzo comunitario entre un técnico promotor de campo y productores de la comunidad adscritos al programa federal Sembrando Vida del municipio de Tezoatlán de Segura y Luna, en Oaxaca. La recolecta de estos ejemplares consistió en la participación ciudadana como parte de las actividades entre los productores, para que, de manera general, conozcan a los insectos asociados con el cultivo de maíz.
Posterior a la recolecta de los ejemplares, éstos fueron etiquetados y transportados al Laboratorio de Entomología Agrícola del INIFAP – Oaxaca para el subsecuente montaje en alfileres entomológicos de acuerdo con los lineamientos descritos por Márquez-Luna (2005). La extracción y montaje de genitales masculinos siguió el procedimiento descrito por Gutiérrez-Carranza (2023). Para la extracción de las placas genitales femeninas se tomó en cuenta, parcialmente, el protocolo de Gutiérrez-Carranza (2023), con la excepción de que estas estructuras fueron limpiadas a mano del tejido adicional, las placas genitales inferiores se separaron entre sí para poder observar a detalle los márgenes completos y, finalmente, estas estructuras fueron preservadas en un microvial con alcohol junto con la respectiva hembra. Todos los ejemplares (n = 10) fueron disecados.
Para la identificación de los ejemplares recolectados, se utilizaron las claves para el reconocimiento de subgéneros y grupos de especies de Morón (1986), así como la monografía del grupo pallida realizada por Morón (2006), también se revisaron las descripciones originales de las especies del grupo descritas en trabajos diferentes a los mencionados: Morón (1992), Deloya y Morón (1998) y Romero-López y Morón (2017). El criterio para reconocer a esta especie como una nueva perteneciente al grupo pallida, fue con base en la revisión de los trabajos citados, así como la comparación detallada tanto de los genitales masculinos y femeninos.
Para la toma de fotografías, se utilizó una cámara Color CMOS C-Mount Microscope Camera, adaptada a un estereoscopio Jewelry Gem Zoom Stereo Microscope con el programa AmScope v. 3.1. Para corregir los efectos de la iluminación artificial se utilizó el domo de iluminación LED armable para fotografía científica y microscópica recomendado por Kawada y Buffington (2016). Se tomaron fotografías a diferentes profundidades, las cuales posteriormente fueron ensambladas mediante el programa Helicon Focus v. 6.7.1.
La terminología usada para la descripción taxonómica de los adultos fue tomada de Morón (2006), la cual es acorde con el grupo de especies tratadas en el presente trabajo. Para establecer la variación, todos los ejemplares (n = 10) fueron medidos.
Resultados
Phyllophaga (Phytalus) dieteriana Deloya et Morón, 1998
Figs. 1, 8
Resumen taxonómico
Distribución conocida. Esta especie ha sido registrada en Morelos y Puebla, en ecosistemas de bosque tropical caducifolio y matorral crasicaule ubicados entre 940 y 1,600 m snm (Morón, 2006).
Registros nuevos.Diecinueve ♂♂, 4 ♀♀ (INIFAP-Oax), México: Oaxaca: Tezoatlán de Segura y Luna, San Isidro el Naranjo (17°36’21.6” N, 97°47’24” O), 27/junio/2022, Col. C. A. Cruz-López. 2 ♀ (INIFAP-Oax), México: Oaxaca: Tezoatlán de Segura y Luna, Tezoatlán centro (17°38’49.2” N, 97°49’1.2” O), 27/junio/2022, Col. C. A. Cruz-López. 2 ♂♂ (INIFAP-Oax) México: Oaxaca: Tezoatlán de Segura y Luna, Juquila de León (17°22’58.8” N, 97°28’8.4” O), 27/junio/2022, Col. C. A. Cruz-López.
Comentarios taxonómicos
Esta especie es fácilmente reconocible dentro del resto del grupo pallida ya que los machos presentan la espuela metatibial inferior muy recurvada (fig. 1B) y la parte distal de los parámeros con numerosas estrías transversales (fig. 1C).

Phyllophaga (Phytalus) psittacina sp. nov.
https://zoobank.org/urn:lsid:zoobank.org:act:F9A4EBC3-67CA-4E61-A7D6-F421DF338E14
Figs. 2-8.
Descripción. Holotipo: (♂, IEXA) con etiqueta roja. Longitud total 19.0 mm, ancho máximo 6.0 mm. Cabeza y pronoto pardo rojizos, élitros, patas y abdomen de color amarillo pajizo brillante (fig. 2A, B). Clípeo semitrapezoidal, con ápice redondeado, ligeramente bilobulado y borde anterior poco elevado; 3.2 veces más ancho que largo; disco clipeal glabro, convexo, con puntos circulares profundos, irregularmente distribuidos (figs. 2A, 3A). Sutura fronto-clipeal bien marcada, ligeramente sinuada. Frente glabra, 2.0 veces más ancha que larga, en sus 2/3 anteriores casi plana con puntos circulares profundos grandes y puntos circulares pequeños irregularmente distribuidos; tercio posterior completamente liso, con muy pequeños puntos someros (figs. 2A, 3A).

Antenas con 10 artejos; segundo artejo más largo y ancho que los artejos III, IV y V, siendo éstos más pequeños, con longitud similar y una proyección anterior semicónica; artejos VI y VII similares en largo y ancho al segundo artejo, y con una proyección anterior semicónica (fig. 3A). Maza antenal más larga que el flagelo (0.9:0.5), formada por 3 lamelas cubiertas con sensilas finas y algunas sedas erectas esparcidas. Anchura dorsal de cada ojo equivalente a 30% de la distancia interocular. Canto ocular corto y estrecho, con 10 sedas (fig. 3A).
Pronoto 1.6 veces más ancho que largo; relación anchura máxima de cabeza-anchura máxima de pronoto de 0.6:1; relación distancia interocular-anchura máxima de pronoto de 0.3:1.
Disco pronotal glabro, con puntos circulares moderadamente profundos, irregularmente esparcidos, más densos hacia la porción media basal. Ángulos anteriores y posteriores obtusos, muy prominentes (figs. 2B, 3A). Bordes laterales levemente redondeados, angulados en su porción central; mitad anterior ligeramente angulada y mitad posterior casi recta; márgenes laterales crenulados en toda su longitud, sobre todo en la mitad anterior (figs. 2B, 3A). Margen basal bien delimitado cerca de ángulos posteriores y ligeramente difuso en parte central (fig. 3A). Escutelo más ancho que largo, con pocos puntos someros dispuestos irregularmente (fig. 3A).
Élitros 3.9 veces más largos que anchos, glabros, con textura punteada de manera irregular pero densa. Reborde de la epipleura muy estrecho, que se desvanece a la altura del propigidio, con sedas cortas muy esparcidas. Callos humerales anteriores prominentes y redondeados; callos posteriores amplios, redondeados, poco prominentes. Alas metatorácicas completamente desarrolladas (fig. 2A, B).

Quinto esternito abdominal poco más largo que el cuarto, con área media central excavada, punteada-rugosa, y algunas sedas erectas cerca del borde posterior, flanqueado por rebordes redondeados transversales (fig. 4A). Placa anal corta, cóncava, con reborde basal transversal y 6 sedas finas cerca de borde posterior.
Protibias más cortas que el tarso respectivo (1:1.3); borde externo con 3 procesos dentiformes, el apical más largo y angosto, ligeramente recurvado, el intermedio grande y ancho, el proximal corto y redondeado (fig. 3A). Espolón protibial ligeramente curvo y aguzado, poco más corto que el segundo protarsómero. Mesotibias con quilla media, setífera transversal oblícuamedia en cara externa bien marcada, precedida por quilla prebasal incompleta. Espolones mesotibiales articulados, rectos, aguzados; externo ligeramente más corto que el interno. Metatibias poco más cortas que tarsos respectivos (1:1.2), con quilla setífera transversal oblicua en tercio distal de la cara externa. Ambos espolones metatibiales articulados; espolón superior más ancho que inferior, poco recurvado, con ápice redondeado, flabelado; espolón inferior recurvado, poco más corto que el superior, con ápice redondeado; espolón superior más largo que primer metatarsómero y el inferior tan largo como éste (fig. 5A). Uñas tarsales profundamente hendidas; dentículo superior de igual longitud que el inferior, ligeramente más estrecho. (fig. 3B).
Cápsula genital masculina con parámeros completamente fusionados en la falobase; extremos apicales punteados y ligeramente divergentes, formando una estructura semicónica, recurvada; ventralmente cada parámero con proyección excavada central que convergen en la parte media, y en la parte basal con una concavidad circular que en conjunto asemejan a un número “8” (fig. 6). Superficie del tectum ampliamente cóncava. Edeago carece de estructura de soporte esclerosada y de ornamentos conspicuos.
Hembra (paratipo IEXA). Muy similar al macho, sin dimorfismo sexual notorio. De tamaño similar al macho, proporciones de las patas similares, abdomen ligeramente más ensanchado y longitud de la maza antenal ligeramente más corta (fig. 2C, D). En cuanto a los espolones metatibiales, el externo tiene casi la misma longitud que el interno, y ambos son apicalmente espatulados (fig. 5B). El carácter sexual más distintivo es la ornamentación del esternito abdominal V, el cual no presenta ningún tipo de rebordes ni ornamentaciones, solo se encuentra cubierto por pocas sedas, esparcidas (fig. 4B). Las placas genitales superiores son alargadas, en forma de lanceta, fusionadas en casi toda su longitud y terminando en un ápice agudo inferior. Por la parte superior, estas placas están ligeramente divergentes, con los bordes engrosados y ligeramente dirigidos hacía la parte ventral, con 6 sedas apicales. Placas inferiores menos esclerosadas, de forma semitriangular y envuelven la mitad inferior de las placas superiores (fig. 7).
Variación. Ambas hembras (n = 2) exhiben tamaños similares, que son ligeramente más grandes que el macho (longitud: 20 mm). Los machos (n = 8) presentan tamaños y proporciones muy similares.
Diagnosis. Esta especie puede ser reconocida de las restantes del grupo pallida por la siguiente combinación de caracteres morfológicos: uñas con dentículo inferior de longitud similar al superior (fig. 3B); maza antenal de similar longitud que el escapo (fig. 3A); quinto esternito abdominal con reborde marcado, en forma de media luna, abarcando más de la mitad del ancho del segmento (fig. 4A); ápices de los parámeros divergentes, con bordes ventrales con concavidad media (fig. 6); placas genitales inferiores fusionadas, en forma de lanceta, con ápices divergentes (fig. 7).

Resumen taxonómico
Material examinado (n = 10): holotipo (♂, IEXA). México: Oaxaca: Santa María Tecomavaca (17°57’46.8” N, 97°01’12” O), 24/agosto/2024, Col. Cortes-Dávila, A. T. 1♂ paratipo (IEXA) con los mismos datos que el holotipo. Un ♂, 2 ♀♀ (IEXA) paratipos con los mismos datos de colecta, pero con fecha del 12/agosto/2023. Un ♂ paratipo (IEXA) con los mismos datos de colecta, pero con fecha del 03/agosto/2024. Dos ♂♂, 1 ♂ y 1 ♂ paratipos (CNIN) con los mismos datos de colecta, pero con las siguientes fechas: 17/agosto/2024, 29/agosto/2024 y 31/agosto/2024, respectivamente.
Etimología. El nombre específico se refiere a la similitud de los parámeros con el pico de la guacamaya verde Ara militaris (Linnaeus, 1766), ave perteneciente al orden Psittaciformes. Adicionalmente, la localidad tipo pertenece al santuario de la guacamaya verde, en la Reserva de la Biosfera Tehuacán-Cuicatlán.
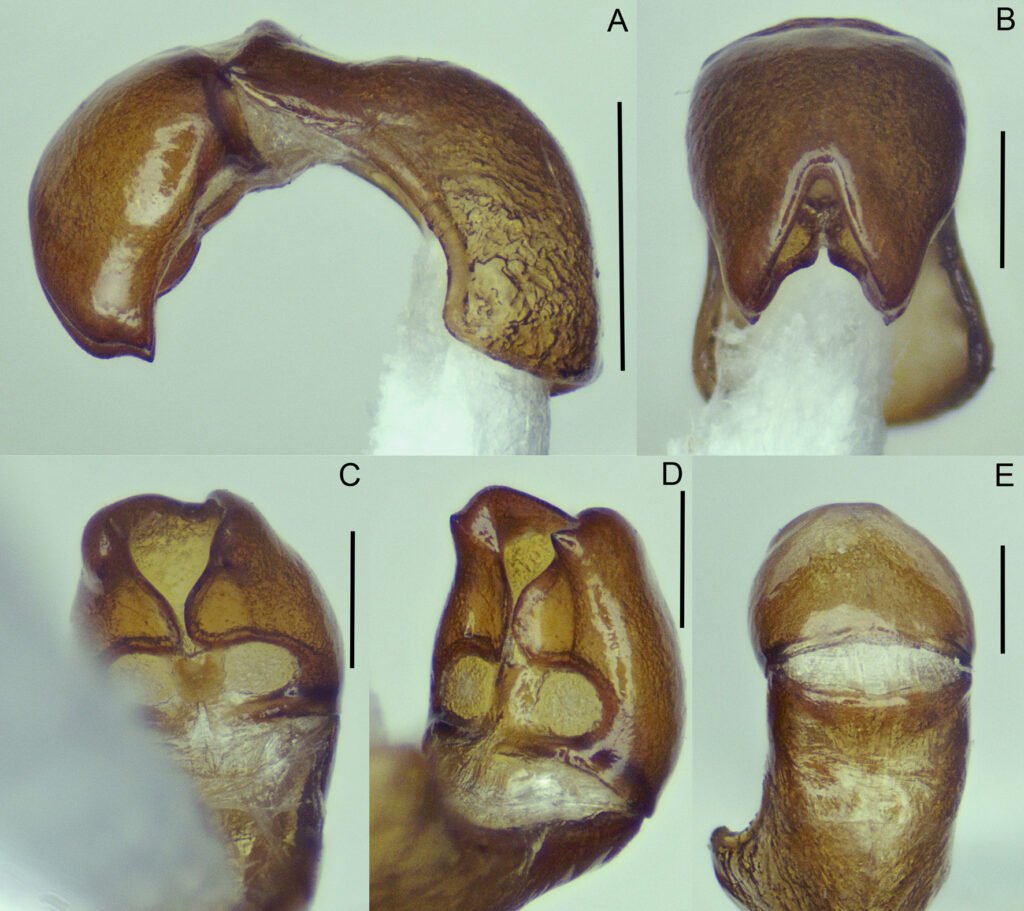
Distribución. Esta especie solo se conoce de la localidad tipo (fig. 8).
Historia natural. Se observó a los escarabajos adultos de P. (Phytalus) psittacina sp. nov. volar y posarse durante la noche en las copas de los árboles de las especies Prosopis laevigata (Humb. et Bonpl. ex Willd) (mezquite blanco), Acacia farnesiana (L.) Willd (huizache) y Citrus aurantifolia (Christm.) Swingle (limón). También se observó que los escarabajos adultos suelen llevar a cabo sus vuelos nupciales en parcelas de cultivo de agave espadín, presumiblemente para alimentarse de las flores o copular en estas plantas. Esta especie convive con los siguientes Rhizotrogina: Phyllophaga (Phyllophaga) lenis (Horn, 1887) y Listrochelus cuicatecus (Morón y Aragón, 1997).
Comentarios taxonómicos
Dentro del grupo pallida, P. (Phytalus) psittacina sp. nov. Es similar a P. (Phytalus) huiteaca Morón, 2006y P. (Phytalus) dieteriana, en la forma del cuerpo, el cual es de tamaño mediano y proporcionalmente esbelto, con una longitud entre 10 a 15 mm, el color pardo rojizo a amarillo, pronoto ligeramente más ancho que la cabeza, antenas formadas por 10 artejos, del tercer al quinto antenómero cortos, maza antenal masculina más larga o de igual longitud que el funículo, pronoto y élitros glabros. En cuanto a la forma de la cápsula genital, P. (Phytalus) psittacina sp. nov. es similar a P. (Phytalus) huiteaca, ya que ambas especies poseen una concavidad media en la parte ventral de los parámeros, sin embargo, en P. (Phytalus) huiteaca los márgenes ventrales que prosiguen de esta concavidad divergen notablemente hasta converger en los ápices, lo cual es inverso en P. (Phytalus) psittacina sp. nov. Desafortunadamente, se desconoce la hembra de P. (Phytalus) huiteaca por lo que las placas genitales no pueden compararse. En cuanto a la morfología genital de la hembra, esta tiene una configuración única entre las especies del grupo, donde las placas genitales inferiores en su parte superior, tienen ligera similitud con aquellas de Phyllophaga (Phytalus) reyesiana, Morón 1992, con los bordes ligeramente divergentes y ornamentados con sedas.
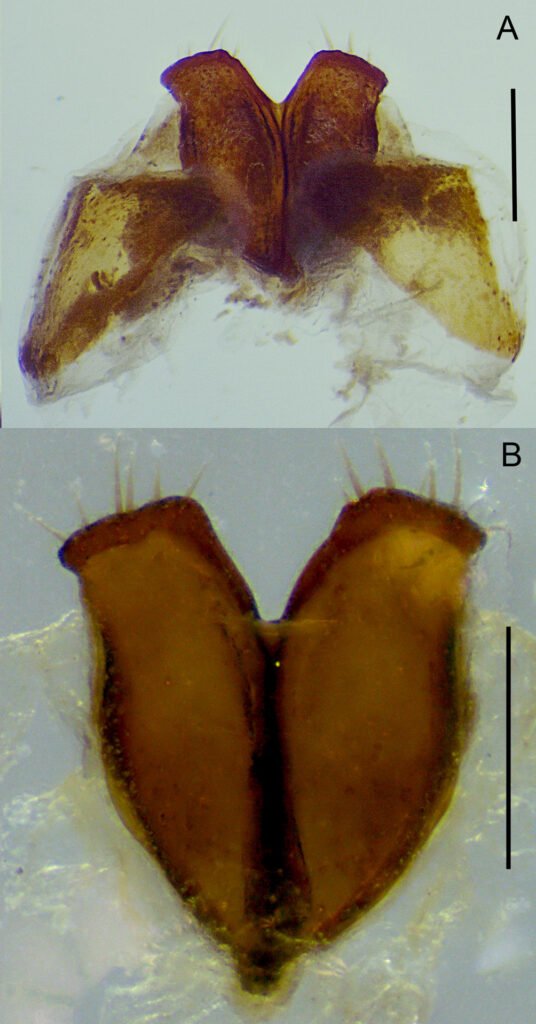
Discusión
De acuerdo con Morón et al. (2014), la abundancia de escarabajos melolóntidos puede categorizarse de acuerdo con el número de ejemplares capturados por métodos convencionales para el grupo. En este sentido, a lo largo del año 2024, se recolectó un total de 7 ejemplares de P. (Phytalus) psittacina sp. nov., todos machos, por lo que esta especie puede considerarse como escasa dentro del rango de clasificación. Las únicas 2 hembras conocidas fueron recolectadas en muestreos aleatorios previos durante el 2023, realizados con la idea de tener una aproximación de la fauna de la zona de estudio. Por otro lado, P. (Phytalus) dieteriana se considera como una especie con abundancia baja (27 ejemplares) en la población reportada como nuevo registro en Oaxaca. Para esta especie, de igual manera la mayor cantidad de ejemplares recolectados fueron machos. La abundancia de los ejemplares de las especies del grupo parece ser baja, capturándose no más de 35 ejemplares por especie (Morón, 2006).
Diversos estudios faunísticos enfocados a la riqueza, diversidad y abundancia de escarabajos melolóntidos en Mesoamérica reportan la presencia de un número variable de especies del género Phyllophaga, así como de otros géneros relacionados de la subtribu Rhizotrogina (e.g., Chlaenobia Blanchard, 1850 y Listrochelus Blanchard, 1850). Los datos de riqueza oscilan entre 4 y 24 especies de Phyllophaga (Aragón-García et al., 2008; Castro-Ramírez et al., 2005; Cuate-Mozo et al., 2016; Delgado et al., 2012; Deloya et al., 1995; Doskocil et al., 2008; Mateos-Escudero et al., 2021; Márquez-Manzano et al., 2023; Ramírez-Ponce et al., 2009). En la localidad tipo de P. (Phytalus) psittacina sp. nov., se está realizando un monitoreo de escarabajos melolóntidos que son atraídos a la luz tipo led blanca. Sorpresivamente, para esta zona, a lo largo de un monitoreo sistemático durante todo el 2024 (además de recolectas aleatorias durante el 2023), solo 3 especies de Rhizotrogina han sido capturadas: P. (Phytalus) psittacina sp. nov. (7 ejemplares), P. (Phyllophaga) lenis (15 ejemplares) y L. cuicatecus (142 ejemplares), consideradas como especies con abundancia moderada y alta, respectivamente. Estos muestreos se realizaron en 3 parcelas diferentes, que abarcan un área aproximada de 2.6 Km2. Morón et al. (2014) mencionan que en ambientes cálido-húmedos y cálido-secos predominan las especies con abundancias moderadas o bajas, como se observa en ambas especies de Phyllophaga. De igual manera, los trabajos enfocados en riqueza y abundancia, indican que existe una menor riqueza de especies en ambientes agrícolas en comparación con ambientes no perturbados, y a su vez, especies con abundancia alta y muy alta en el caso de escarabajos con hábitos alimenticios rizófagos. En la zona de estudio, los muestreos se están realizando en una parcela de agave, por lo que el tipo de ambiente cálido-seco (matorral xerófilo), más la presencia de cultivos agrícolas perennes, pueden ser los factores determinantes para la poca riqueza y abundancia, por lo menos de las especies de Rhizotrogina.
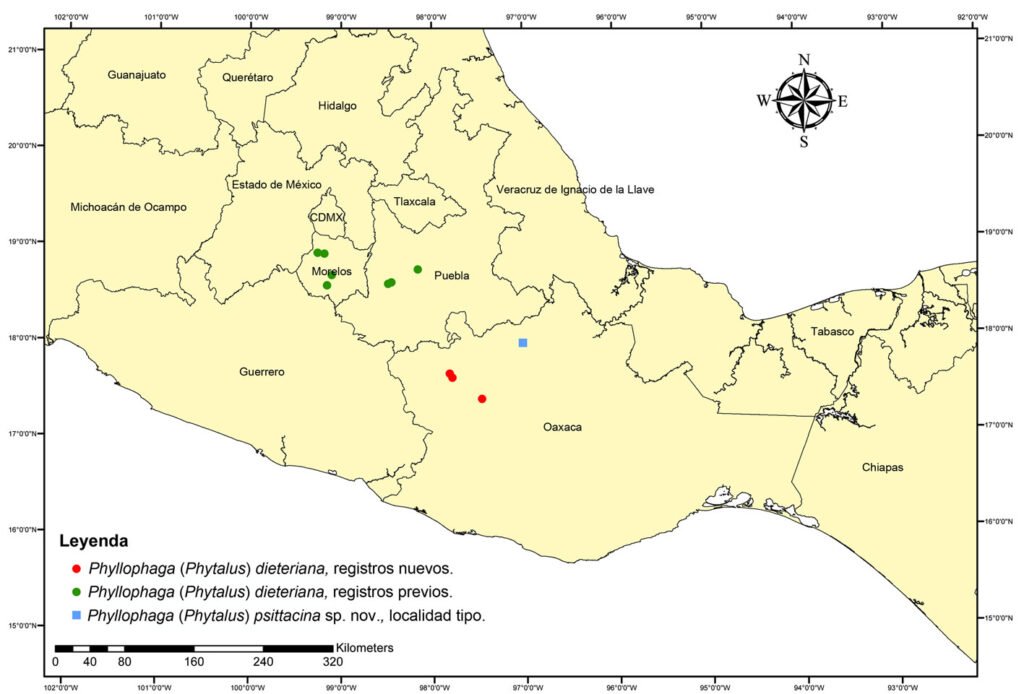
Con respecto a las restantes especies del grupo pallida, todas ellas se distribuyen en ambientes cálido-secos, abarcando matorrales xerófilos, bosques espinosos y bosques tropicales caducifolios, lo que nos indica la preferencia de estas especies por estos ambientes. La mayoría de las especies de este grupo se distribuye a lo largo de la vertiente del Pacífico, abarca casi en su totalidad a la provincia biogeográfica Tierras Bajas del Pacífico, con excepción de Phyllophaga (Phytalus) pallida (Horn, 1885) y Phyllophaga (Phytalus) sonora Saylor, 1939, las cuales se distribuyen entre los límites al norte de las provincias Tierras Bajas del Pacífico y Sonora (sensu Morrone, 2019; Morrone et al., 2017). Cabe resaltar el caso excepcional de P. (Phytalus) dieteriana, que es la única especie que se distribuye en el centro del país, en Morelos y Puebla, en la parte central de la provincia biogeográfica Cuenca del Balsas, incluyendo los nuevos registros reportados en el presente trabajo. Con la adición de P. (Phytalus) psittacina sp. nov., esta especie se distribuye en la provincia biogeográfica Valle de Tehuacán-Cuicatlán (sensu Rzedowski, 1978), la cual corresponde a la provincia más pequeña, de acuerdo con el criterio de Rzedowski. Sin embargo, de acuerdo con la propuesta de Morrone (2019), el área de distribución geográfica de la especie nueva, corresponde a los límites más hacia el centro-sur de la provincia Cuenca del Balsas. También Morrone (2019) indica que esta provincia se encuentra estrechamente relacionada con las Tierras Bajas del Pacífico, lo cual explicaría la distribución geográfica del grupo pallida. Fitogeográficamente, por lo menos la flora de Asteraceae del Valle de Tehuacán-Cuicatlán se relaciona con las de otras regiones áridas y semiáridas de la provincia Cuenca del Balsas y Tierras Bajas del Pacífico (Villaseñor, 1990); así como ambas provincias comparten un gran número de taxones endémicos de la familia Burseraceae (Becerra y Venable, 1999).
Agradecimientos
El primer autor agradece al Consejo Nacional de Humanidades, Ciencias y Tecnologías (Conahcyt), actualmente Secretaría de Ciencia, Humanidades, Tecnología e Innovación (Secihti), por la beca 1320122 y al Instituto Tecnológico del Valle de Oaxaca (ITVO) por su apoyo a la investigación. Este trabajo fue financiado por el Conahcyt, bajo el proyecto número 320715: “Modelo predictivo como estrategia de control de la gallina ciega (Phyllophaga spp.) con base en información climática y del microhábitat en el suelo en sistemas de mono- y policultivos de maíz nativo en Oaxaca”, apoyo otorgado al autor de correspondencia (JACL). Agradecemos el respaldo durante el trabajo de campo a los productores, por facilitar el acceso a sus parcelas donde se realizaron los muestreos. También agradecemos a César Augusto Cruz López, por su apoyo durante la recolecta de melolóntidos en la región de la Mixteca, Oaxaca. De igual manera, agradecemos a los estudiantes que nos apoyaron con la toma de fotografías y las recolectas en campo, especialmente a Eunice Raquel Avendaño Zavaleta (UMAR) y a Marlon Federico Cortes Dávila (UDEA). Finalmente agradecemos a 2 revisores anónimos quienes dieron acertadas sugerencias a la versión previa del manuscrito.
Referencias
Allsop, P. G. y Schoolmeesters, P. (2024). All genera of the world: Subfamilies Prototroginae, Cretoscarabaeinae, Dynamonodinae, Electrorubesopsinae, Phaenomeridinae, Orphninae, Allidiostomatinae, Aclopinae, Lichniinae, Melolonthinae, Oncerinae, Podolasiinae, Sericinae, Sericoidinae (Animalia: Arthropoda: Insecta: Coleoptera: Scarabaeidae). Megataxa, 12, 104–175. https://doi.org/10.11646/megataxa.12.
Aragón-García, A., Nochebuena-Trujillo, C. D., Morón, M. A. y López-Olguín, J. F. (2008). Uso de trampas de luz fluorescente para el manejo de la gallina ciega (Coleoptera: Melolonthidae) en maíz (Zea mays L.) Agrociencia, 42, 217–223.
Aragón-García, A., Pérez-Torres, B., Aragón-Sánchez, M., Cuate-Mozo, V. A., López-Olguín, J. F. y Lugo-García, G. L. (2018). Estrategias agroecológicas para el control de gallina ciega en cultivos agrícolas. En M. B. Nájera R. y Aragón-García, G. (Eds.), Diversidad, ecología y manejo de insectos rizófagos (pp. 135–147). Ciudad de México: Instituto Nacional de Investigaciones Forestales, Agrícolas y Pecuarias/ La Benemérita Universidad Autónoma de Puebla.
Barria, M. D., Clavijo-Bustos, J. y Ramírez-Ponce, A. (2022). A new species of Phyllophaga (s. str.) schizorhina species group (Coleoptera: Scarabaeidae: Melolonthinae: Rhizotrogini) from Panama. Faunitaxys, 10, 1–5. https://doi.org/10.57800/faunitaxys-10(58)
Becerra, X. J. y Venable, L. (1999). Nuclear ribosomal DNA and its implications for evolutionary trends in Mexican Bursera (Burseraceae). American Journal of Botany, 86, 1047–1057. https://doi.org/10.2307/2656622
Castro-Ramírez, A. E., Delfín-González, E. H., Parra-Tabla, V. y Morón, M. A. (2005). Fauna de melolóntidos (Coleoptera: Scarabaeoidea) asociados al maíz (Zea mays L.) en los Altos de Chiapas, México. Folia Entomológica Mexicana, 44, 339–365.
Cuate-Mozo, V. A., Aragón-García, A., Pérez-Torres, B. C., López-Olguín, J. F., Morón, M. A. y Rojas-Martínez, R. I. (2016). Manejo del complejo gallina ciega (Coleoptera: Melolonthidae) asociado al cultivo de amaranto (Amaranthus hypochrondriacus L.) en Puebla, México. Agrociencia, 50, 889–900.
Delgado, L., Mora-Aguilar, E. F. y Escobar-Hernández, F. (2012). Scarabaeoidea (Coleoptera) of the municipality of Xalapa, Veracruz, Mexico: Inventory and analysis. The Coleopterists Bulletin, 66, 319–332. https://dx.doi.org/10.1649/072.066.0405
Doskocil, J. P., Walker, N. R., Bell, G. E., Marek, M., Reinert, J. A. y Royer, T. A. (2008). Species composition and seasonal occurrence of Phyllophaga (Coleoptera: Scarabaeidae) infesting intensely managed bermudagrass in Oklahoma. Journal of Economic Entomology, 101, 1624–1632. https://doi.org/10.1603/0022-0493(2008)101[1624:SCASOO]2.0.CO;2
Deloya, C. y Morón, M. A. (1998). Nuevas especies de Phyllophaga Harris (Coleoptera: Melolonthidae) de Morelos, Puebla y Chiapas, México. Folia Entomológica Mexicana, 104, 109–119.
Deloya, C., Morón, M. A. y Lobo, J. M. (1995). Coleoptera Lamellicornia (Macleay, 1819) del sur del Estado de Morelos, México. Acta Zoológica Mexicana (nueva serie), 65, 1–42. https://doi.org/10.21829/azm.1995.65651653
Gutiérrez-Carranza, I. G. (2023). Protocolo para disección y montaje de luciérnagas (Coleoptera: Lampyridae). Boletín de la Asociación Mexicana de Sistemática de Artrópodos, 7, 2–6.
Hernández-Cruz, J., Morón, M. A., Sánchez-García, J. A., Martínez-Martínez, L., Jarquín-López, R. y Cerqueda-Reyes, H. (2016). Listado de especies del género Phyllophaga Harris, 1827 (Coleoptera: Melolonthidae, Melolonthinae) en el estado de Oaxaca. Entomología Mexicana (nueva serie), 3, 924–929.
Kawada, R. y Buffington, M. L. (2016). A scalable and modular dome illumination system for scientific microphotograph on a budget. Plos One, 11, e0153426. https://doi.org/10.1371/journal.pone.0153426
Mateos-Escudero, M., Guzmán-Vázquez, H. M., Lozano-Trejo, S., Sánchez-García, J. A. y Pérez-León, M. I. (2021). White grub adults associated with maize (Zea mays L.) at Zaachila, Oaxaca, Mexico. Southwestern Entomologist, 46, 709–724. https://doi.org/10.3958/059.046.0312
Márquez-Luna, J. (2005). Técnicas de colecta y preservación de insectos. Boletín Sociedad Entomológica Aragonesa, 37, 385–408.
Márquez-Manzano, J. P., Aragón-García, A., Cuate-Mozo, V. A. y Pérez-Torres, B. C. (2024). Diversidad de gallinas ciegas (Coleoptera: Melolonthidae) asociadas a pastos ornamentales de la Ciudad de Puebla, México. Entomología Mexicana, 11, 1–5.
Morón, M. A. (1986). El género Phyllophaga en México. Morfología, distribución y sistemática supraespecífica (Insecta: Coleoptera). Publicación 20. Ciudad de México: Instituto de Ecología.
Morón, M. A. (1992). Nuevas especies mexicanas del subgénero Chlaenobia de Phyllophaga (Coleoptera: Melolonthidae). Giornale Italiano di Entomologia, 6, 35–51.
Morón, M. A. (2006). Revisión de las especies de Phyllophaga (Phytalus) grupos obsoleta y pallida (Coleoptera: Melolonthidae: Melolonthinae). Folia Entomológica Mexicana, 45 (Suplemento 1), 1–104.
Morón, M. A. (2015a). Revisión del nuevo grupo de especies “guapilana” de Phyllophaga (s. str.) (Coleoptera: Melolonthidae: Melolonthinae). Boletín de la Sociedad Entomológica Aragonesa, 56, 27–38.
Morón, M. A. (2015b). Revisión del nuevo grupo de especies “quetzala” de Phyllophaga (s. str.) (Coleoptera: Melolonthidae: Melolonthinae). Elytron, 27, 3–28.
Morón, M. A. (2018). Revisión del grupo de especies “pruinosa” de Phyllophaga (Phytalus) (Coleoptera: Melolonthidae: Melolonthinae). Dugesiana, 25, 37–59.
Morón, M. A. y Terrón, R. A. (1988). Entomología práctica. Ciudad de México: Instituto de Ecología.
Morrone, J. J. (2019). Regionalización biogeográfica y evolución biótica de México: encrucijada de la biodiversidad del Nuevo Mundo. Revista Mexicana de Biodiversidad, 90, e902980. https://doi.org/10.22201/ib.20078706e.2019.90.2980
Morrone, J. J., Escalante, T. y Rodríguez-Tapia, G. (2017). Mexican biogeographic provinces: map and shapefiles. Zootaxa, 4277, 277–279. https://doi.org/10.11646/zootaxa.
4277.2.8
Morón, M. A., Nogueira, G., Rojas-Gómez, C. V. y Arce-Pérez, R. (2014). Biodiversidad de Melolonthidae (Coleoptera) en México. Revista Mexicana de Biodiversidad, 85 (Supl.), 293–302. https://doi.org/10.7550/rmb.31834
Ramírez-Ponce, A., Allende-Canseco, J. y Morón, M. A. (2009). Fauna de coleópteros lamelicornios de Santiago Xiacui, Sierra Norte, Oaxaca, México. Acta Zoológica Mexicana (nueva serie), 25, 323–343. https://doi.org/10.21829/azm.2009.252640
Rivera-Gasperín, S. L. y Morón, M. A. (2013). Análisis filogenético del subgénero Phyllophaga (Triodonyx) (Coleoptera: Melolonthidae: Melolonthinae). Revista Mexicana de Biodiversidad, 84, 802–817. https://doi.org/10.7550/rmb.34034
Rivera-Gasperín, S. L. y Morón, M. A. (2017a). Phylogenetic relationships within Phyllophaga Harris (sensu lato) (Coleoptera: Melolonthidae, Melolonthinae) with emphasis on Listrochelus Blanchard. Neotropical Entomology, 46, 1–13. https://doi.org/10.1007/s13744-017-0482-6
Rivera-Gasperín, S. L. y Morón, M. A. (2017b). Relaciones filogenéticas de las especies de Chlaenobia con otros miembros de Phyllophaga s. lato (Coleoptera: Melolonthidae: Melolonthinae). Revista Mexicana de Biodiversidad, 88, 592–607. https://doi.org/10.1016/j.rmb.2017.07.011
Romero-López, M. y Morón, M. A. (2017). Dos nuevas especies de Coleoptera Melolonthidae de la Costa Grande de Guerrero, México. Southwestern Entomologist, 42, 889–900. https://doi.org/10.3958/059.042.0327
Rzwedowski, J. (1978). Vegetación de México. Ciudad de México: Editorial Limusa.
Villaseñor, J. L. (1990). The genera of Asteraceae endemic to Mexico and adjacent regions. Aliso, 12, 685–692.
A new gall wasp species (Hymenoptera: Cynipidae), Andricus protector Pujade Villar and Cuesta-Porta sp. nova (Hymenoptera: Cynipidae: Cynipini), from Mexico
Una especie nueva de avispa agalladora (Hymenoptera: Cynipidae), Andricus protector Pujade-Villar and Cuesta-Porta sp. nova (Hymenoptera: Cynipidae: Cynipini) de México
Juli Pujade-Villar a, Ricardo Clark-Tapiab, Victor Aguirre-Hidalgo c, George Melikac, Víctor Cuesta-Portaa, *
a Universitat de Barcelona, Facultat de Biologia, Departament de Biologia Evolutiva, Ecologia i Ciències Ambientals, Avda. Diagonal 645, 08028-Barcelona, Spain
b Universidad de la Sierra Juárez, Laboratorio de Estudios Ambientales, Cam. a la Universidad s/n, 68725 Ixtlán de Juárez, Oaxaca, Mexico
c National Food Chain Safety Office, Plant Health Diagnostic National Reference Laboratory, Keleti Károly u. 24, 1024, Budapest, Hungary
*Corresponding author: victorcp93@gmail.com (V. Cuesta-Porta)
Received: 20 January 2025; accepted: 20 August 2025
http://zoobank.org/urn:lsid:zoobank.org:pub:510B361C-ED79-48EB-A484-9100684E3D92
Abstract
A new species of oak gall wasp, Andricus protector Pujade-Villar and Cuesta-Porta sp. nova, known only from its asexual generation that induces deciduous galls on buds of Quercus crassifolia Humb. and Bonpl. (Lobatae section), is described from Mexico. Diagnosis, distribution and biological data of the new species are given. The validity of Andricus species from Mexico is commented. Four species are proposed here to have dubious affiliation: Andricus burnetti (Dailey and Sprenger, 1983), A. marmoreus Kinsey, 1920, A. setifer (Karsch, 1880) and A. strues (Kinsey, 1938).
Keywords: Hymenoptera; Cynipidae; Andricus; New species; Mexico; Quercus crassifolia
Resumen
Se describe una especie nueva de avispa agalladora, Andricus protector Pujade-Villar y Cuesta-Porta sp. nova de México, conocida solo por su generación asexual, que induce agallas deciduas en yemas de Quercus crassifolia Humb. y Bonpl. (sección Lobatae). Se proporcionan la diagnosis, los datos de distribución y biológicos de la nueva especie. Se comenta la validez de las especies de Andricus de México. Cuatro especies tienen afiliación dudosa: Andricus burnetti (Dailey y Sprenger, 1983), A. marmoreus Kinsey, 1920, A. setifer (Karsch, 1880) y A. strues (Kinsey, 1938).
Palabras clave: Hymenoptera; Cynipidae; Andricus; Especie nueva; México; Quercus crassifolia
Introduction
Oak gall wasps (Hymenoptera: Cynipidae: Cynipini) represent the most diverse group within the Cynipidae family, comprising over 1,000 described species in 59 genera (Buffington et al., 2020; Melika, Pujade-Villar et al., 2021; Ronquist et al., 2015; Stone et al., 2002). These wasps induce galls on Fagaceae, primarily on oaks (Quercus L.). In America, north of Mexico, nearly 500 cynipid species have been recorded (Burks, 1979) in association with 90 species of Quercus (Nixon, 2008). In contrast, Europe has approximately 150 described species of cynipids (Melika 2006), despite having only about 30 Quercus species (Uotila, 2011). More specifically, only in the Iberian Peninsula, about 70 cynipid species associated with 10 Quercus species have been documented (Nieves-Aldrey, 2001). Under the same rule-of-thumb, the Mexican oak gall wasp fauna is likely extraordinarily rich, with many species yet to be described, as Mexico is home to 135-161 Quercus species, 86 of which are endemic (Nixon, 1993a, b; Zavala, 1998; Valencia, 2004).
The recorded number of Mexican cynipid species has varied considerably over time. Pujade-Villar et al. (2009) listed 157 cynipid species, with 42 species attributed to the genus Andricus Hartig, 1840. In a subsequent review, Pujade-Villar and Ferrer-Suay (2015) increased the number of Mexican cynipid species to 183, and 43 species of Andricus. More recently, Martínez-Romero et al. (2022) listed 205 species, 30 of which were assigned to Andricus. The genus Andricus is the most diverse, with more than 400 species described worldwide (Melika, Nicholls et al., 2021; Stone et al.,2002), and one of the most taxonomically complex among oak gall wasps.
Since the review by Pujade-Villar et al. (2009), several species previously classified under Andricus have been reassigned to other genera, including Disholandricus Melika, Pujade-Villar and Nicholls, 2021, Dros Kinsey, 1937, Druon Kinsey, 1937, Erythres Kinsey, 1937, Femuros Kinsey, 1937, Feron Kinsey, 1937, Protobalandricus Melika, Nicholls and Stone, 2018, Striatoandricus Pujade-Villar, 2020, and Trichoteras Ashmead, 1897. In Mexico, a total of 31 species of Andricus are listed (Martínez-Romero et al., 2022), later also A. cylindratum (Kinsey, 1937), A. tecturnarum Kinsey, 1920, A. tibialis Kinsey, 1937 (= A. tostum Kinsey, 1937) (= A. uterinus Kinsey, 1937), A. vitreus Kinsey, 1937 (= A. validus Kinsey, 1937) and A. verutus Kinsey, 1937 were transferred to Feron by Cuesta-Porta et al. (2023). Furthermore, since Martínez-Romero et al. (2022), 2 additional Andricus species have been described from Mexico, A. coombesi Pujade-Villar and Pérez-Torres, 2024 and A. mazahua García-Martinón and Pujade-Villar, 2024 (Pujade-Villar et al., 2024, García-Martiñón et al., 2024), bringing the current number of Andricus species in Mexico to 25. Among these, 18 species induce galls on branches, 4 attack buds (rarely leaves or petioles), 1 is found exclusively on leaves, and 1 in acorns. The host of A. aztecus remains unknown, although it is suspected to induce tuberous galls (Martínez-Romero et al.,2022). The new species described here attacks the buds of Q. crassifolia (Lobatae section), producing a distinctive spherical gall with the larval chamber surrounded by longitudinal air chambers divided by thin partitions of spongious tissue.
Materials and methods
Asexual adult gall wasps (ŏ) of the new species described herein were extracted dead from galls on Quercus crassifolia in Oaxaca (Mexico) years after being collected. They were preserved in 100% ethanol in the laboratory by the second author.
The description of the new species follows the current terminology of morphological structures specific for Cynipidae (Liljeblad & Ronquist, 1998; Melika, 2006) and more general sources such as the Hymenoptera Anatomy Ontology Portal (Yoder et al., 2010). Abbreviations for the fore wing venation follow Ronquist and Nordlander (1989); cuticular surface terminology follows that of Harris (1979). Measurements and abbreviations used here include: F1-F11, 1st and subsequent flagellomeres; POL (postocellar distance), the distance between the inner margins of the posterior ocelli; OOL (ocellar-ocular distance), the distance from the outer edge of a posterior ocellus to the inner margin of the compound eye; LOL, the distance between lateral and frontal ocelli. The width of the fore wing radial cell is measured from the margin of the wing to the Rs vein.
The scanning electron microscope (SEM) pictures were taken by the first author at the University of Barcelona (UB) using a field-emission gun environmental scanning electron microscope (FEI Quanta 200 ESEM), with low-resolution imaging without gold-coating the specimens. The photographs of the galls and habitus were taken by the second author with a Canon PowerShot SX510 HS digital camera and a digital camera mounted on Carl Zeiss microscopy III followed by processing with GIMP 2.8 (GNU Image Manipulation Program), respectively.
The type material of the new species is deposited in the University of Barcelona (UB), Catalonia (J. Pujade-Villar coll.).
Andricus protector Pujade-Villar and Cuesta-Porta sp. nova
(Figs. 1-3)
http://zoobank.org/urn:lsid:zoobank.org:act:D806CCCD-2240-4F39-AF38-94D670D404A5
Diagnosis. The new species differs from all Mexican Andricus species by the following combination of characters: mesoscutum transversally rugose; median mesoscutal line present, with smooth bottom; margin of the fore wing shortly ciliated; second metasomal tergum smooth anteriorly, anterolaterally with dense white setae, and a band of punctures in the posterior third; subsequent terga and hypopygium punctuate, without setae. Andricus protector is closely related to Andricus formosalis Weld, 1944 (Fig. 3B), but differs from it by the following characters: antenna and legs brown to black (yellowish in A. formosalis); inner margins of eyes parallel (converging ventrally in A. formosalis); head quadrate and slightly broader than the mesosoma (triangular, narrower than the mesosoma in A. formosalis); F1 1.2 × as long as F2 (only slightly longer in A. formosalis); placodeal sensilla inconspicuous (present in A. formosalis); pronotum completely rugose (with parallel ridges in the posterior margin in A. formosalis); median mesoscutal line long and wide (short and becoming almost inconspicuous in A. formosalis); mesoscutellar foveae quadrate, separated by a carina, not delimited posteriorly (triangular, widely separated and well-delimited posteriorly in A. formosalis); 2nd metasomal tergum occupying most of the metasoma (very short in A. formosalis). Also, the galls of both species are different: with a velvety reddish surface when young and spherical when mature in A. proctector sp. nova, and with a velvety whitish surface when young and ovoid when mature in A. formosalis.
The new species can be morphologically similar to other Andricus inducingtuberous galls in branches. Despite the great divergence in gall morphology between A. protector and the tuberous gallers, Andricus guanajuatensis, A. montezumus, A. peredurus and A. protector have the mesoscutum transversally carinate, whilst the rest of Mexican species that induce tuberous galls have the mesoscutum strongly wrinkled. Andricus protector has a wide band of punctuation in the second metasomal tergum and a medial mesoscutal line (both characters absent in A. guanajuatensis, A. montezumus, and A. peredurus).
Description. Asexual female. Body black (Fig. 3A); mandibles brown, maxillary and labial palpi light brown; antenna brown, scape black; tegula dark brown to black; legs black, tibiae I and II and all tarsomeres brown; metasoma chestnut brown to black; fore wing veins light.
Head (Fig. 1A-D) 1.3 × as broad as high and very slightly broader than mesosoma in frontal view, 2.1 × as broad as long in dorsal view. Gena coriaceous, broadened behind eye, around 1.4 × as broad as transverse diameter of eye; malar space with striae radiating from clypeus and reaching eye; eye 3.1 × as high as length of malar space. Inner margins of eyes parallel. POL 2.0× as long as OOL, OOL 1.3 × as long as diameter of lateral ocellus, 1.3 × as long as LOL, ocelli rounded, all 3 equal in size. Transfacial distance as long as height of eye and 1.35 × as long as height of lower face (distance between antennal rim and ventral margin of clypeus); diameter of antennal torulus 3.3 × as long as distance between them, distance between torulus and eye as long as diameter of torulus. Lower face coriaceous, setose, with some irregular weak striae, with elevated median area coriaceous, with irregular weak striae, without setae. Clypeus quadrate, flat, carinated, ventrally slightly curved, not emarginate and slightly incised medially; with anterior tentorial pit, epistomal sulcus and clypeo-pleurostomal line distinct. Frons, interocellar area and vertex strongly coriaceous, with some weak rugae between ocelli and eye; occiput carinate, with few short setae; postgena punctuate, pubescent; postocciput and postocciput around occipital foramen weakly carinate, glabrous; posterior tentorial pit oval; hypostomal carina emarginate, present at the basis of postgenal sulci, gular sulci present; occipital foramen longer than height of postgenal bridge.
Antenna (Fig. 1E, F) as long as head + mesosoma, with 13 flagellomeres, sometimes with a partial suture towards the middle of F12, rarely F13 fused; pedicel 1.6 × as long as broad, F1 0.8 × as long as length of scape + pedicel and 1.25 × as long as F2, F3 0.8 × as long as F2, F3 as long as F4, F5 as long as F6, F7 slightly shorter than F6 and equal as F8, F9 slightly shorter than F8 and equal as F10, F11 slightly shorter than F10, F12 + 13 shorter than F10 + F11, F12 as long as F11 and slightly longer than F13; placodeal sensilla inconspicuous on F5 – F12, absent on F1 – F4. Antennal formula: 14:11 x 7: 20: 16: 13: 13: 12: 12. 11: 11: 10: 10: 9: 9: 8.
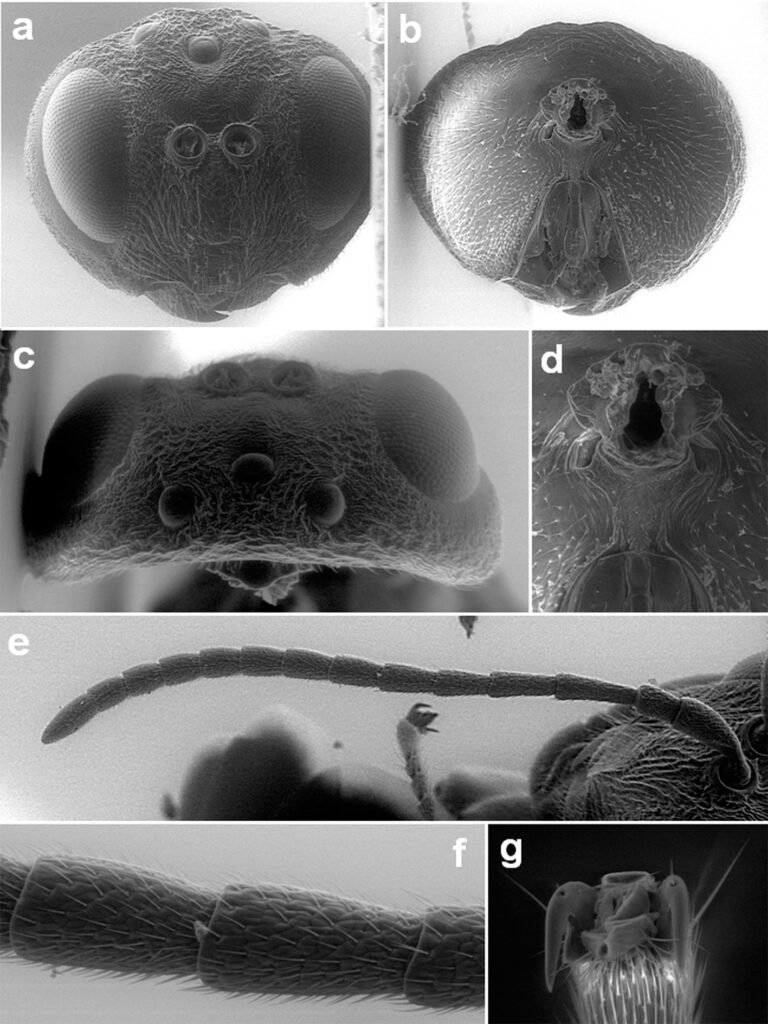
Mesosoma (Fig. 2A-C) slightly longer than high in lateral view. Pronotum rugose with coriaceous interspaces, glabrous, emarginate along lateral and dorsal edges. Propleuron alutaceous with weak irregular rugae and sparse setae. Mesoscutum rugose, with coriaceous interspaces, glabrous, broader than long (width measured across base of tegulae). Notaulus complete; median mesoscutal line present, extends 1/3 of mesoscutum length, with smooth bottom; anterior parallel line impressed, extends to half of mesoscutum length, coriaceous, glabrous; parapsidal line incised and surrounded by coriaceous surface, extends 2/3 of mesoscutum; parascutal carina narrow, coriaceous, reaching notaulus. Transscutal articulation deep, distinct. Mesoscutellum quadrate, shorter than mesoscutum, uniformly rugose, slightly overhanging metanotum; mesoscutellar foveae big, quadrate, with smooth to alutaceous bottom, open basally with some rugae, distinctly divided by a central carina with coriaceous sculpture. Mesopleuron including speculum rugose-carinate, with coriaceous interspaces, sparsely pubescent; mesopleural triangle coriaceous-rugose, with sparse white setae. Metapleural sulcus reaching mesopleuron above half its height, upper part of sulcus inconspicuous; dorsal axillar area rugose, glabrous; lateral axillar area and axillar carina weakly carinated, glabrous; subaxillular bar with parallel sides smooth, glabrous; metanotal trough smooth, setose. Metascutellum sub-rectangle coriaceous, glabrous, ventral impressed area smooth. Lateral propodeal carinae distinct, broad, parallel but basally curved towards the nucha; central propodeal area coriaceous, glabrous, without central longitudinal carina, with some rugae; lateral propodeal area coriaceous rugose, glabrous; nucha with delicate rugae.
![Figure 3. Lateral habitus of a) Andricus protector sp. nova., b) A. formosalis [Picture obtained from Type Search of Smithsonian Entomological Collections, http://n2t.net/ark:/65665/m36fc57b94-797e-4d41-b88c-58776f4ed1d3]](https://rev-mex-biodivers.mx/wp-content/uploads/5448-Fig_3-12cm-921x1024.jpg)
Fore wing (Fig. 4A) slightly longer than body, hyaline, with the margin shortly ciliate; radial cell open, 3.0 × as long as broad, R1 and Rs not reaching the wing margin; areolet present, large; Rs + M visible in 2/3 of its length, its projection reaching basal vein slightly below half of its height.
Legs with a reticulate sculpture. Tarsal claws with a basal lobe (Fig. 1G).
Metasoma (Fig. 2D, E) shorter than head + mesosoma, longer than high in lateral view, 2nd metasomal tergum smooth anteriorly with dense white setae anterolaterally and a band of punctures in the posterior third; subsequent terga and hypopygium punctuate, without setae; prominent part of the ventral spine of the hypopygium 5.0 × as long as broad in ventral view, with short and sparse white setae, without apical tuft. Body length 3.7-3.8 mm (n = 3).
Gall (Fig. 4B, C). Unilocular, spherical gall located on the axillary buds of Q. crassifolia, in clusters of 2-4, with average size of 11.6 mm (SD ± 0.46 mm, N = 20). Light yellowish red with short pubescent surface when young, turning brown and glabrous when mature. Inside a single larval cell located in the center, surrounded by multiple radiating empty chambers. All chambers are surrounded by spongy tissue. The consistency of the gall is not very hard.
Taxonomic summary
Type material. Holotype: asexual female “MEX, La Resinera, Santa Catarina Ixtepeji (Ixtepec, Oaxaca), Q. crassifolia, (30.vii.2021) extr. 18.v.24”, R. Clark leg. (black label); “holotype Andricus protector Pujade-Villar and Cuesta-Porta n. sp., desig. JP-V 2023” (red label). Paratypes: 3 asexual females with same data as the holotype. Holotype and 3 paratypes are deposited in the collection of JP-V (University of Barcelona, UB), one of them dissected for SEM imaging.
Etymology. Named for the species’ gall model in which the larval chamber is protected (surrounded) by multiple empty chambers.

Host plant. Known only from Q. crassifolia (Lobatae section). This oak species is distributed in Mexico (Chiapas, Chihuahua, Guerrero, Guanajuato, Hidalgo, Jalisco, Estado de México, Michoacán, Oaxaca, Puebla, Querétaro, San Luis Potosí, Tlaxcala, Veracruz, and Zacatecas) and Guatemala. Distribution. Mexico (Oaxaca).
Biology. Only the asexual generation is known. The galls appear in mid-June and fall to the ground between August and September, rarely in October. Once in the ground, the galls absorb a lot of soil moisture during the rainy season, which explains why they were very soft when they were collected. This might have affected the development of the adults and prevented them from emerging. The extracted adults were dead, but perfectly preserved.
Discussion
Of the total of 25 Andricus species known from Mexico (Table 1), Andricus burnetti (Dailey & Sprenger, 1983) was described in the genus Trichoteras and transferred to Andricus by Melika and Abrahamson (2002), it is associated with oaks of the Protobalanus section which matches with most Trichoteras species, however the gall differs from the rest of Trichoteras and is more similar to Feron. Andricus marmoreus Kinsey, 1920 has a gall similar to those of Disholcaspis, but the adult has simple tarsal claws and an unusual mesoscutal sculpture for a Mexican species of Disholcaspis. The gall of A. setifer (Karsch, 1880) may correspond to some of the genus Striatoandricus. Lastly, A. strues (Kinsey, 1938) was originally described in the genus Conobius Kinsey, 1938, which was later synonymized under Andricus by Melika and Abrahamson (2002), but unlike what is mentioned in the original description of Conobius, A. strues has the margin of its fore wings ciliated. Concerning Callirhytis quercusbatatoides, Pujade-Villar et al. (2014) mentioned that this species probably belongs to Andricus; later, in Pujade-Villar and Ferrer-Suay (2015), it is considered as an Andricus species but the formal change is not indicated; and finally, in Martínez-Romero et al. (2022) it is definitively named as Andricus quercusbatatoides (Ashmead, 1881).
Among the rest of Andricus species reported from Mexico, the gall morphology and host association is still remarkably diverse (Table 1): one species attacks acorns, 4 induce galls on leaves and/or buds, 3 attack branches without causing globular tuberous galls and 13 induce tuberous galls. The gall of A. aztecus (Cameron, 1888) is unknown, although it may induce tuberous galls.
The alternating life cycle is only known in one of the species (A. quercuslaurinus); the rest remain known by their asexual form except in A. protuberans and A. sphaericus, of which only the sexual generation is known. The Mexican Andricus induce galls in the sections Quercus, Lobatae, Virentes, and Protobalanus (Martínez-Romero et al., 2022; Table 1). The species described here corresponds to an asexual form inducing galls in Lobatae section.
Andricus species with linear elements in the mesoscutum (wrinkles or carinae) are restricted to tuberous gall specie (except A. mazahua). This trait is also present in the species here described, but A. protector does not induce tuberous galls. Among the Mexican Andricus species that produce galls on buds, A. burnetti is associated with Q. palmeri (Protobalanus section), while A. protuberans, A. rochai, and A. strues are associated with species in the Quercus section. Although A. protuberans has also been reported on oaks from the Lobatae section, Cuesta-Porta, Cibrián-Tovar et al. (2022) suggested that these records require further confirmation. Andricus protector has a very peculiar gall containing a set of empty chambers radially surrounding the central larval chamber. The gall of the newly described species is very similar to the gall of A. formosalis known from Arizona also associated with the Lobatae section, but the adults are morphologically different (see Diagnosis and Fig. 3).
Table 1
Andricus species reported in Mexico after this study, indicating the known generation of their lifecycle (Known generation), the plant organ they attack (Plant organ), and the host oak association (Host section). Species marked with an asterisk are considered of unclear affiliation in the Discussion section.
| Species | Authors | Known generation | Plant organ | Host section | |
| Andricus | aztecus | (Cameron, 1888) | Asexual | Unknown | Unknown |
| Andricus | bonanseai | Mayr, 1905 | Asexual | Branch (tuberous) | Lobatae, Quercus |
| Andricus | breviramuli | Pujade-Villar, 2014 | Asexual | Branch (non-tuberous) | Quercus |
| Andricus | burnetti * | (Dailey and Sprenger, 1983) | Asexual | Buds/leaf | Protobalanus |
| Andricus | carrilloi | Pujade-Villar, 2013 | Asexual | Branch (tuberous) | Quercus |
| Andricus | coombesi | Pujade-Villar and Pérez-Torres, 2024 | Asexual | Acorn | Lobatae |
| Andricus | dugesi | Beutenmüller, 1917 | Asexual | Branch (tuberous) | Lobatae, Quercus |
| Andricus | durangensis | Beutenmüller, 1911 | Asexual | Branch (tuberous) | Lobatae |
| Andricus | furnaceus | Kinsey, 1920 | Asexual | Branch (tuberous) | Quercus |
| Andricus | fusiformis | Pujade-Villar, 2014 | Asexual | Branch (non-tuberous) | Lobatae, Quercus |
| Andricus | guanajuatensis | Pujade-Villar, 2013 | Asexual | Branch (tuberous) | Lobatae, Quercus |
| Andricus | marmoreus * | Kinsey, 1920 | Asexual | Branch (non-tuberous) | Quercus |
| Andricus | mazahua | García-Martinón and Pujade-Villar, 2024 | Asexual | Branch (tuberous) | Lobatae |
| Andricus | montezumus | Beutenmüller, 1913 | Asexual | Branch (tuberous) | Quercus |
| Andricus | peredurus | Kinsey, 1920 | Asexual | Branch (tuberous) | Quercus |
| Andricus | protector | Pujade-Villar & Cuesta-Porta, sp. nova. | Asexual | Buds/leaf | Lobatae |
| Andricus | protuberans | Pujade-Villar and Ferrer-Suay, 2015 | Sexual | Buds/leaf | Lobatae |
| Andricus | quercusbatatoides | (Ashmead, 1881) | Asexual | Branch (tuberous) | Virentes |
| Andricus | quercuslaurinus | Melika and Pujade-Villar, 2009 | Sex. and asex. | Branch (non-tuberous) | Lobatae |
| Andricus | rochai | Pujade-Villar, 2018 | Asexual | Buds/leaf | Quercus |
| Andricus | santafe | Pujade-Villar, 2013 | Asexual | Branch (tuberous) | Quercus |
| Andricus | setifer * | (Karsch, 1880) | Asexual | Branch (non-tuberous) | Unknown |
| Andricus | sphaericus | Pujade-Villar, 2016 | Sexual | Buds/leaf | Quercus |
| Andricus | strues * | (Kinsey, 1938) | Asexual | Buds/leaf | Quercus |
| Andricus | tumefaciens | Pujade-Villar and Paretas-Martínez, 2012 | Asexual | Branch (tuberous) | Lobatae, Quercus |
| Andricus | tumeralis | Pujade-Villar, 2009 | Asexual | Branch (tuberous) | Quercus |
Air chambers in galls are hypothesized to act as a defensive mechanism against parasitoids by hindering oviposition or confusing parasitoids simulating larval chambers and (Pujade-Villar et al., 2025; Stone & Cook 1998; Stone et al., 2002). Several independent cynipid lineages have converged into gall morphologies with hollow internal spaces in the parenchyma, such as the galls of Atrusca, Kinseyella, and some Amphibolips in the Nearctic and Trichagalma in Eastern Palearctic, usually exhibit radiating filaments that connect the larval chamber with the outer shell of the gall; or less common cases of free rolling larval chambers like some Disholcaspis or Belizinella (Cuesta-Porta et al.,2020, 2024, 2025; Ide & Koyama, 2023; Pujade-Villar et al., 2010). In the case of A. protector and A. formosalis, galls have hollow longitudinal chambers divided by longitudinal parenchymatous partitions. Despite the gall similarity, the adults of A. protector and A. formosalis exhibit significant differences (see Diagnosis). Further research is needed to determine the phylogenetic relationships between A. protector and the rest of Andricus species. The generic limits of the American Andricus species are still under revision and more data is needed to confirm that the similarities between A. protector and A. formosalis are phylogenetically informative or the result of morphological convergence.
Acknowledgements
We thank Denis Brothers (School of Biological and Conservation Sciences, University of KwaZulu-Natal, Pietermaritzburg, South Africa) for his help with a ZNC question. This research was funded by the project “PID2021-128146NBI00/MCIN/AEI/10.13039/501100011033/” and “FEDER una manera de hacer Europa” from the Ministry of Science and Innovation of Spain and the European Region al Development Fund (ERDF). Also is supported by UNSIJ 2-EA-2203.
References
Buffington, M. L., Forshage, M., Liljeblad, J., Tang, C. T., & van Noort, S. (2020). World Cynipoidea (Hymenoptera): A key to higher-level groups. Insect Systematics and Diversity, 4, 1–69. https://doi.org/10.1093/isd/ixaa003
Burks, B. D. (1979). Superfamily Cynipoidea. In K.V. Krombein, P. D. Hurd Jr., D. R. Smith, & B. D. Burks (Eds.), Catalog of Hymenoptera in America North of Mexico. Vol. 1. (pp. 1045–1107). Symphyta and Apocrita. Washington D.C.: Smithsonian Institution Press.
Cuesta-Porta, V., Equihua-Martínez, A., Estrada-Venegas, E. G., Cibrián-Tovar, D., Barrera-Ruiz, U. M., Ordaz-Silva, S. et al. (2020). Revision of the Amphibolips species of the “nassa” complex from Mexico and Central America (Hymenoptera: Cynipidae). Zootaxa, 4877, 1–50. https://doi.org/10.11646/zootaxa.4877.1.1
Cuesta-Porta, V., Cibrián-Tovar, D., Barrera-Ruiz, U. M., Melika, G., & Pujade-Villar, J. (2022). Andricus protuberans Pujade-Villar and Ferrer-Suay is a sexual form (Hym., Cynipidae, Cynipini). Folia Entomológica Mexicana (nueva serie), 8, e0081004. https://doi.org/10.53749/fem.0081004
Cuesta-Porta, V., Melika, G., Nicholls, J. A., Stone, G. N., & Pujade-Villar, J. (2023). Re-establishment of the Nearctic oak cynipid gall wasp genus Feron Kinsey, 1937 (Hymenoptera: Cynipidae: Cynipini), including the description of six new species. Zootaxa, 5366, 001–174. https://doi.org/10.11646/zootaxa.5366.1.1
Cuesta-Porta, V., Cooke-Mcewen, C., Melika, G., Romero-Rangel, S., Equihua-Martínez, A., Estrada-Venegas, E. G. et al. (2024). Review of the Mexican species of Disholcaspis Dalla Torre & Kieffer with the description of eleven new species. Zootaxa, 5498, 1–100. https://doi.org/10.11646/zootaxa.5498.1.1
Cuesta-Porta, V., Melika, G., Ferrer-Suay, M., Vera-Ortiz, A., & Pujade-Villar, J. (2025). Review of the Nearctic and Neotropical genus Atrusca Kinsey, 1930 (Hymenoptera: Cynipidae, Cynipini). Zootaxa, 5617, 1–195. https://doi.org/10.11646/zootaxa.5617.1.1
García-Martiñón, R. D., Equihua-Martínez, A., Estrada-Venegas, E. G., & Pujade-Villar, J. (2024). Una nueva especie mexicana de Andricus inductora de agallas tumorales en Quercus crassipes. Southwestern Entomologist, 49, 1–11.
Harris, R. (1979). A glossary of surface sculpturing. State of California, Department of Food and Agriculture, Occasional Papers in Entomology, 28, 1–31.
Ide, T., & Koyama, A. (2023). The formation of a rolling larval chamber as the unique structural gall of a new species of cynipid gall wasps. Scientific Reports, 13, 18149. https://doi.org/10.1038/s41598-023-43641-6
Liljeblad, J., & Ronquist, F. (1998). A phylogenetic analysis of higher-level gall wasp relationships (Hymenoptera: Cynipidae). Systematic Entomology, 23, 229–252. https://doi.org/10.1046/j.1365-3113.1998.00053
Martínez-Romero, A., Cuesta-Porta, V., Equihua-Martínez, A., Estrada-Venegas, E. D., Barrera-Ruiz, U. M., Cibrián-Tovar, D. et al. (2022). Aportación al conocimiento de las especies de Cynipini (Hymenoptera: Cynipidae) en los estados mexicanos. Revista Mexicana de Biodiversidad, 93, e933998. https://doi.org/10.22201/ib.20078706e.2022.93.3998
Melika, G. (2006) Gall wasps of Ukraine. Cynipidae. Vestnik Zoologii, 21, 1–300.
Melika, G. & Abrahamson, W. G. (2002). Review of the world genera of oak cynipid wasps (Hymenoptera: Cynipidae: Cynipini). In G. Melika, & C. Thuróczy (Eds.), Parasitic wasps: evolution, systematics, biodiversity and biological control (pp. 150–190). Budapest: Agroinform.
Melika, G., Nicholls, J. A., Abrahamson, W. G., Buss, E. A., & Stone, G. N. (2021). New species of Nearctic oak gall wasps (Hymenoptera: Cynipidae, Cynipini). Zootaxa,
5084, 001–131. https://doi.org/10.11646/zootaxa.5084.1.1
Melika, G., Pujade-Villar, J., Nicholls, J. A., Cuesta-Porta, V., Cooke-McEwen, C., & Stone, G. N. (2021). Three new Nearctic genera of oak cynipid gall wasps (Hymenoptera: Cynipidae: Cynipini): Burnettweldia Pujade-Villar, Melika and Nicholls, Nichollsiella Melika, Pujade-Villar and Stone, Disholandricus Melika, Pujade-Villar and Nicholls; and re-establishment of the genus Paracraspis Weld. Zootaxa, 4993, 1–81. https://doi.org/10.11646/zootaxa.4993.1.1
Nieves-Aldrey, J. L. (2001). Fauna Ibérica, Vol. 16. Hymenoptera: Cynipidae. Madrid: Consejo Superior de Investigaciones Científicas.
Nixon, K. C. (1993a). The genus Quercus in Mexico. In R. Bye, T. P. Ramamoorthy, A. Lot, & J. Fay (Eds.), Biological diversity of Mexico: origins and distribution (pp. 447–458). Nueva York: Oxford University Press.
Nixon, K. C. (1993b). Infrageneric classification of Quercus (Fagaceae) and typification of sectional names. Annales des Sciences Forestières, 50, 25–34. https://doi.org/10.1051/forest:19930701
Nixon, K. C. (2008). Quercus. In Flora of North America @ eFloras. Missouri Botanical Garden, St. Louis, MO y Harvard University Herbaria, Cambridge, MA. Retrieved on September 15, 2020 from: http://www.efloras.org/florataxon.aspx?flora_id=1andtaxon_id=127839
Pujade-Villar, J., Romero-Rangel, S., Chagoyán-García, C., Equihua-Martínez, A., & Estrada-Venegas, E. G. (2010). A new genus of oak gallwasps, Kinseyella Pujade-Villar & Melika, with a description of a new species from Mexico (Hymenoptera: Cynipidae: Cynipini). Zootaxa, 2335, 16–28. https://doi.org/10.11646/zootaxa.2335.1.2
Pujade-Villar, J., Equihua.Martínez, A., & Estrada-Venegas, E. G. (2014). Actualización del conocimiento de los Cynipidae mexicanos que producen agallas en encinos (Hymenoptera: Cynipidae: Cynipini). Entomología Mexicana, 13, 559–564.
Pujade-Villar, J., & Ferrer-Suay (2015). Adjudicació genèrica d’espècies mexicanes d’ubicació dubtosa descrite2s per Kinsey i comentaris sobre la fauna mexicana (Hymenoptera: Cynipidae: Cynipini). Butlletí de la Institució Catalana d’Historia Natural, 79, 7–14.
Pujade-Villar, J., Equihua-Martínez, A., Estrada-Venegas, E. G., & Chagoyán-García, C. (2009). Estado del conocimiento de los Cynipini (Hymenoptera: Cynipidae) en México: perspectivas de estudio. Neotropical Entomology, 38, 809–821. https://doi.org/10.1590/S1519-566X2009000600015
Pujade-Villar, J., Pérez-Torres, B. C., Coombes, A. J., Aragón-García, A., Rodríguez-Acosta, M., López-Olguin, J. F. et al. (2024). Description of the first species of gall wasp (Hym., Cynipidae: Cynipini) on Quercus grahamii (Fagaceae). Zootaxa,5403, 369–376. https://doi.org/10.11646/zootaxa.5403.3.6
Pujade-Villar, J., Melika, G., & Cuesta-Porta, V. (2025). Structure and function of oak gall wasps. In D. C. de Oliveira, & R. M. S. dos Santos Isaias (Eds), Plant galls (pp. 313–347). Cham, Switzerland: Springer.
Ronquist, F., Nieves-Aldrey, J. L., Buffington, M. L., Liu, Z., Liljeblad, J., & Nylander, J. A. A. (2015). Phylogeny, evolution and classification of gall wasps: the plot thickens. Plos One, 10, 1–40. https://doi.org/10.1371/journal.pone.0123301
Ronquist, F., & Nordlander, G. (1989). Skeletal morphology of an archaic cynipoid, Ibalia rufipes (Hymenoptera: Ibaliidae). Entomologica Scandinavica, 33, 1–60
Stone, G. N., & Cook, J. M. (1998). The structure of cynipid oak galls: patterns in the evolution of an extended phenotype. Proceedings of the Royal Society of London. Series B: Biological Sciences, 265,979–988. https://doi.org/10.1098/rspb.1998.0387
Stone, G. N., Schönrogge, K., Atkinson, R. J., Bellido, D., & Pujade-Villar, J. (2002). The population biology of oak gall wasps (Hymenoptera: Cynipidae). Annual Review of Entomology, 47, 633–668. https://doi.org/10.1146/annurev.ento.47.091201.145247
Uotila P. (2011). Quercus. In Euro + Med Plantbase-the information resource for Euro-Mediterranean plant diversity. Retrieved on November 12, 2024 from: http://www.europlusmed.org
Valencia, A. S. (2004). Diversidad del género Quercus (Fagaceae) en México. Boletín de la Sociedad Botánica de México, 75, 33–53. https://doi.org/10.17129/botsci.1692
Yoder, M. J., Mikó, I., Seltmann, K. C., Bertone, M. A., & Deans, A. R. (2010). A gross anatomy ontology for Hymenoptera. Plos One, 5, e15991. https://doi.org/10.1371/journal.pone.0015991
Zavala, F. (1998). Observaciones sobre la distribución de encinos en México. Polibotánica, 8, 47–64.


